Clear Sky Science · en
Single-cell multi-omics sequencing reveals cell-specific transcriptomic and chromatin accessibility profiles in gut microbiome metabolite butyrate-produced pain modulation
Why your gut might matter for jaw pain
People with temporomandibular joint (TMJ) disorders often live with stubborn jaw pain that makes talking, eating, and sleeping difficult. Many current treatments offer only partial or temporary relief and can carry side effects, especially when they rely on painkillers like opioids. This study explores an unexpected ally in the search for safer therapies: tiny molecules made by gut bacteria. By looking deep inside individual brain cells, the researchers show how a gut-derived substance called butyrate can dial down TMJ pain and point toward future, non-opioid treatments.
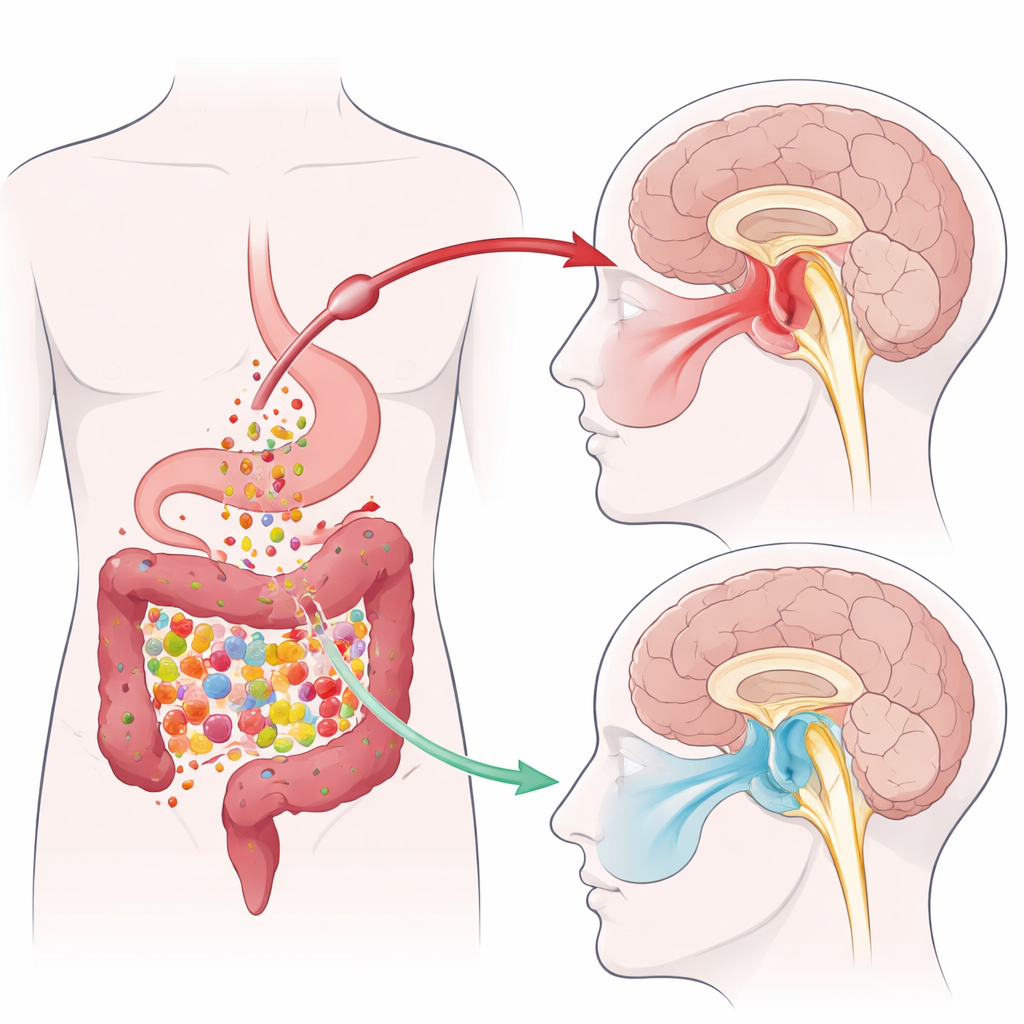
From gut bacteria to jaw pain relief
The team used a mouse model of inflammatory TMJ pain created by injecting an irritant into the jaw joint. This procedure made the animals more sensitive to touch on the side of the face connected to the injured joint. Earlier work from the same group had shown that TMJ inflammation lowers levels of short-chain fatty acids produced by gut microbes, especially butyrate. In this study, the scientists gave mice tributyrin, a stable compound that breaks down into butyrate in the intestine, by mouth for 10 days. This treatment raised the animals’ pain thresholds on the affected side of the face, meaning they reacted less strongly to mechanical stimulation, while leaving the opposite side unchanged. Measurements of feces, blood, and a brainstem region called the spinal trigeminal nucleus caudalis (Sp5C) showed that TMJ inflammation reduced butyrate throughout the body—and that tributyrin restored it to normal.
