Clear Sky Science · en
Methylomic signatures of tau and amyloid-beta in transgenic mouse models of Alzheimer’s disease neuropathology
Why this research matters for understanding memory loss
Alzheimer’s disease slowly robs people of memory and independence, but we still do not fully understand what drives the damage inside the brain. This study looks at tiny chemical tags on DNA, called methylation marks, in mouse models that mimic core Alzheimer’s features. By tracking how these marks change as harmful tau tangles and amyloid-beta plaques build up, the researchers aim to uncover early warning signals and potential new targets for treatment long before brain cells die. 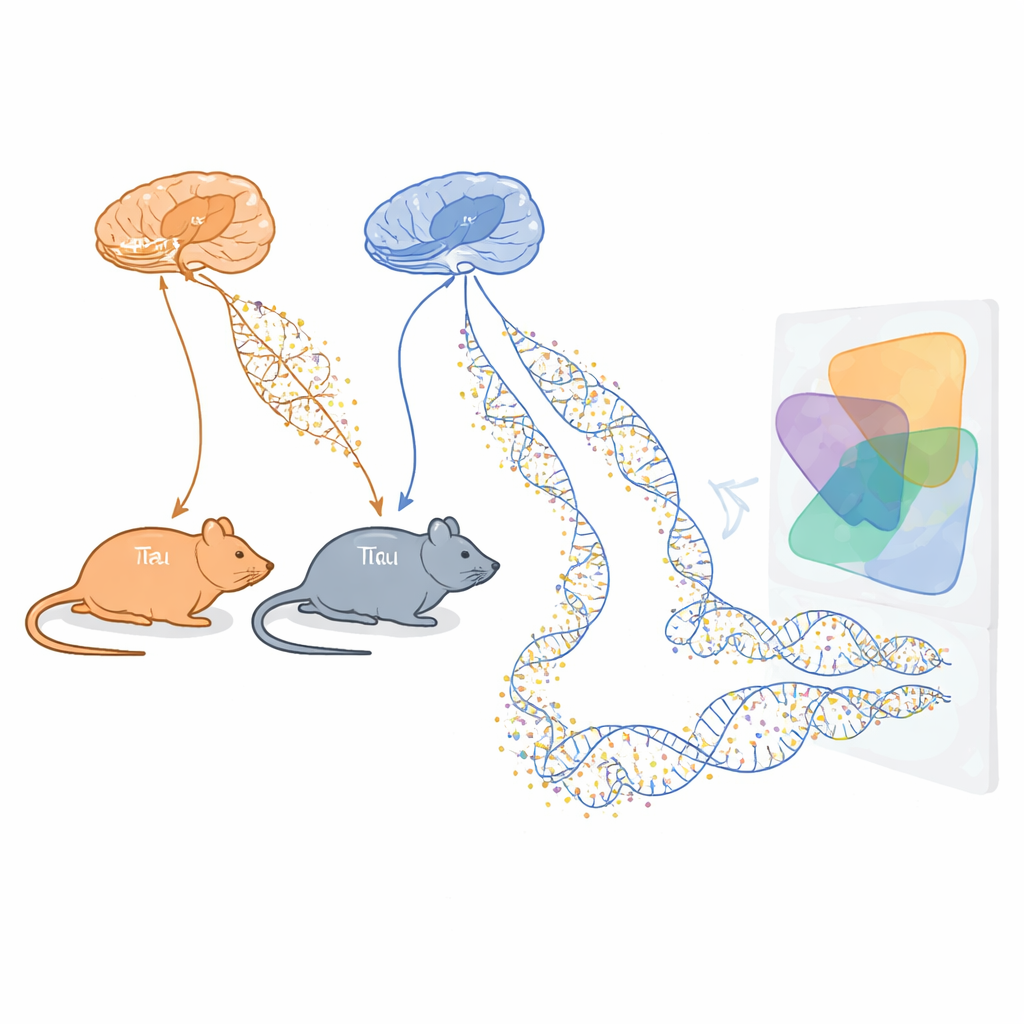
Two mouse stand-ins for human Alzheimer’s
The team used two well-established mouse lines that each model one major aspect of Alzheimer’s disease. One, called rTg4510, produces an abnormal human tau protein that forms tangles inside nerve cells. The other, called J20, carries mutations that cause excess amyloid-beta, a protein that clumps into plaques between cells. For both models, scientists collected samples from two memory-critical brain regions, the entorhinal cortex and the hippocampus, at several ages spanning early to more advanced disease-like stages. They then measured DNA methylation across more than a million sites in the genome, using both deep sequencing and a custom array to obtain a broad and reliable view of epigenetic changes over time.
What changes with tau tangles in the brain
In the tau mouse, DNA methylation changed extensively, especially in genes linked to nerve cell communication, cell death, and fat-related signaling in the brain. Many sites gained methylation as tau pathology worsened, including in genes that help control whether neurons survive or self-destruct. Some of these genes, such as those involved in protein disposal systems and in maintaining healthy synapses, also showed shifts in activity levels, suggesting that altered methylation may help switch disease pathways on or off. The hippocampus, a key hub for forming memories, showed even more pronounced methylation changes than the entorhinal cortex, and its "epigenetic age" appeared older than expected, hinting that tau may speed up the molecular aging of this vulnerable region.
