Clear Sky Science · en
SLIT3: a novel regulator of odontogenic differentiation through Akt/GSK3β/β-catenin signaling pathway
Why this tooth story matters
Many children and teenagers suffer injuries or deep cavities that kill the pulp inside a still-developing tooth. When that happens, the root may stop growing, leaving a short, fragile tooth that can break easily. This study explores a natural protein called SLIT3 and how it helps special stem cells at the tip of young teeth build stronger root dentin, pointing toward future treatments that could help damaged young teeth keep growing instead of being lost.
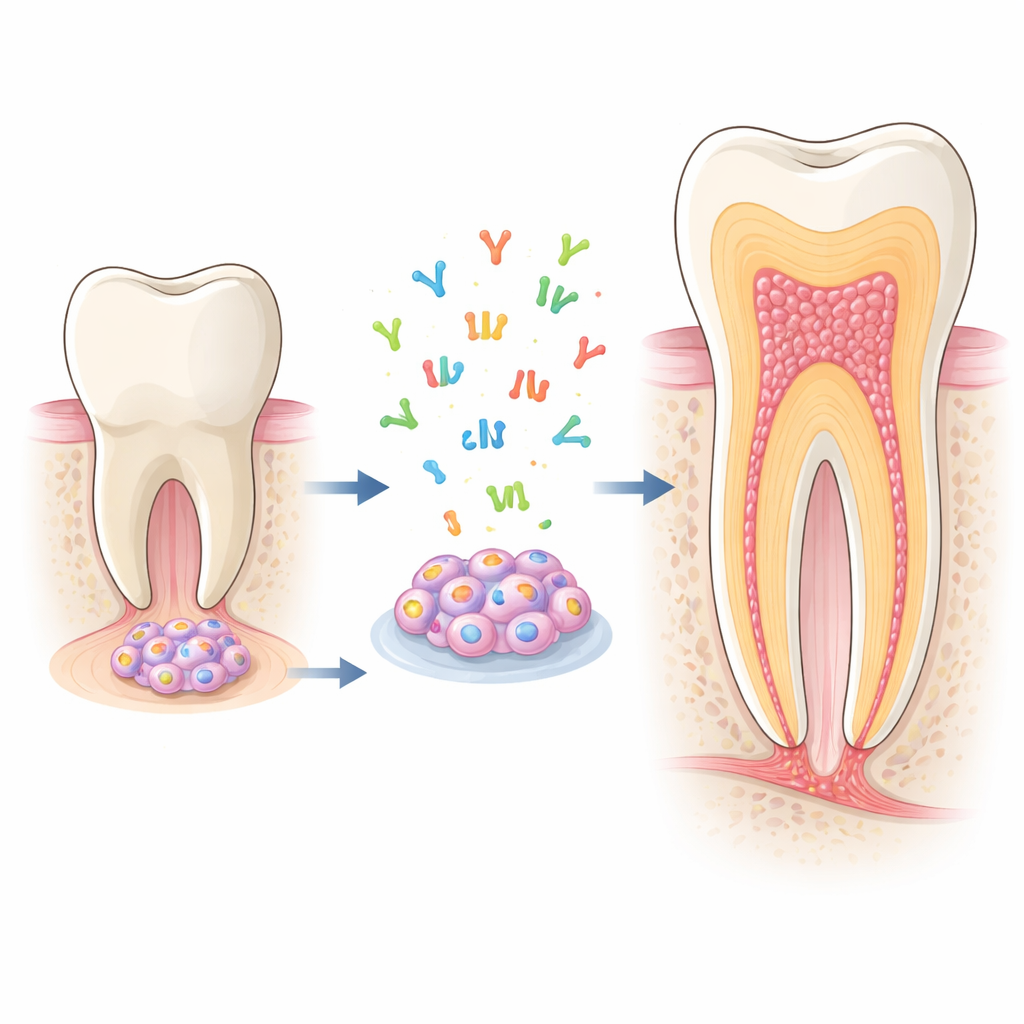
Hidden builders inside young teeth
At the growing tip of an immature tooth root sits a soft tissue called the apical papilla. It houses stem cells from apical papilla (SCAP), which can turn into several types of tooth-supporting cells, especially odontoblasts—the cells that lay down dentin, the hard tissue forming most of the tooth. Because SCAP grow well in the lab and can readily become odontoblast-like cells, they are an ideal model to study how roots develop and how we might coax them to repair themselves after disease or injury.
