Clear Sky Science · en
Catalytic asymmetric activation of bicyclobutanes
Why tiny, tense rings matter
Chemists are always looking for new ways to reshape simple oil-like molecules into the complex, three-dimensional structures found in medicines and advanced materials. Some of the most promising building blocks are tiny, highly strained carbon rings that store a great deal of chemical "spring" energy. The problem is that once you touch that spring, it tends to snap open in many different ways at once, making it hard to control the final shape. This article describes a way to tame one of the most unruly of these rings and turn it into useful, single-handed (chiral) products with remarkable precision.
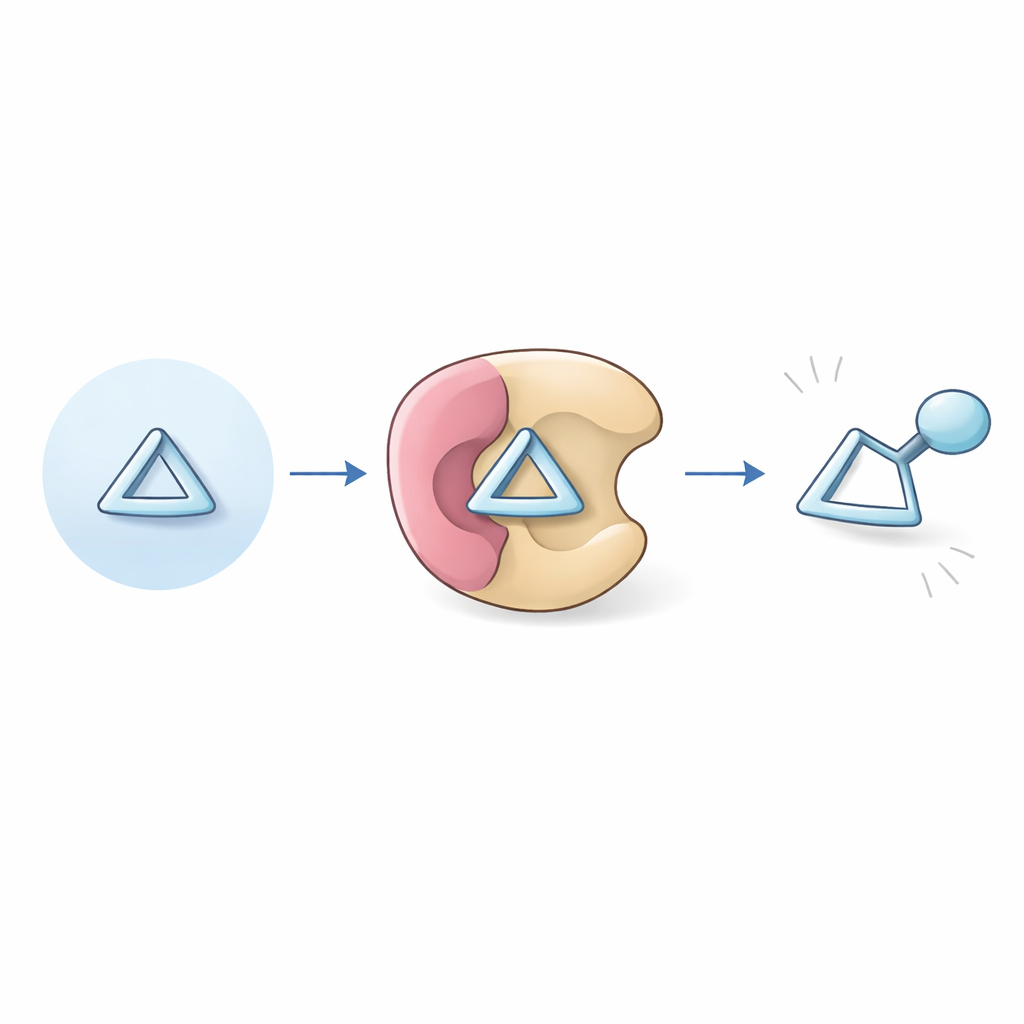
A chemical spring under control
The study focuses on bicyclobutanes, extremely compact four-carbon rings that resemble two fused triangles. Because their bonds are bent far from their preferred angles, they are primed to burst open, releasing stored strain energy. Earlier work had shown how related strained structures could be coaxed into forming new rings or bioactive shapes, but bicyclobutanes made only of carbon and hydrogen were considered especially difficult: they lack helpful "handles" such as oxygen or nitrogen atoms that usually guide reactions, and they tend to break apart or rearrange in multiple, competing ways.
A tailor-made catalyst pocket
To solve this, the authors designed a family of strong, organic acids that act a bit like artificial enzyme pockets. These catalysts, called iminoimidodiphosphoric acids (iIDPs), are both very acidic and very crowded in three dimensions. When a bicyclobutane and an alcohol enter this pocket together, the catalyst briefly protonates (acidifies) the tiny ring, nudging it toward opening, while the tight space around it steers how that opening happens. Under optimized conditions—using a carefully tuned iIDP, cold temperatures, and a simple hydrocarbon solvent—the team converts bicyclobutanes into tertiary cyclopropylcarbinyl ethers: molecules that still contain a compact three-membered ring, now bearing an attached alcohol fragment.
