Clear Sky Science · en
Microglial CR3-mediated synaptic pruning in the dmPFC promotes the generation and maintenance of chronic muscle pain via glutamatergic dysfunction
Why sore muscles can linger for months
Most of us expect muscle aches to fade after a few days of rest. Yet for many people, muscle pain becomes a stubborn companion that lasts for months and is often tied to anxiety or low mood. This study in rats looks beyond the sore muscle itself and into the brain, asking why chronic muscle pain can become so persistent and emotionally draining. The researchers uncover a surprising culprit: tiny immune cells in the brain that quietly reshape nerve connections in a pain‑related region of the cortex.
From brief ache to long‑lasting pain
The team began by creating a well‑established model of chronic muscle pain in rats, caused by a small injection of salty solution into a calf muscle. At first, the animals reacted strongly to touch and heat, as expected after injury. But even weeks later, when the original irritation should have settled, their sensitivity to pain remained high and they moved less and avoided the center of an open arena—signs of anxiety‑like behavior. Brain scans and cellular markers pointed to reduced activity in a region called the dorsomedial prefrontal cortex, an area known to shape how we feel and react to pain.
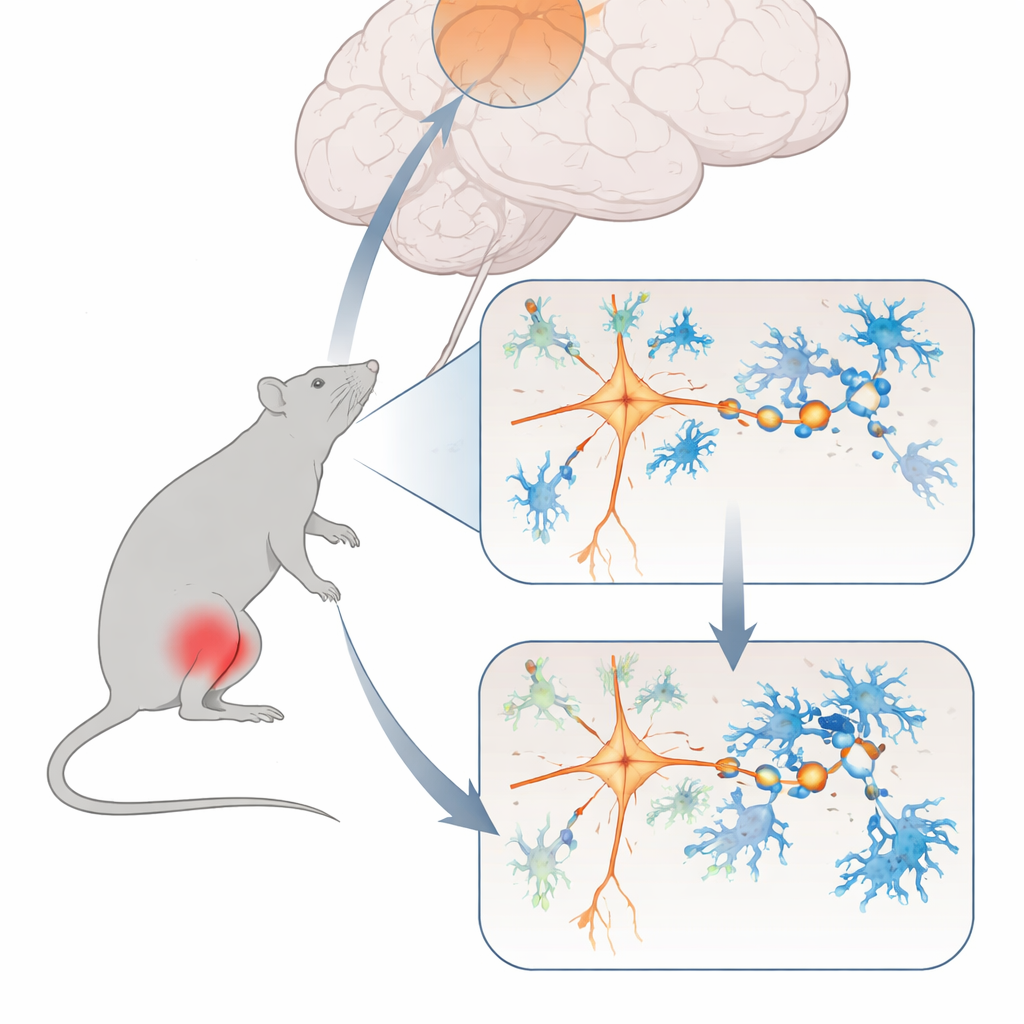
A quieted pain‑control hub in the brain
To test whether this brain region actually helps control chronic muscle pain, the researchers used light‑based and drug‑based genetic tools to turn its excitatory nerve cells up or down like a dimmer switch. When they boosted activity in these cells, the rats became less sensitive to mechanical pain and showed fewer anxiety‑like behaviors. When they dampened the same cells, even healthy animals started to behave more like chronic pain sufferers, and a relay area in the spinal cord that carries pain signals became overactive. Detailed electrical recordings revealed that, in chronic pain, the affected cortical neurons fired less and their excitatory synapses—tiny contact points that pass signals between nerve cells—became weaker and fewer in number.
