Clear Sky Science · en
Human pluripotent stem cell engineering with CRISPR–Cas9 for Parkinson’s disease
Why this research matters to everyday life
Parkinson’s disease robs people of smooth movement, speech, and independence, and today’s drugs only ease symptoms for a while. This article explores how scientists are combining two powerful tools, human stem cells and DNA-cutting proteins known as CRISPR, to better understand why key brain cells die in Parkinson’s and to design safer, longer lasting cell therapies that could one day restore lost function rather than just mask the disease.
Understanding a common brain disorder
Parkinson’s disease affects millions of people worldwide and becomes more common with age. The condition centers on the slow loss of dopamine-making nerve cells in a small midbrain region and on clumps of a sticky protein called alpha synuclein inside cells. Standard treatments like levodopa can temporarily replace missing dopamine, but they do not stop these cells from dying. Animal models have helped uncover clues, yet they often fail to capture the gradual, human-specific course of the illness, leaving big gaps between lab discoveries and useful treatments for patients.
Growing human nerve cells in the dish
Human pluripotent stem cells, which can turn into almost any cell type, now let researchers grow large numbers of human dopamine-producing neurons in the lab. These lab-grown cells mimic many hallmarks of Parkinson’s, including problems with the energy factories of the cell, waste disposal systems, and the build-up of alpha synuclein. Because they can be made from a person’s own tissues, these cells also carry that person’s exact genetic makeup, allowing scientists to model both inherited and more common sporadic forms of the disease in a human setting and to test candidate drugs directly on vulnerable cell types.
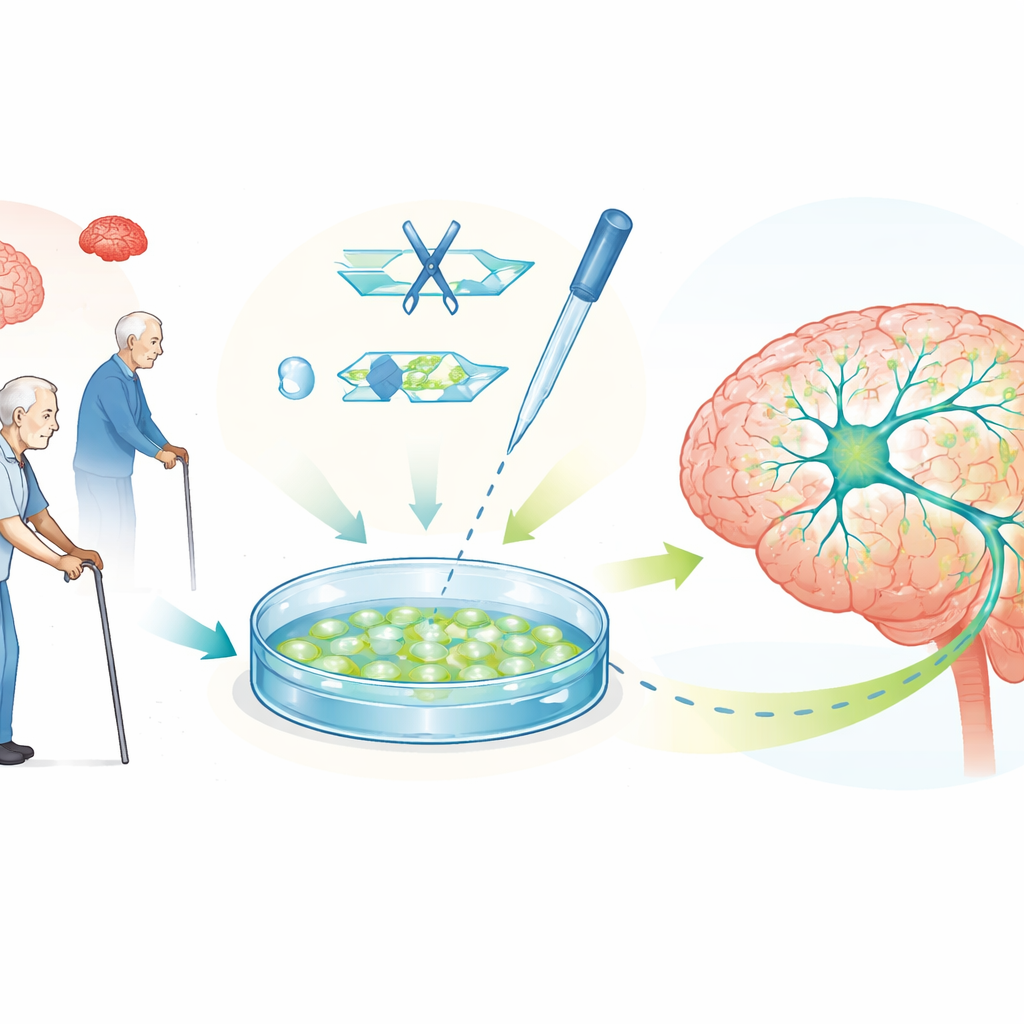
Editing genes to probe causes and test ideas
CRISPR–Cas9, a precise gene editing tool, makes these stem cell models far more powerful. By changing single letters of DNA, deleting genes, or correcting mutations, scientists can create pairs of cell lines that are identical except for one Parkinson’s risk factor. Comparing them reveals exactly how particular genes affect dopamine neuron health, energy balance, and protein build-up. The review describes many such examples, including edits in genes that control mitochondria, protein recycling, and nerve cell communication. It also covers “switch-like” versions of CRISPR that turn genes up or down without cutting DNA, and designer systems that tweak how DNA is packaged, all of which help dial alpha synuclein levels to safer ranges.