How amyloid-beta plaques leave a different mark
In contrast, the amyloid-beta mouse showed fewer and generally smaller methylation changes, and these were more tightly tied to immune responses and energy factories inside cells. Sites near genes involved in inflammation, mitochondrial health, and synapse structure were particularly affected as plaques accumulated. The patterns differed strongly between entorhinal cortex and hippocampus, implying that amyloid-beta’s epigenetic effects are more region-specific and perhaps more dependent on local cell types or timing than those of tau. Together, the two models suggest that the brain responds to tau tangles and amyloid-beta plaques through overlapping but distinct molecular routes, with tau having a broader footprint on the genome.
Shared epigenetic fingerprints across mice and humans
To test whether mouse findings truly reflect human disease, the researchers compared their results with large DNA methylation studies of human Alzheimer’s brains. They found striking overlaps. Key genes such as ANK1 and PRDM16 showed similar methylation shifts in mouse models and in brain tissue from people with Alzheimer’s. Many shared changes occurred in non-neuronal support cells, matching human data that highlight the role of glial cells in disease progression. These cross-species echoes suggest that the mouse methylation patterns are not just by-products of genetic engineering, but capture core mechanisms that also operate in human brains. 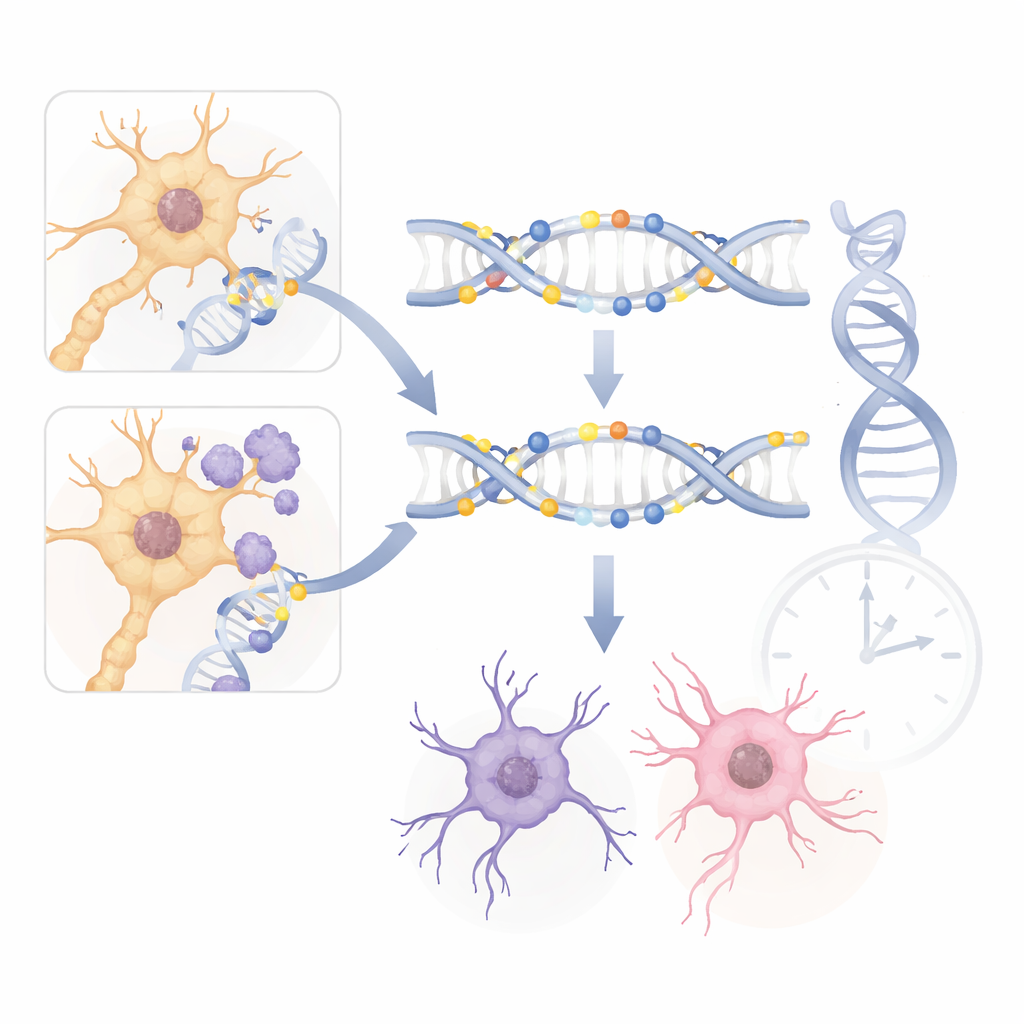
What it all means for future treatments
For a layperson, the main message is that Alzheimer’s-related brain changes leave a detectable chemical imprint on DNA long before symptoms appear. Tau tangles, in particular, are associated with widespread shifts in methylation that track with disease severity and accelerated molecular aging. Amyloid-beta plaques trigger their own set of changes, especially in immune and energy pathways. Because many of these epigenetic signatures mirror those in human brains, they could help scientists identify early biomarkers or design drugs that nudge harmful gene programs back toward a healthier state. This work does not yet offer a cure, but it maps out a more detailed molecular trail from protein build-up to brain cell damage, bringing researchers a step closer to new ways of slowing or preventing Alzheimer’s disease.
Citation: Leung, S.K., Walker, E.M., Policicchio, S. et al. Methylomic signatures of tau and amyloid-beta in transgenic mouse models of Alzheimer’s disease neuropathology. npj Dement. 2, 23 (2026). https://doi.org/10.1038/s44400-026-00074-y
Keywords: Alzheimer’s disease, epigenetics, DNA methylation, tau and amyloid-beta, mouse models