Clear Sky Science · en
Single-cell imaging analysis, therapeutic modeling and a Phase Ib trial validate BCL-2 as a target across heterogeneous castration-resistant prostate cancer
Why this research matters
Many men with advanced prostate cancer eventually face a stage where standard hormone-blocking drugs stop working. This study asks a simple but urgent question: when tumors learn to live without male hormones, do they all rely on a common “survival switch” that doctors can target? By tracking individual cancer cells, testing drugs in models, and running an early clinical trial, the researchers pinpoint one such switch—a protein called BCL-2—and show that blocking it may help treat otherwise resistant disease.
The problem with stubborn prostate tumors
Modern drugs that shut down male hormones, or block their receptor, have extended lives for many men. Yet thousands still die every year from metastatic castration-resistant prostate cancer, a form that grows despite these treatments. One key reason is variety: within a single tumor, some cells depend heavily on hormone signals, while others barely use them at all. When hormone-blocking drugs are given, the sensitive cells die or shrink, but the hardier ones survive, adapt, and eventually dominate. Doctors have long suspected that these survivors rely on backup survival programs, but it has not been clear which programs are shared across different resistant tumor types.
Finding the survival switch in single cells
The team focused on BCL-2, a protein that helps cells avoid programmed death. Using advanced imaging tools that can measure many proteins in thousands of cells at once, they examined benign prostate tissue, untreated primary tumors, and resistant tumors from patients, as well as several mouse models. In healthy glands, hormone receptors and BCL-2 sit in mostly separate cell layers. In untreated cancers, most cells show strong hormone receptors but little BCL-2. After long-term hormone-blocking, however, that pattern flips: resistant cancers become rich in BCL-2–positive cells, including cells that still carry the hormone receptor and those that have largely lost it. Across multiple datasets, BCL-2 activity rose as hormone signaling fell, suggesting a tight, see-saw relationship between the two.
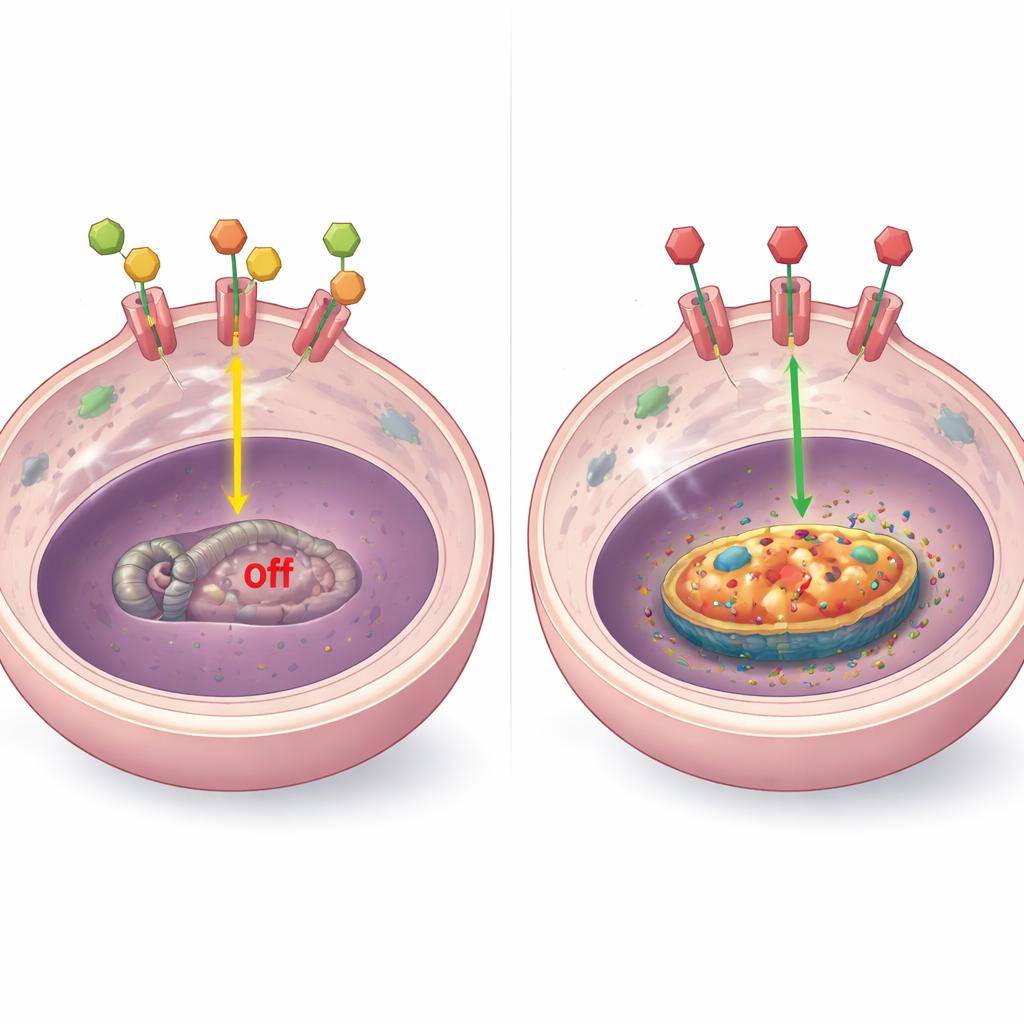
How hormone signals control BCL-2
To understand the mechanism, the researchers examined how the hormone receptor interacts with the BCL-2 gene. In laboratory models, they found that when hormones are present, the receptor physically binds to several sites near the BCL-2 gene and keeps it turned down. When hormones are removed or the receptor is blocked by drugs, this binding is lost, the local DNA becomes more “open,” and BCL-2 levels climb. This pattern showed up in diverse resistant tumor types, including those with high receptor levels, low or absent receptor, and receptor shifted out of the nucleus. In some special cases, other factors such as altered receptor fragments or the stress-response receptor (GR) also influenced BCL-2, but the central theme remained: shutting down normal hormone signaling removes a brake on this survival protein.
