Clear Sky Science · en
Coupling inertial, viscoelastic, and enhanced secondary flow in a composite microchannel: achieving high-precision multi-sized particle 3D central co-focusing
Bringing Tiny Travelers into Line
Imagine trying to photograph thousands of fast‑moving cars on a highway, but some are motorcycles and some are buses. To measure each one accurately, you would want them to pass through your camera in a neat single file, not scattered across lanes. In biomedical labs, scientists face a similar problem with cells and microscopic beads rushing through hair‑thin channels. This paper describes a new microfluidic chip that gently forces particles and white blood cells of different sizes into the same narrow three‑dimensional path, improving how we count, sort, and analyze them for diagnostics and research. 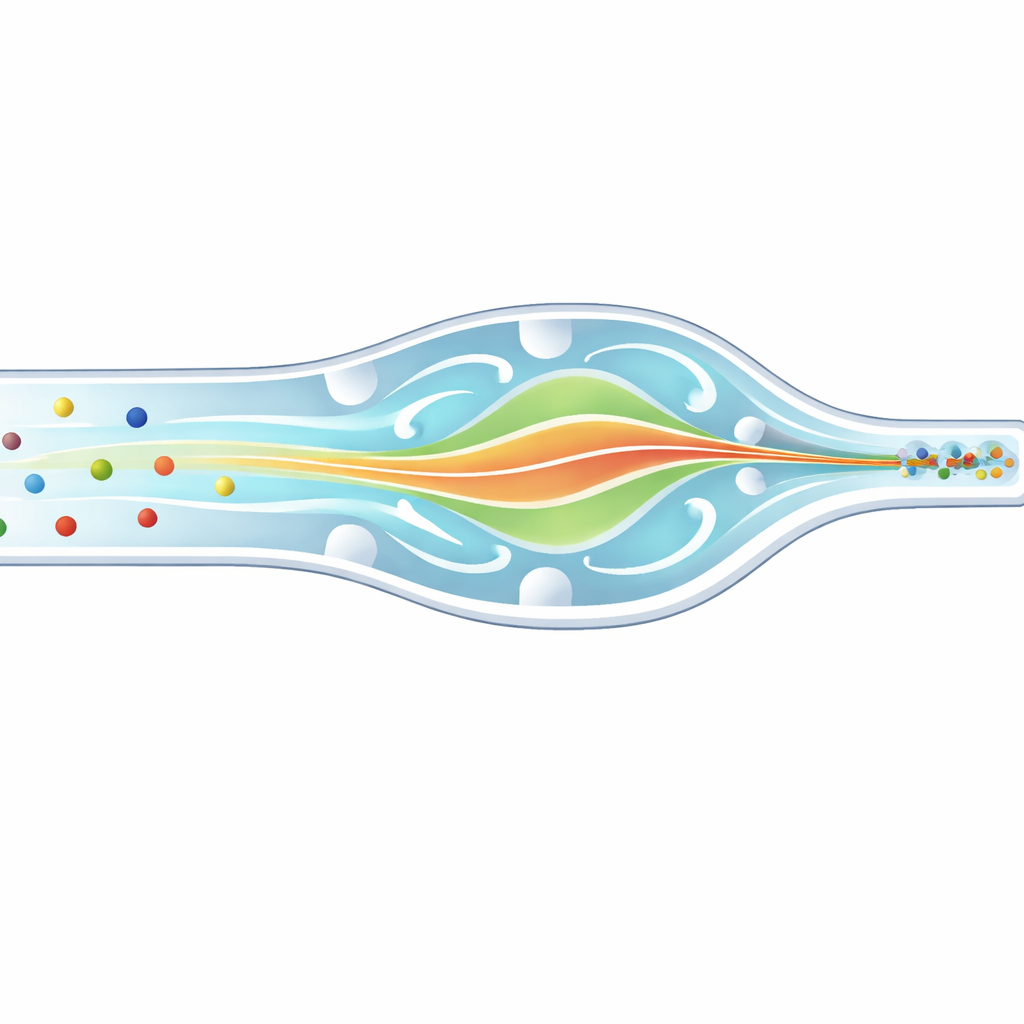
Why lining up cells really matters
Many cutting‑edge medical tools, such as flow cytometers and single‑cell analyzers, work by sending cells through tiny channels and interrogating them with light or electrical signals. If some cells pass near the wall and others glide through the center, they experience different illumination and forces, which blurs the measurements and hides subtle differences between cells. Conventional microfluidic designs can focus particles, but usually only of one size at a time, or they push them toward corners rather than the true center. That means mixtures of cells—like the diverse white blood cells in our blood—are hard to measure cleanly in a single pass.