A guidance protein with a new job
SLIT3 is best known as a guidance cue that helps nerve fibers find their paths during brain development, and it also links bone breakdown to new bone formation. Since bone and dentin share many building rules, the researchers asked whether SLIT3 might also guide the formation of dentin in teeth. By reanalyzing single-cell RNA sequencing data from mouse tooth germs at different stages, they found that the Slit3 gene is active mainly in the tooth’s inner supporting tissue (dental mesenchyme), not in the outer epithelium. Yet, the SLIT3 protein itself showed up strongly on both dentin-forming odontoblasts and enamel-forming ameloblasts, hinting that it is secreted and then acts on nearby cells to coordinate hard tissue formation.
Helping stem cells grow and harden
To test SLIT3’s role directly, the team isolated human SCAP from immature wisdom teeth and grew them under conditions that encourage dentin formation. As these cells matured, both SLIT3 gene and protein levels rose steadily. When the researchers artificially lowered SLIT3 with small interfering RNA, SCAP multiplied more slowly and formed weaker mineral deposits, with reduced levels of two key dentin proteins, DMP-1 and DSPP. In contrast, adding extra human SLIT3 protein made SCAP grow faster, boosted mineralization, and increased DMP-1 and DSPP. When SCAP with more or less SLIT3 were transplanted into tiny ceramic scaffolds in mice, the high-SLIT3 constructs produced richer dentin-like tissue, while low-SLIT3 constructs produced poorer tissue, confirming SLIT3’s importance in living systems.
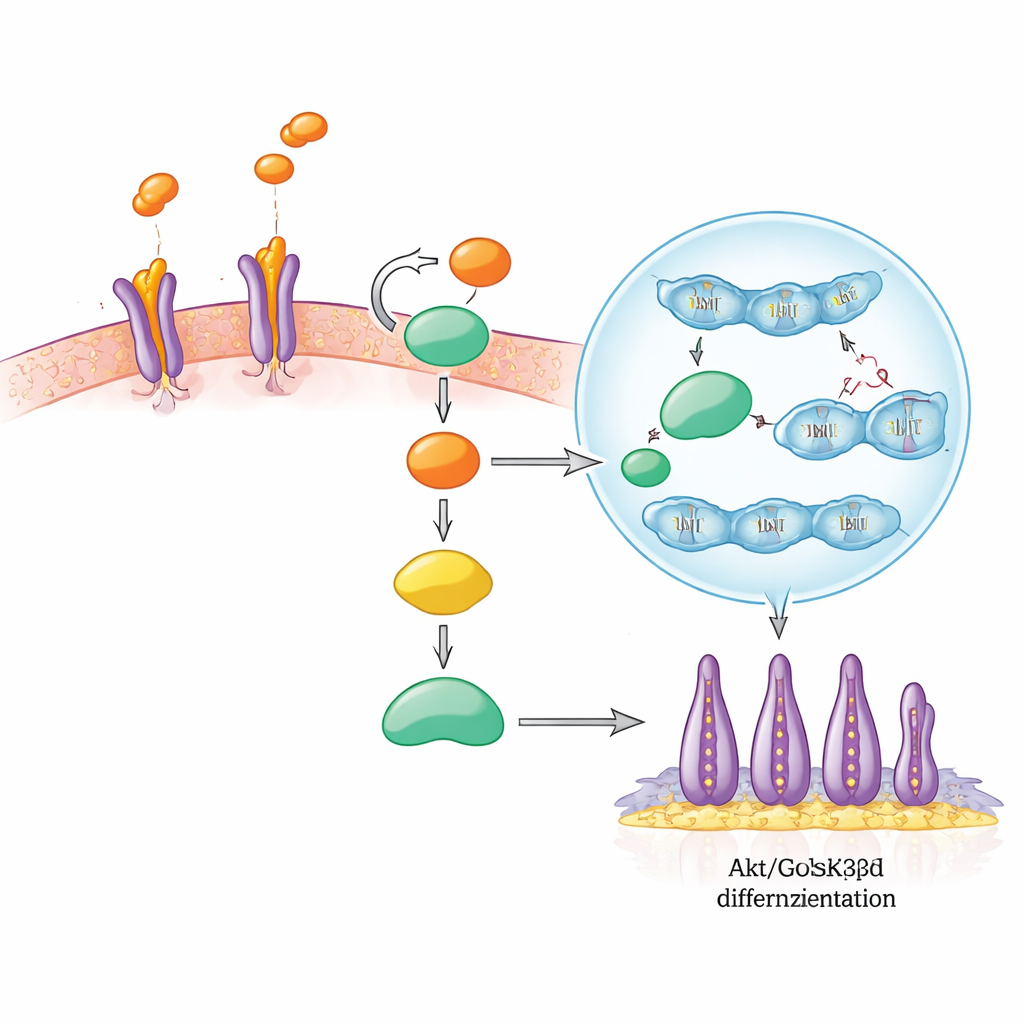
Inside the cell’s decision circuit
The researchers then followed the chain of signals SLIT3 triggers inside SCAP. SLIT3 first binds to ROBO2 and ROBO3 receptors on the cell surface, much like a key turning a lock. This activates a well-known growth and survival hub called Akt, which then switches off another protein, GSK3β. Normally, active GSK3β helps mark the messenger β-catenin for