Clear Sky Science · en
Haloperidol induces neuroprotection and enhances neuromuscular function in both murine and human models of spinal muscular atrophy
Turning a Psychiatric Drug into a Nerve Protector
Spinal muscular atrophy (SMA) is a genetic disease that weakens muscles and can be fatal in childhood. Existing treatments help many patients but are difficult to deliver, very costly, and do not fully restore muscle and nerve health. This study asks a simple but powerful question: can haloperidol, a long‑used antipsychotic drug, be repurposed at much lower doses to protect the nerve cells and muscles affected in SMA, offering a more accessible and complementary option?
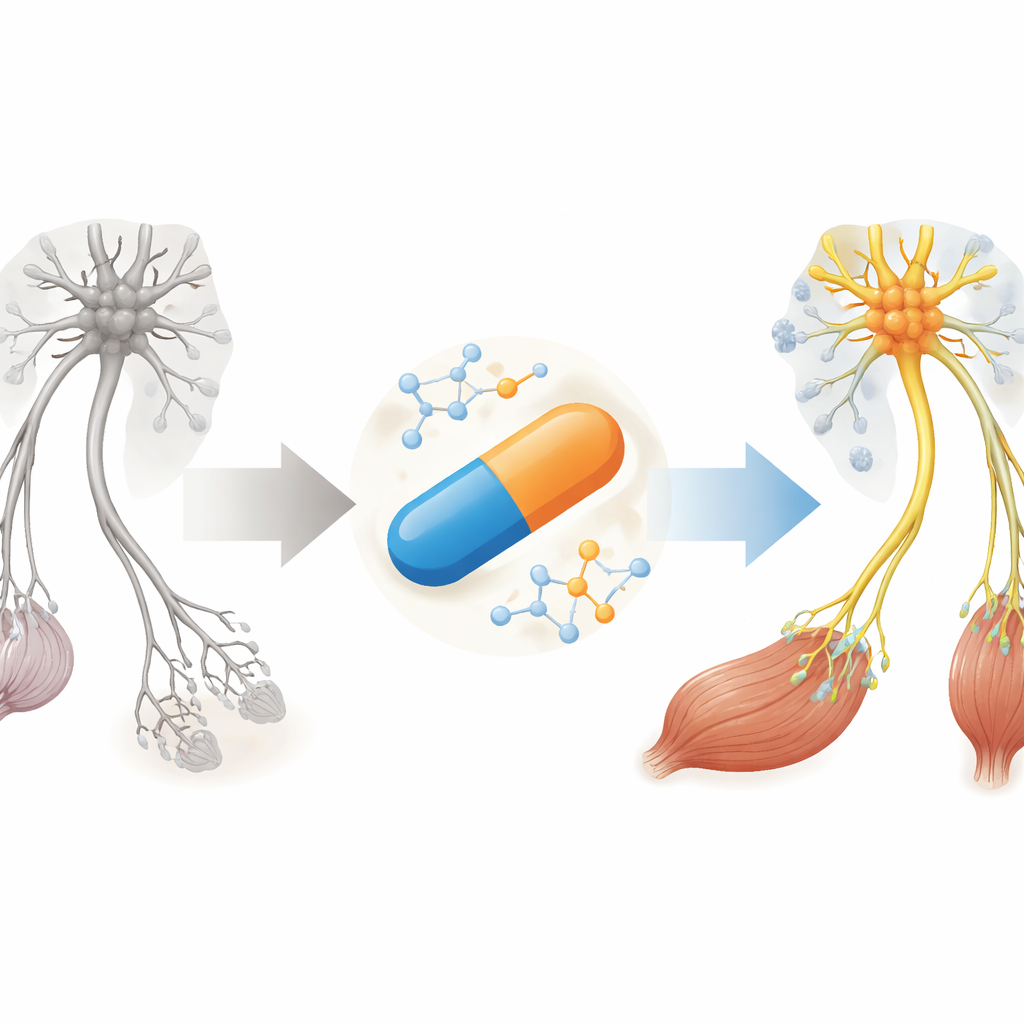
Why a Muscle‑Wasting Disease Needs New Options
SMA is caused by a shortage of a protein called SMN, which motor neurons—the nerve cells that control movement—need to survive. Current drugs focus almost entirely on boosting SMN in the nervous system, often through spinal injections or expensive gene therapy. Yet many patients still show defects at the neuromuscular junction, the tiny contact points where nerves talk to muscles, even after months of therapy. The authors argue that future treatments must not only raise SMN levels more broadly in the body but also calm damaging inflammation and directly support muscle and junction health.
Testing Haloperidol in Mice and Human Cells
The team previously screened approved medicines in fruit flies and human cells and found that haloperidol could improve how the backup SMN2 gene is processed, increasing SMN protein. In this study, they gave a low dose of haloperidol to a severe SMA mouse strain from just after birth. The treated mice lived longer, gained more weight, and showed better balance, posture, and limb strength than untreated littermates. In parallel, the researchers grew motor neurons and muscle cells from SMA patients using stem‑cell methods. When these human cells were exposed to tiny amounts of haloperidol, more motor neurons survived, formed richer networks of branches, and made larger, better‑connected muscle fibers in co‑culture.