Combining three gentle pushes on a micro scale
The researchers tackled this by building a carefully sculpted “composite” microchannel that exploits three fluid behaviors at once: inertia (the tendency of moving fluid and particles to keep going), elasticity (extra springiness added by dissolving a biopolymer called hyaluronic acid), and swirling secondary currents created by bends and obstacles. In their design, the channel curves like a slow spiral and carries a series of semicircular bumps both along the side wall and on the ceiling and floor. These features create controlled vortices that sweep particles away from corners, while the elastic forces in the fluid nudge them toward regions of gentle flow. By tuning the channel geometry and the fluid’s properties, the authors create a synergistic regime they call INVEST, in which the three effects cooperate rather than compete. 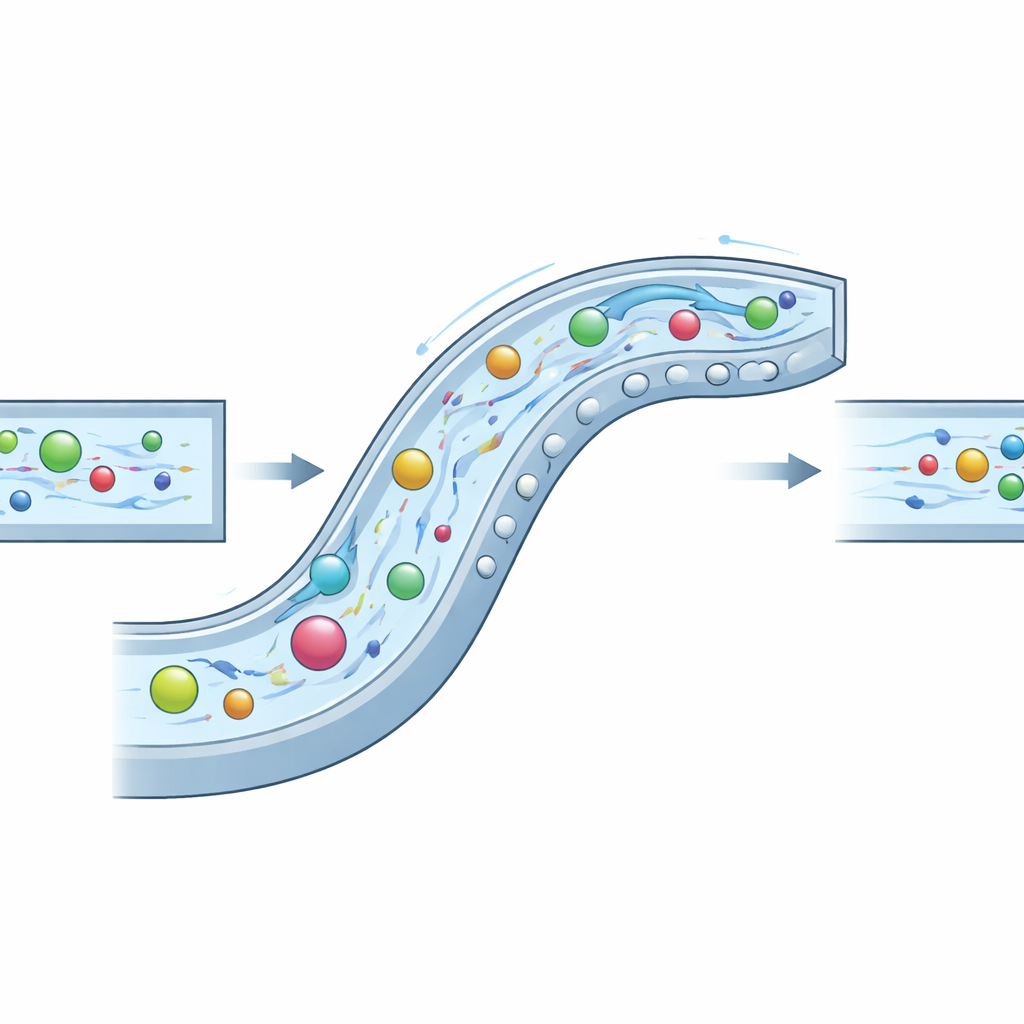
Testing the new microchannel highway
To understand how this system behaves, the team first ran detailed computer simulations of the flow patterns, the internal swirling, and how sharply the fluid shears near the walls. They introduced a new yardstick—the “equilibrium zone width”—that estimates how tightly particles can be confined at the center of the channel. The best design, with side and vertical obstacles aligned in phase, produced a very narrow equilibrium zone of about 16 micrometers, suggesting that particles would gather into a thin core stream. Simulations also showed that the added elasticity barely changed the strength of the swirls but did increase the restoring forces toward the center, sharpening the focus without destabilizing the flow.
Tuning the fluid and handling different particle sizes
Next, the researchers built the chip and pumped in fluorescent plastic beads suspended in saline solutions containing different amounts of hyaluronic acid. By filming the particles from above and from the side at thousands of frames per second, they measured both how wide the focused band was and what fraction of particles actually joined it. With the right polymer concentration, the channel produced a single bright streak only slightly wider than the particles themselves over a wide span of flow rates. Beads from 10 to 20 micrometers in diameter—which would normally settle at different positions—were all brought into the same central path with focusing efficiencies above 95% under optimal conditions. A more complex, staggered obstacle layout worked, but not as well or as robustly.