From strained rings to medicine-ready building blocks
One of the key achievements is control over handedness. Many biologically active molecules exist in left- and right-handed forms, and often only one works as intended. The new reaction consistently delivers one hand with high preference, reaching "enantioselectivity" values up to 98:2. The method works for a broad range of bicyclobutane starting materials and alcohol partners, including simple alcohols such as methanol and butanol, more complex fragments related to natural fragrances (citronellol), and others. The ether products can then be smoothly transformed into chiral tertiary alcohols—highly sought-after components in pharmaceutical and materials chemistry—without losing their carefully set handedness.
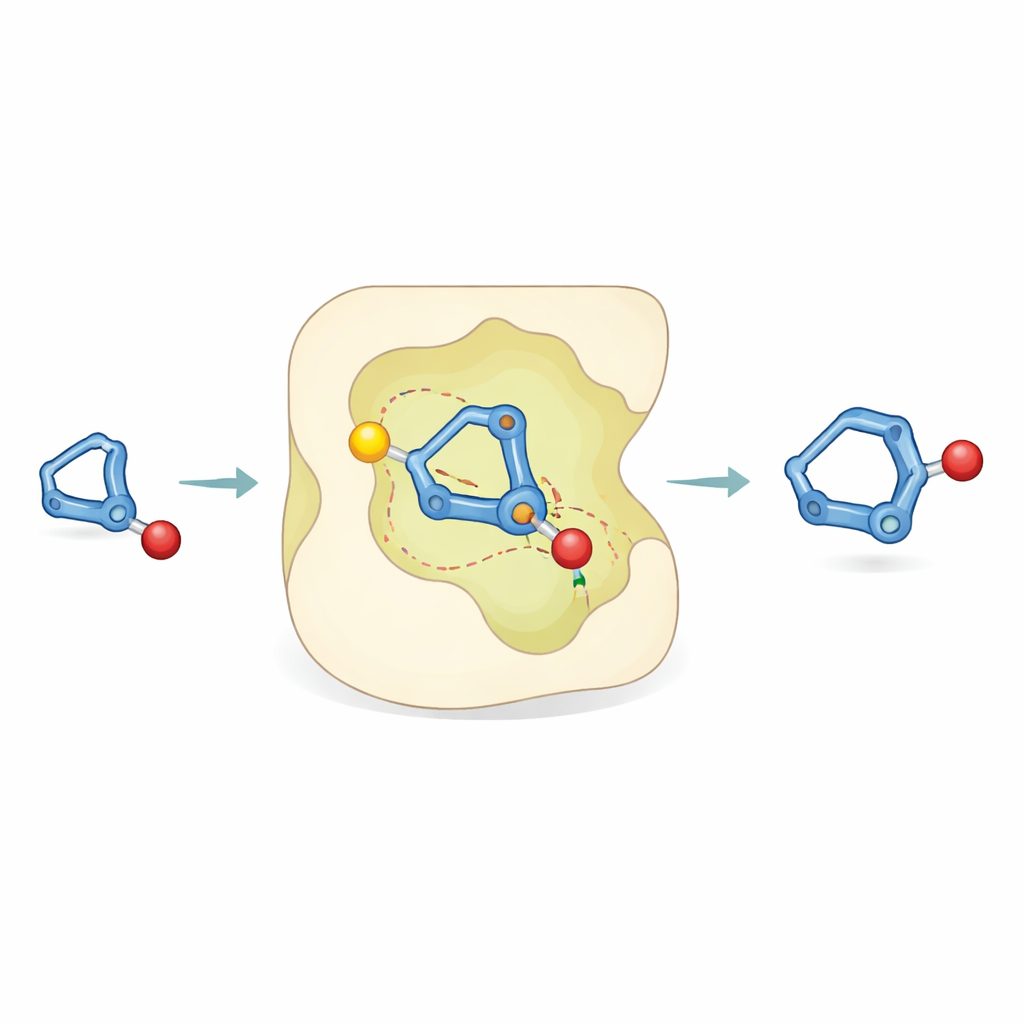
Peeking inside the reaction
To understand how such accuracy is possible, the researchers combined experiments with computer simulations. Lab studies using labeled hydrogen atoms showed that protonation of the bicyclobutane happens in a preferred way, and that unwanted breakdown occurs in parallel rather than feeding into the desired product. Computational chemistry then revealed that the key step is an "asynchronous concerted" process: protonation of the strained ring and bond formation to the alcohol happen almost together. Within the catalyst pocket, the developing positively charged intermediate forms specific, directional contacts—hydrogen-bond-like attractions—between its carbon–hydrogen bonds and a Lewis-basic (electron-rich) region of the catalyst. These non-covalent interactions stabilize one transition shape more than its mirror image, explaining why one hand of the product dominates.
What this means for future molecules
In simple terms, the researchers have built an artificial chemical glove that can grasp an especially tense carbon ring, let it relax in a controlled way and attach an alcohol in a single, preferred orientation. This shows that even the most reactive, spring-loaded hydrocarbons can be steered with enzyme-like precision using carefully shaped organic catalysts, without relying on metals or pre-installed guiding groups. The strategy opens a path to a wide variety of compact, three-dimensional, chiral building blocks that could feed into drug discovery, polymers and other advanced materials, all by learning to direct how molecular springs release their energy.
Citation: Shi, F., Frank, N., Leutzsch, M. et al. Catalytic asymmetric activation of bicyclobutanes. Nat. Synth 5, 527–533 (2026). https://doi.org/10.1038/s44160-025-00951-z
Keywords: strain-release chemistry, bicyclobutane activation, asymmetric catalysis, chiral tertiary alcohols, organocatalyst design