Clear Sky Science · en
Research progress in heterogeneity of dental mesenchymal stem cells
Why tiny tooth stem cells matter
Teeth look simple on the surface, but inside them lies a bustling world of stem cells that quietly build, maintain, and repair our smiles. This review article explores a special family of cells in and around teeth—dental mesenchymal stem cells—and explains how their hidden diversity may open the door to better treatments for tooth decay, gum disease, bone loss in the jaw, and even nerve injuries. By understanding the “cast of characters” behind tooth development and healing, researchers hope to design smarter, more reliable regenerative therapies instead of relying only on drilling, fillings, and implants.
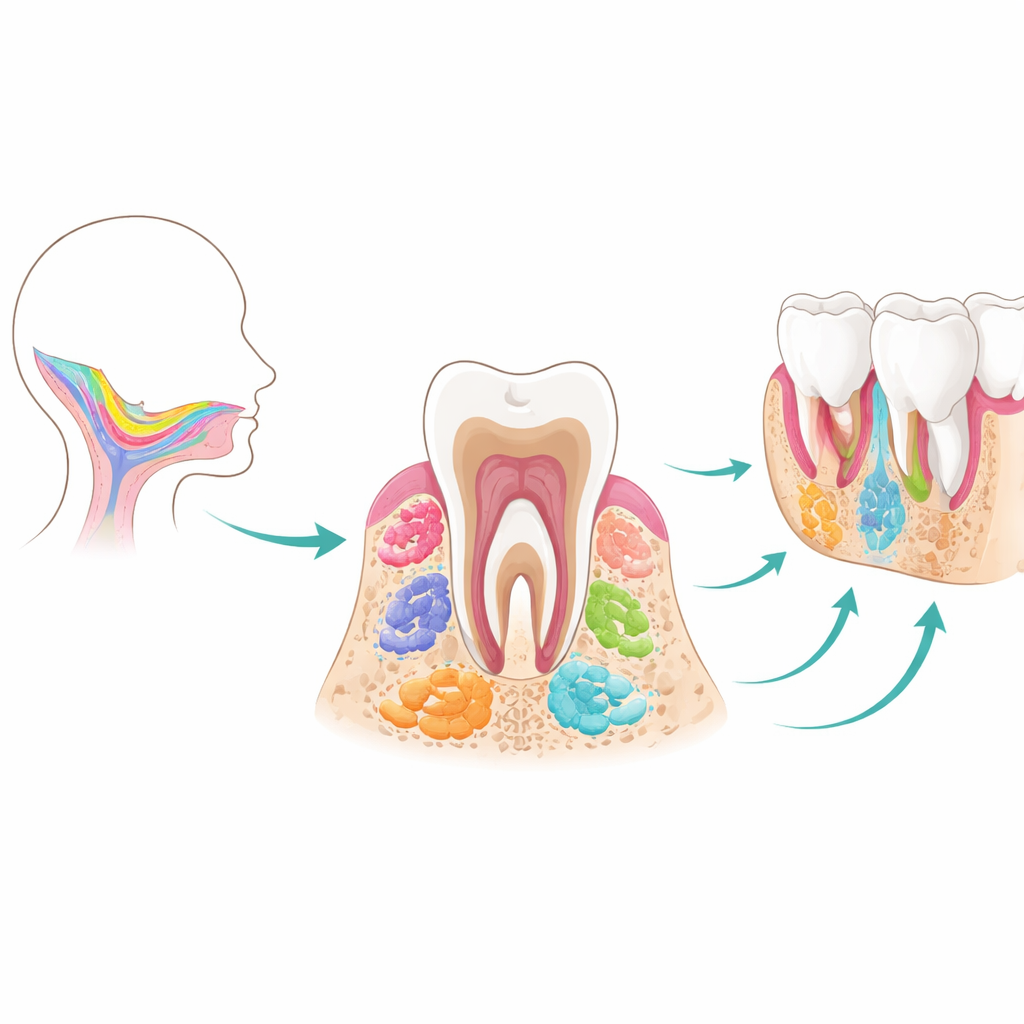
From early tooth buds to full-grown teeth
Teeth and their supporting tissues form through a tightly choreographed partnership between two main layers: an outer covering that will later make enamel, and inner cells that give rise to dentin, pulp, ligament, and jaw bone. The mesenchymal stem cells at the heart of this review all trace back to migratory cells from the early embryo called the cranial neural crest. As they settle in the forming jaw, these cells first produce tooth germs—tiny “buds” that pass through bud, cap, and bell stages. Along this timeline, several temporary stem cell populations appear in sequence. Tooth germ progenitor cells act as early, versatile builders; dental follicle stem cells help assemble the tissues that hold teeth in place; and stem cells in the apical papilla at the root tip guide root formation and show surprising potential to become nerve-like cells. Once tooth roots mature, many of these early players disappear, making them hard to study or reuse later in life.
