Clear Sky Science · en
VAP1 promotes cardiac fibrosis by enabling PDGFR signaling in myofibroblasts
Why scarring of the heart matters
Heart failure affects millions of people and often develops as the heart slowly fills with stiff scar tissue. This scarring process, called fibrosis, makes it harder for the heart to relax and pump blood. The study behind this article explores how certain support cells in the heart turn into aggressive scar-forming cells and identifies a surface molecule, VAP1, that helps drive this change. Understanding this switch opens the door to new ways to slow or even reverse harmful scarring in heart disease. 
From quiet helpers to overactive builders
The healthy heart contains many fibroblasts, quiet support cells that maintain the tissue’s structure. When the heart is injured or under long-term pressure, some of these fibroblasts switch into myofibroblasts, which are highly active builders of scar tissue. In small bursts this response can be protective, but when it continues unchecked it fills the heart wall with tough fibers that limit its movement. The researchers focused on how this fibroblast-to-myofibroblast shift is controlled, especially in a common model of pressure overload that mimics forms of human heart failure.
A surface gatekeeper called VAP1
Using large-scale gene activity screens, the team found that a surface protein named VAP1 sharply increased when a master switch for scarring, the protein MKL1, was turned on. VAP1 itself is known for helping white blood cells stick to blood vessels, but its role inside the heart was unclear. In heart fibroblasts grown in the lab, turning down VAP1 reduced hallmark features of myofibroblasts, such as cell growth, movement, and the ability to contract and pull on their surroundings. Boosting VAP1 had the opposite effect, pushing fibroblasts more strongly toward the active, scar-forming state. 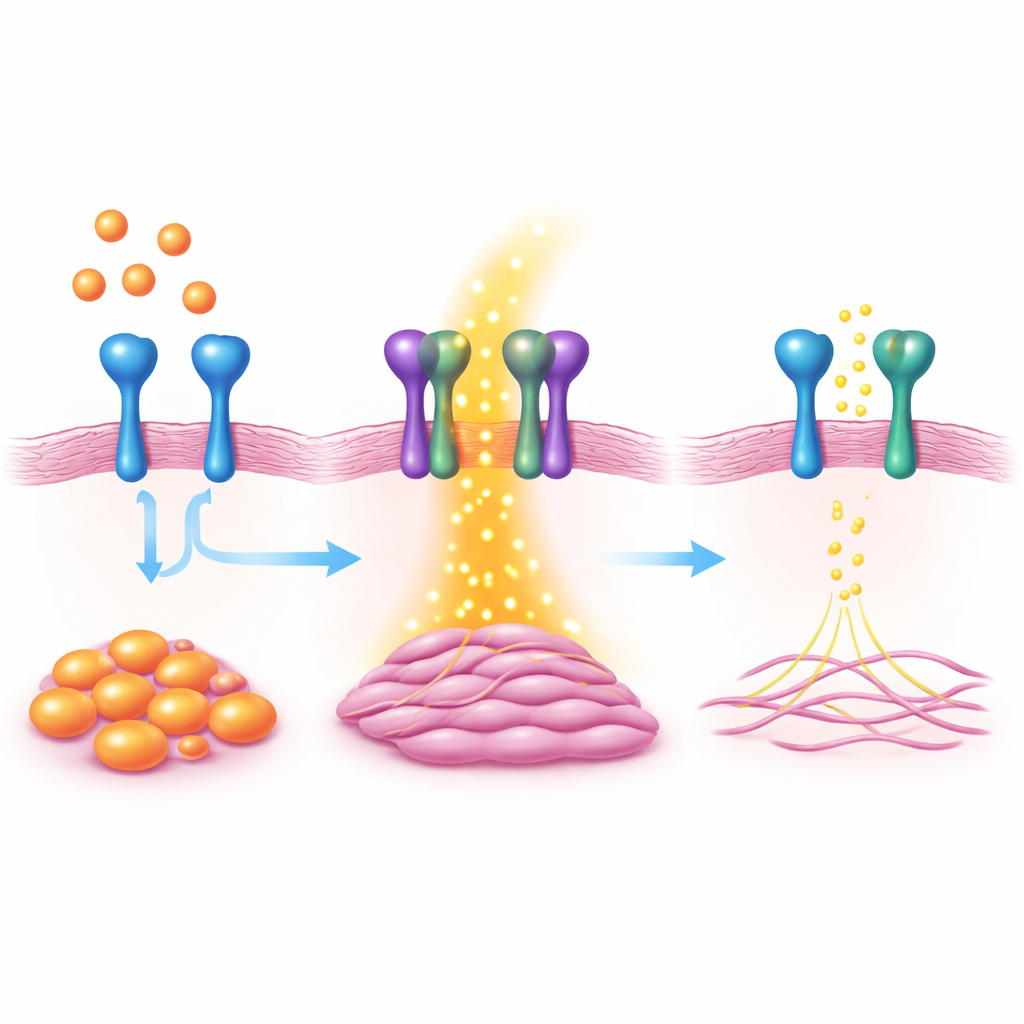
Testing VAP1 in living hearts
To see how VAP1 behaves in a full organism, the scientists engineered mice in which VAP1 could be removed only from heart fibroblasts. When these mice were exposed to long-term pressure on the main artery leaving the heart, they developed far less fibrous scarring than normal mice, even though overall heart enlargement was similar. Measures of heart pumping function were also better preserved. Removing VAP1 specifically from already activated myofibroblasts produced a similar reduction in scarring and improvement in function, showing that VAP1 is important both as fibroblasts switch on and as they remain active.
How VAP1 boosts pro-scar signals
To uncover how VAP1 works, the team combined gene activity analysis with protein surveys. They discovered that VAP1 physically interacts with another surface protein called PDGFRβ, a receptor that senses growth signals known to trigger fibrosis. When VAP1 was missing, PDGFRβ became less active and a series of downstream signal pathways that encourage cell growth, movement, and collagen production were weakened. A portion of the VAP1 protein that sits outside the cell membrane was enough to restore this missing signal when added back, suggesting that the physical partnership between VAP1 and PDGFRβ on the cell surface is key.
Blocking VAP1 with a small molecule
Because VAP1 also functions as an enzyme, the researchers tested a drug-like compound that blocks its activity. In heart fibroblasts in dishes, this inhibitor reduced myofibroblast markers and curbed their ability to divide, migrate, and contract. When given to mice with pressure overload, the compound did not stop the heart from growing larger, but it did markedly reduce scar buildup and improved measures of how well the heart pumped blood. These results suggest that pharmacological blocking of VAP1 can soften the scarring response without disrupting all aspects of the heart’s adaptation to stress.
What this means for people with heart failure
Together, the findings show that VAP1 acts as a crucial helper for signals that push heart support cells into a rigid, scar-forming mode. By tuning down VAP1, either genetically in fibroblasts or with an inhibitor, the heart develops less stiff collagen and retains better function under stress. While more work is needed in additional animal models and human tissue, the study positions VAP1 as a promising target for future therapies aimed at limiting harmful cardiac fibrosis and easing the burden of heart failure.
Citation: Huang, S., Zhao, Q., Shao, T. et al. VAP1 promotes cardiac fibrosis by enabling PDGFR signaling in myofibroblasts. Exp Mol Med 58, 1284–1296 (2026). https://doi.org/10.1038/s12276-026-01690-7
Keywords: cardiac fibrosis, heart failure, fibroblasts, VAP1, PDGFR signaling