Brain immune cells that eat synapses
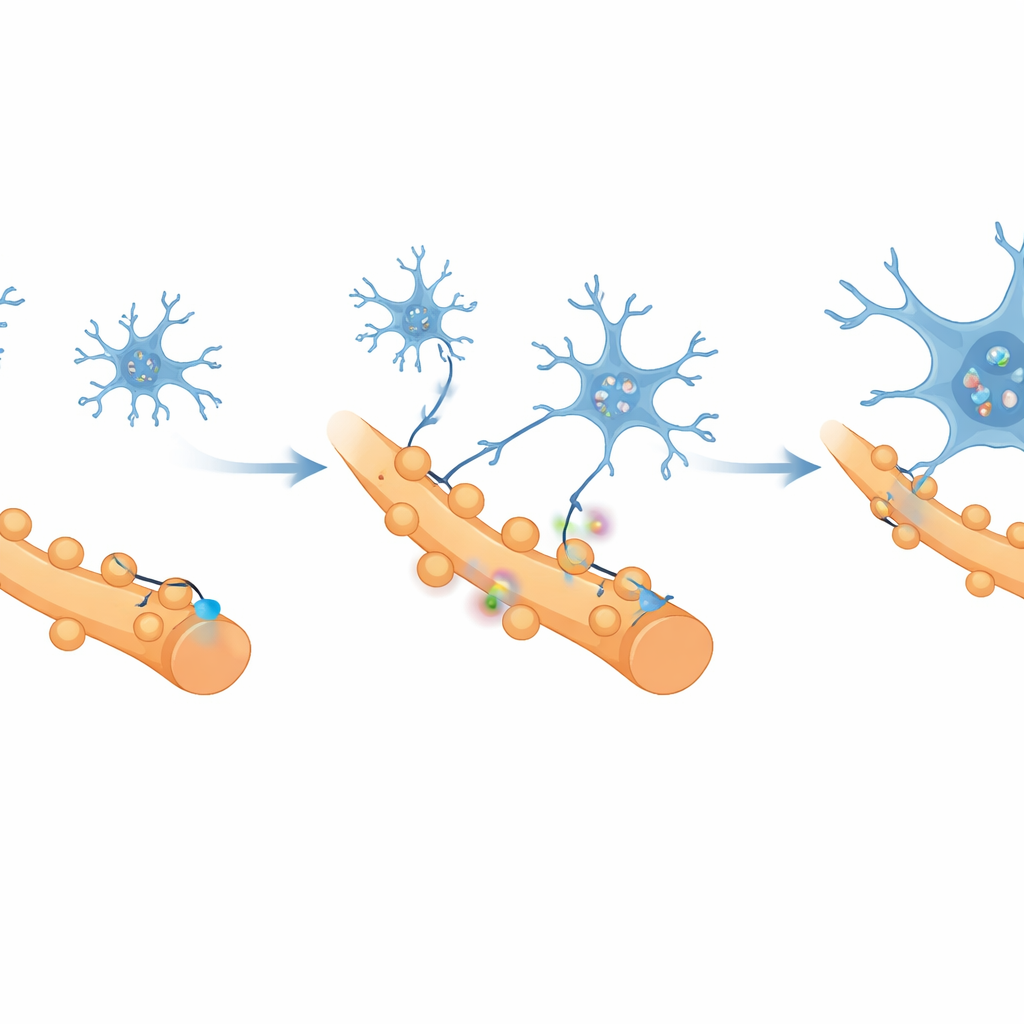
What causes this loss of healthy connections? The investigators turned to microglia, the brain’s resident immune cells, which act as gardeners that trim synapses during development and in disease. Using single‑cell RNA sequencing, they found that microglia in the pain‑related cortex shifted into a strongly pro‑inflammatory state, showing high activity in genes linked to synapse pruning. One molecule, a receptor called CR3, stood out as closely tied to pathways that remove synapses. Microscopy and flow‑cytometry experiments showed that, as pain became chronic, microglia multiplied, gathered around active neurons, and increasingly engulfed pieces of excitatory synapses, while inhibitory connections were largely spared.
Blocking the pruning rescues brain function
To see whether these microglia actually drive chronic pain, the team temporarily suppressed them with a drug delivered into the brain’s fluid. Silencing microglia reduced pain behaviors, restored cortical neuron responses to painful stimuli, and reversed structural damage at synapses. Going a step further, the scientists used a targeted viral approach to reduce CR3 specifically in microglia. This cut back their synapse‑eating behavior, increased the number and strength of excitatory synapses, and made cortical neurons more excitable again. Rats with CR3‑reduced microglia showed higher pain thresholds and were more willing to explore, suggesting both pain and anxiety had eased. When the researchers then artificially shut down the same excitatory neurons, the benefits disappeared, confirming that the relief depends on restoring these cells’ activity.
What this means for people with chronic muscle pain
Overall, the work paints a picture in which persistent muscle pain is maintained not just by ongoing signals from the body but by a remodeled brain circuit. In this model, overactive microglia in a key prefrontal area strip away excitatory synapses through CR3‑dependent pruning, leaving the region underpowered and less able to dampen pain and anxiety. By blocking microglia or their CR3 receptor, the researchers could reverse these brain changes and relieve symptoms in rats. While more studies are needed to translate this to humans, the findings suggest that future treatments for chronic muscle pain may come from calming, or carefully retraining, the brain’s own immune cells rather than targeting nerves or muscles alone.
Citation: Luo, M., Wang, L., Liang, Y. et al. Microglial CR3-mediated synaptic pruning in the dmPFC promotes the generation and maintenance of chronic muscle pain via glutamatergic dysfunction. Exp Mol Med 58, 664–680 (2026). https://doi.org/10.1038/s12276-026-01666-7
Keywords: chronic muscle pain, microglia, synaptic pruning, prefrontal cortex, neuroinflammation