destruction. When GSK3β is turned off, β-catenin accumulates and travels into the cell nucleus, where it partners with other factors to turn on genes that drive cell division and specialization. In this study, SLIT3 stimulation rapidly increased Akt and GSK3β phosphorylation, boosted nuclear β-catenin, and raised levels of β-catenin target proteins linked to growth. Blocking ROBO2/ROBO3 prevented these changes, showing those receptors are essential for the signal to pass.
Proving the pathway really matters
To show that this internal circuit is not just a side effect but actually required for dentin formation, the team used a chemical called Resibufogenin to block the Akt/GSK3β/β-catenin route. When this pathway was inhibited, SLIT3 could no longer increase β-catenin in the nucleus or raise DMP-1 and DSPP levels, and its ability to enhance odontogenic differentiation was essentially lost. Together with the receptor experiments, this firmly supports a model in which SLIT3 acts through ROBO2/3 to flip on the Akt/GSK3β/β-catenin switch, which in turn tells SCAP to multiply and mature into dentin-producing cells.
What this means for future tooth repair
The work positions SLIT3 as a new regulator of dentin formation at the root tip, extending our map of how tooth roots grow and harden. For patients with damaged, immature teeth, understanding this SLIT3-centered pathway could eventually inspire biologically based treatments that encourage the tooth’s own stem cells to continue building root dentin. While any clinical application will require much more research, this study adds a key missing piece to the puzzle of how young teeth develop—and how we might help them heal themselves.
Citation: Jiang, L., Liu, L., Yang, F. et al. SLIT3: a novel regulator of odontogenic differentiation through Akt/GSK3β/β-catenin signaling pathway. Int J Oral Sci 18, 35 (2026). https://doi.org/10.1038/s41368-026-00426-7
Keywords: tooth root regeneration, dentin formation, dental stem cells, SLIT3 signaling, Wnt beta-catenin pathway