Mapping the pain gateway in the brainstem
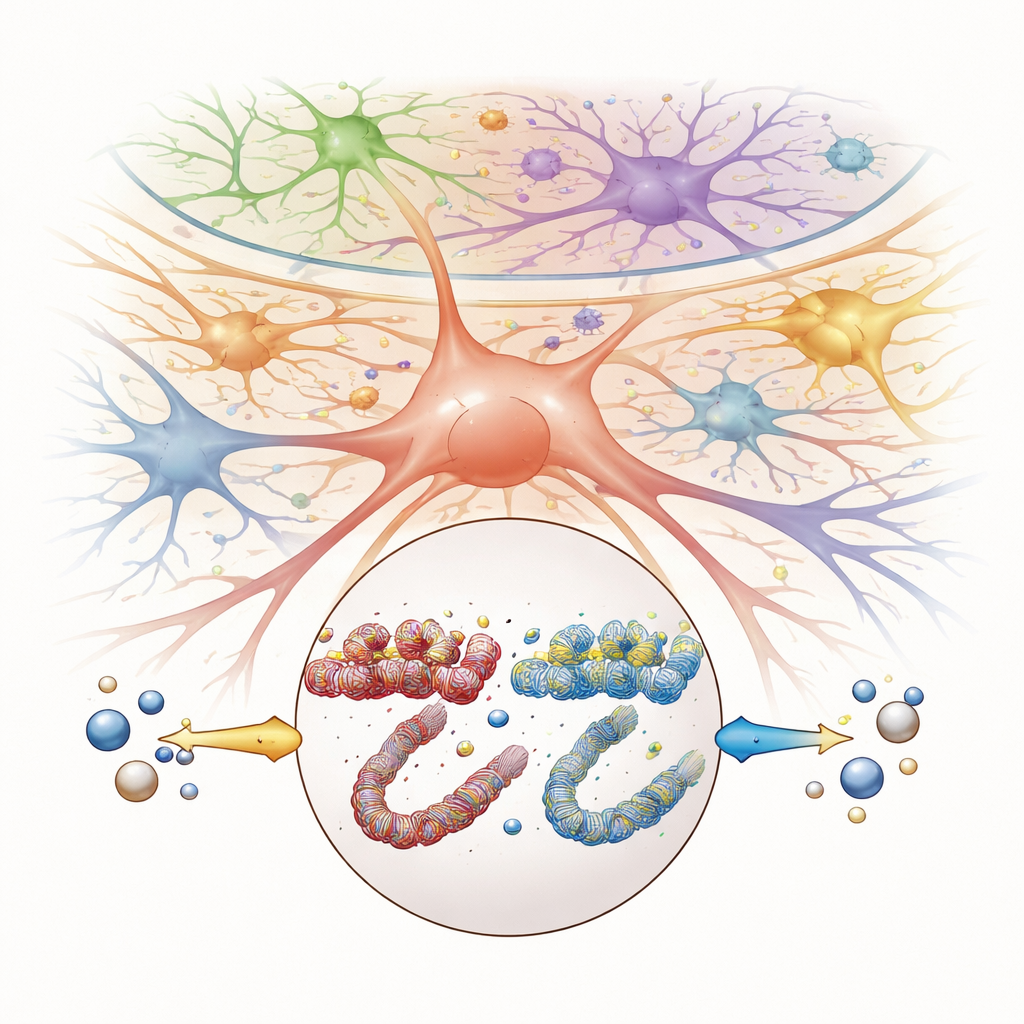
The Sp5C acts as a key relay station for facial pain, including signals from the TMJ. To understand what happens there during pain and recovery, the researchers used a powerful “single-cell multi-omics” approach. They isolated nuclei from Sp5C tissue and, for each individual cell, measured both which genes were turned on and how open the surrounding DNA was—a sign of how easy it is for genes to be activated. This allowed them to identify 12 distinct cell types, including several kinds of neurons, immune cells, and support cells called glia, and to see how each one responded to TMJ inflammation and tributyrin. Interestingly, the overall mix of cell types did not change much; instead, pain and treatment mainly altered how existing cells used their genes.
Key switches flipped by pain and reset by butyrate
By comparing healthy mice, mice with TMJ pain, and mice treated with tributyrin, the team pinpointed specific genes whose activity was consistently disturbed by pain and then reset by butyrate. Across several neuron groups and precursor cells, they highlighted five genes—Nop14, Matk, Idh3b, Ndst2, and Tomm6—as central players. TMJ inflammation changed both the activity of these genes and the openness of nearby DNA regions, while tributyrin reversed these shifts. The researchers then zoomed in on Nop14 in a class of cells called neuropeptide neurons, which showed particularly strong changes. They used additional analyses to map how local DNA control elements and regulatory proteins interact to boost or suppress this gene under pain conditions and after treatment.
How gene packaging connects to pain signals
Because butyrate is known to influence how tightly DNA is wrapped around histone proteins, the researchers also examined histone acetylation, a chemical mark linked to more permissive gene activity. TMJ inflammation lowered overall histone acetylation in the Sp5C, while tributyrin brought it back toward normal. At the same time, a specific region near the Nop14 gene became more open and more heavily acetylated during pain, consistent with increased gene activity. When the team used a virus-based method to reduce Nop14 levels directly in the Sp5C, they observed two effects: histone acetylation in this region moved back toward baseline, and the animals’ TMJ pain responses decreased. This suggests that Nop14 sits at a crucial junction where changes in gene packaging, triggered by the loss or restoration of butyrate, can amplify or calm pain pathways.
A new path toward non-opioid pain therapies
Overall, the study links gut-derived butyrate to TMJ pain relief through precise, cell-specific changes in gene regulation within a brainstem pain hub. Rather than simply numbing pain signals, tributyrin appears to correct molecular imbalances that TMJ inflammation creates in particular neurons and support cells, with Nop14 emerging as a promising target. For patients, this work does not yet translate into an immediate therapy, but it supports the idea that manipulating gut metabolites—or directly targeting the same gene switches they control—could offer future, non-opioid treatments for TMJ disorders and possibly other forms of chronic inflammatory pain.
Citation: Tao, R., Liu, S., Crawford, J. et al. Single-cell multi-omics sequencing reveals cell-specific transcriptomic and chromatin accessibility profiles in gut microbiome metabolite butyrate-produced pain modulation. Int J Oral Sci 18, 37 (2026). https://doi.org/10.1038/s41368-026-00432-9
Keywords: temporomandibular joint pain, gut microbiome, butyrate, epigenetic regulation, single-cell sequencing