From plastic beads to living white blood cells
Finally, the team challenged the device with white blood cells, which vary widely in size and can deform under strong forces. Even so, the channel aligned most of these cells—ranging roughly from 7 to 20 micrometers—into a single narrow stream at moderate flow rates, achieving a peak focusing efficiency of just over 96%. At very high speeds, cells stretched and shrank, making them more vulnerable to the swirling currents and causing them to drift off the central line, but within practical operating windows the focusing remained strong. This shows that the INVEST strategy is compatible with delicate biological samples, not just rigid test beads.
What this means for future lab tools
In essence, this work shows that by artfully shaping a microchannel and choosing a slightly elastic fluid, it is possible to herd a diverse crowd of microscopic travelers—rigid particles or soft cells—into the same three‑dimensional lane. That makes optical and electrical measurements more consistent, boosts the throughput of micro‑scale analyzers, and reduces the need for complex alignment hardware. The authors’ composite channel turns an engineering headache—different‑sized particles taking different paths—into an advantage by letting inertia, elasticity, and swirling flow work together. This approach could help shrink powerful diagnostic instruments onto chips, bringing more precise blood analysis and single‑cell studies closer to routine clinical practice.
Citation: Zhao, T., Zeng, P., Ji, C. et al. Coupling inertial, viscoelastic, and enhanced secondary flow in a composite microchannel: achieving high-precision multi-sized particle 3D central co-focusing. Microsyst Nanoeng 12, 134 (2026). https://doi.org/10.1038/s41378-026-01254-9
Keywords: microfluidics, cell focusing, inertial flow, viscoelastic fluids, flow cytometry