Testing BCL-2 as a weak point
Armed with this insight, the team asked whether BCL-2 is simply a marker of resistance or a true weakness they could exploit. They used organoids (mini-tumors grown in gel) and mouse tumors representing three major resistant subtypes: those still rich in nuclear hormone receptor, those with low receptor, and those with receptor largely trapped in the cell body rather than the nucleus. In all three, the clinically used BCL-2 blocker venetoclax (ABT-199) shrank tumors or slowed their growth, often more effectively than hormone-blocking drugs alone. In models that still had some hormone signaling, combining venetoclax with the anti-androgen enzalutamide produced stronger effects than either drug by itself, while tumors that were hormone-independent still responded to BCL-2 blockade alone.
Early hints from patients
To see if these ideas might translate to people, the investigators ran a small Phase Ib trial combining enzalutamide and venetoclax in men with advanced resistant prostate cancer. Using a device to sift rare tumor cells from blood, they tracked BCL-2, hormone-related genes, and a marker of tumor-cell burden over time. A few patients who stayed on treatment for many cycles showed falling BCL-2 and hormone signals in their circulating tumor cells, a drop in these cells’ numbers, and declines in blood PSA levels. In contrast, patients who did not benefit tended to have very low BCL-2 to begin with and signs that an alternative stress hormone pathway was taking over. Although this early trial was small and drug levels were limited by drug–drug interactions, it supports the idea that BCL-2–driven tumors are especially vulnerable to this approach.
What this means going forward
This work paints a coherent picture: when powerful hormone-blocking treatments pressure prostate tumors, many surviving cells flip on BCL-2 as a shared escape route. By combining detailed single-cell imaging, genetic analysis, animal experiments, and a pilot clinical study, the authors make a strong case that BCL-2 is more than a bystander—it is a common survival handle across several resistant tumor flavors. For patients, the long-term promise is a more tailored strategy in which doctors measure both hormone-receptor and BCL-2 status, then match men whose tumors lean on this switch to treatments that directly turn it off.
Citation: Jamroze, A., Liu, X., Hou, S. et al. Single-cell imaging analysis, therapeutic modeling and a Phase Ib trial validate BCL-2 as a target across heterogeneous castration-resistant prostate cancer. Sig Transduct Target Ther 11, 161 (2026). https://doi.org/10.1038/s41392-026-02700-w
Keywords: prostate cancer, drug resistance, BCL-2, hormone therapy, targeted therapy