Many stem cell types, one integrated tooth system
Even after teeth are fully formed, multiple kinds of stem cells remain tucked into protected niches and continue to support tissue upkeep. Dental pulp stem cells live inside the tooth, where blood vessels and nerves enter; periodontal ligament stem cells sit between tooth root and bone; gingival stem cells line the gums; and mesenchymal stem cells in the alveolar (jaw) bone form the “soil” that anchors teeth. Each population shares basic traits like the ability to self-renew and to turn into bone- or dentin-making cells, but they are not interchangeable. For example, pulp stem cells tend to favor dentin and nerve-like fates, periodontal cells excel at rebuilding the fibrous attachment and cementum on the root surface, and jaw bone stem cells are specialized for fast, direct bone formation in the face. This built-in division of labor helps explain why the mouth can respond differently to injury in pulp, ligament, or bone.
Hidden differences within each stem cell pool
A key message of the review is that heterogeneity exists not only between different stem cell types, but also within each one. New techniques such as single-cell RNA sequencing can profile thousands of individual cells at once, revealing subgroups with distinct gene activity and behavior. In dental pulp, for example, researchers have mapped multiple clusters: some maintain the reserve pool and drive proliferation, others are primed for dentin production, and still others are biased toward forming blood vessels or nerve-like cells. Similar subgroups have been found in periodontal ligament stem cells, jaw bone stem cells, stem cells from baby teeth, and gingival stem cells. Marker molecules on the cell surface and inside the nucleus help distinguish these subpopulations, which differ in their growth capacity, aging pattern, and responsiveness to mechanical forces, inflammation, or bacterial attack.
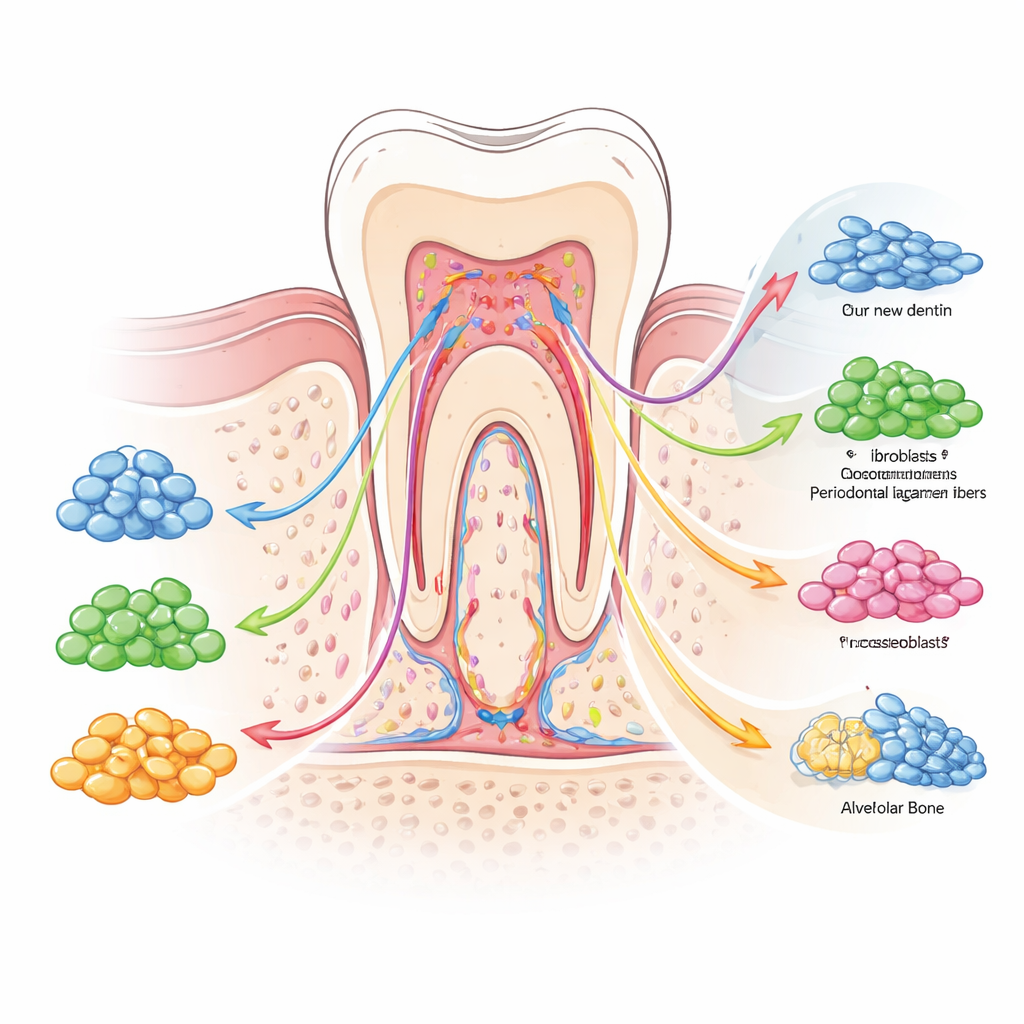
Signals and switches that shape stem cell behavior