Protecting Nerves, Calming Support Cells, and Strengthening Muscles
Inside the mouse spinal cord, haloperidol raised SMN protein by about half and nearly doubled SMN in leg muscles. Motor neurons in treated animals were more numerous and showed fewer molecular signs of programmed cell death. Gene‑activity surveys revealed that hundreds of messages tied to RNA processing, cell survival, and synapse structure shifted toward a healthier pattern, including many known SMN‑dependent targets. Importantly, haloperidol also reduced markers of overactive astrocytes and reshaped microglia—the brain’s immune cells—toward a calmer, more resting form. In the muscles, fibers in a vulnerable thigh muscle grew larger, and more neuromuscular junctions were fully innervated and properly matured, indicating stronger and more reliable communication between nerves and muscles.
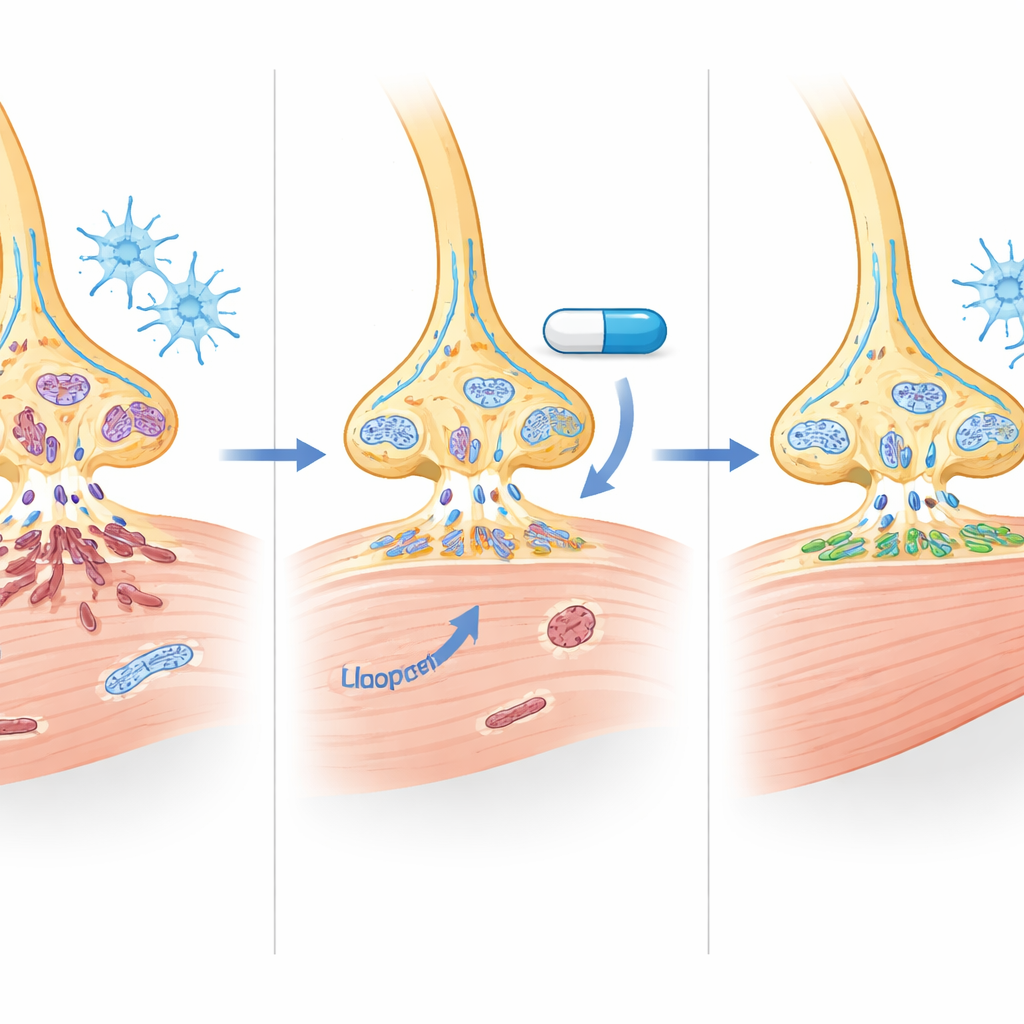
Clues to How the Drug Works Under the Hood
Haloperidol is best known for blocking dopamine D2 receptors at psychiatric doses, but the low doses used here did not measurably change those receptors in mouse brain, spinal cord, or patient‑derived neurons. Instead, the data point to a dual action: modest but meaningful increases in SMN protein together with broader "housekeeping" effects on RNA processing, cell‑death pathways, inflammation, and mitochondrial function. Some genes tied to energy production and fat handling in muscle also shifted, hinting that the drug may partially correct the metabolic disturbances seen in SMA muscle, although the exact impact on mitochondria will require future work.
What This Could Mean for People with SMA
For lay readers, the key message is that an old, inexpensive drug—haloperidol—may, at carefully chosen low doses, help preserve the nerve‑muscle connection in SMA rather than just altering mood or behavior. In both mice and human cell models, it extended survival of motor neurons, improved muscle size, and strengthened the microscopic junctions that power movement, all while dampening harmful inflammation. While much remains to be tested, especially regarding long‑term safety and how best to combine it with existing SMA therapies, this research suggests haloperidol could eventually become a practical add‑on or alternative treatment that makes current gains in strength and independence more durable.
Citation: Menduti, G., Perez-Gomez, R., Berenger-Currias, N. et al. Haloperidol induces neuroprotection and enhances neuromuscular function in both murine and human models of spinal muscular atrophy. Exp Mol Med 58, 1216–1229 (2026). https://doi.org/10.1038/s12276-026-01689-0
Keywords: spinal muscular atrophy, drug repurposing, haloperidol, motor neurons, neuromuscular junction