Smart tags, stress tests, and large-scale searches
Beyond simple gene cuts, CRISPR lets researchers insert glowing tags or tiny light-producing pieces into key genes. These tags allow them to watch dopamine neurons develop, track alpha synuclein clumps, and measure protein levels with great sensitivity during drug testing. Other engineered tools use light to trigger rapid alpha synuclein clumping so that toxic events unfold in days rather than years, speeding up discovery. The article also highlights genome-wide CRISPR screens that knock out thousands of genes in parallel to find hidden regulators of alpha synuclein and of the mitochondrial clean-up pathway, pointing to new drug targets that would have been hard to guess in advance.
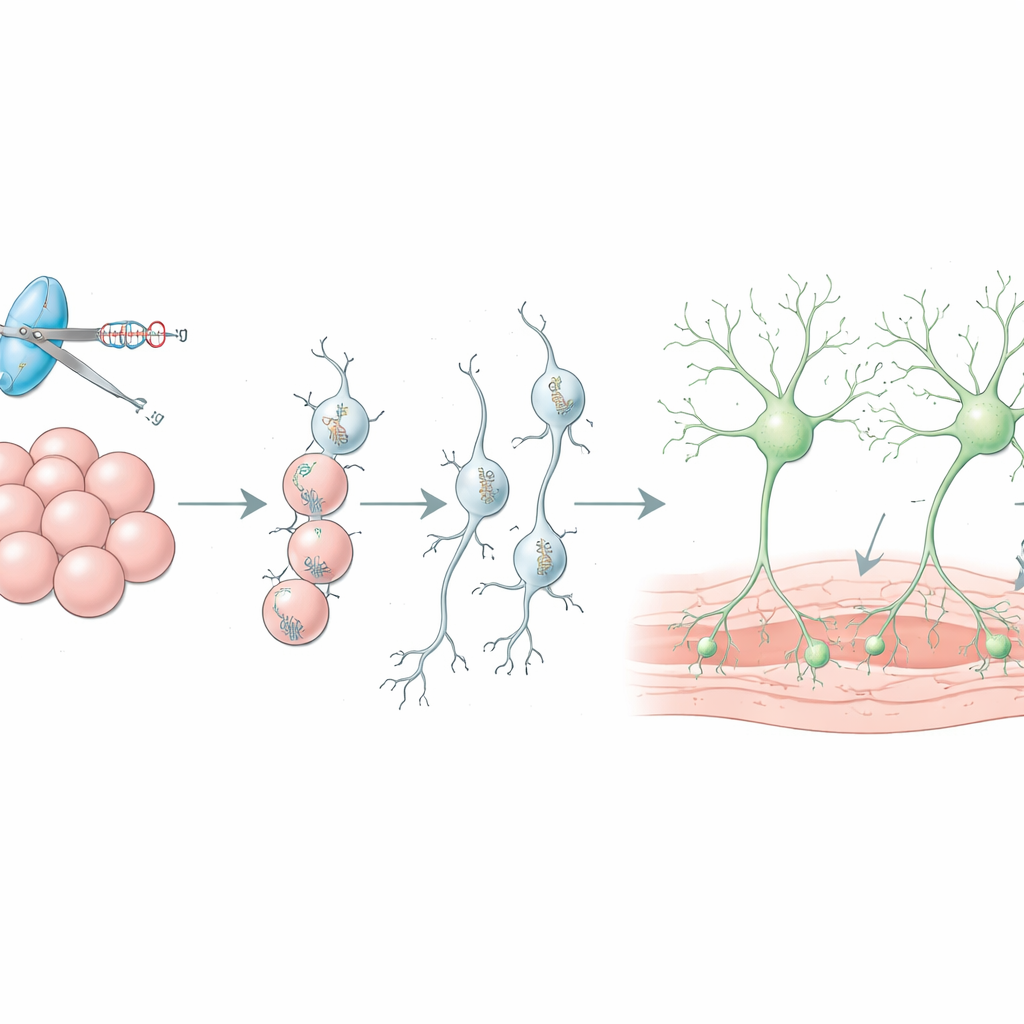
Building better cell grafts for the brain
Stem cell-derived dopamine neurons are already being tested in people as a way to replace lost cells in Parkinson’s disease, but more than ninety percent of transplanted cells often die soon after surgery, and unwanted cell types can slip into the graft. The review explains how CRISPR is used in living animals to hunt for genes that control graft survival, leading to strategies such as blocking specific immune signals with existing drugs to protect new neurons. It also describes how reporter tags help sort purer batches of dopamine neurons before implantation and how chemogenetic switches, inserted with CRISPR, allow doctors to remotely dial graft activity up or down using harmless designer drugs.
Looking ahead to future treatments
Taken together, the work summarized in this article shows that combining human stem cells with precise gene editing is reshaping how scientists study and potentially treat Parkinson’s disease. These tools create human-based models that more closely mirror the slow, age-related damage seen in patients and help design transplanted cells that are safer, more robust, and more resistant to the spread of toxic proteins. While many scientific, safety, and ethical questions remain before such approaches can become standard care, this roadmap suggests a future in which therapies aim not just to ease Parkinson’s symptoms but to protect or rebuild the vulnerable brain circuits themselves.
Citation: Park, S.B., Kim, JS., Ha, Y. et al. Human pluripotent stem cell engineering with CRISPR–Cas9 for Parkinson’s disease. Exp Mol Med 58, 993–1009 (2026). https://doi.org/10.1038/s12276-026-01679-2
Keywords: Parkinson’s disease, human stem cells, CRISPR gene editing, dopamine neurons, cell therapy