The authors highlight that this diversity is not random. It is organized by a network of biochemical signals and “epigenetic” switches that act like traffic lights and road signs for cell fate. Pathways driven by bone morphogenetic proteins, Wnt signals, and transforming growth factor beta push cells toward building hard tissues such as dentin and bone, while others, including Notch and PI3K/AKT, help maintain a balanced pool of stem and progenitor cells. At the same time, chemical tags on DNA and packaging proteins adjust which genes are accessible, pre-setting certain cells for rapid response when damage occurs. Mechanical forces from chewing, inflammatory molecules from infection, and cues from nearby blood vessels or nerves further tilt the balance toward repair, scarring, or breakdown. Together, these layers of control create a flexible, but sometimes unpredictable, healing system.
What this means for future dental care
To a non-specialist, the main takeaway is that tooth and gum regeneration is no longer science fiction, but its success will depend on managing stem cell diversity rather than ignoring it. The review argues that knowing which specific subpopulations build dentin, ligament, bone, or nerves—and which signals steer them—will help researchers refine cell selection, biomaterials, and drug cues for predictable outcomes. At the same time, the authors caution that most detailed maps come from animal studies and lab cultures, and that technical noise can blur the picture. They call for integrated “multi-omics” approaches in human tissue, better standardization, and rigorous testing in living systems. Ultimately, decoding the heterogeneity of dental mesenchymal stem cells could allow dentists to move from replacing damaged structures with inert materials to truly regenerating living, functional teeth and their supporting tissues.
Citation: Fu, H., Chen, P., Wu, Z. et al. Research progress in heterogeneity of dental mesenchymal stem cells. Int J Oral Sci 18, 31 (2026). https://doi.org/10.1038/s41368-026-00433-8
Keywords: dental stem cells, tooth regeneration, periodontal repair, single-cell sequencing, oral tissue engineering