Clear Sky Science · en
Delivering biomedicines with stimuli‑responsive biomaterials
Smart Materials That Know Where Medicines Should Go
Many powerful modern medicines never reach their full potential because they spread throughout the body, wear off too quickly, or cause harsh side effects. This article explores a new class of “smart” materials designed to carry drugs, proteins, genetic treatments, vaccines, and even living cells directly to where they are needed, and to release them only when the right signal appears. These responsive delivery systems could make treatments more effective, gentler, and easier for patients to live with.
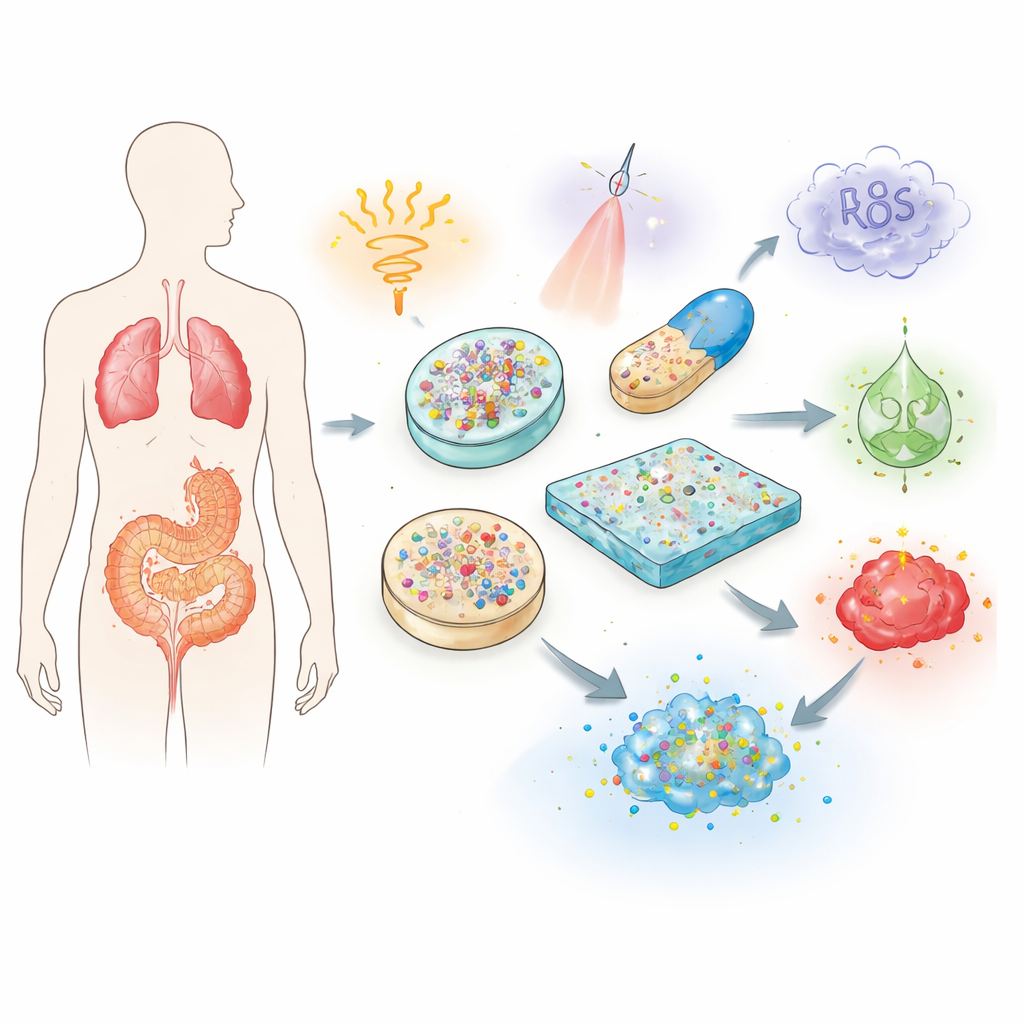
Why Conventional Treatments Need an Upgrade
Traditional pills and injections send medicine on a largely uncontrolled journey through the body. Much of a drug may be broken down, washed out, or soaked up by healthy organs long before it reaches a tumor, an inflamed joint, or a damaged heart. To compensate, doctors often prescribe higher or repeated doses, raising the risk of side effects such as organ damage or immune reactions. The review explains how stimuli-responsive biomaterials tackle these problems by acting as protective shuttles. Built from biocompatible polymers, gels, and nanoparticles, they can shield fragile payloads, extend their lifetime in the body, and fine-tune when and where the medicine is released.
Materials That Listen to Their Environment
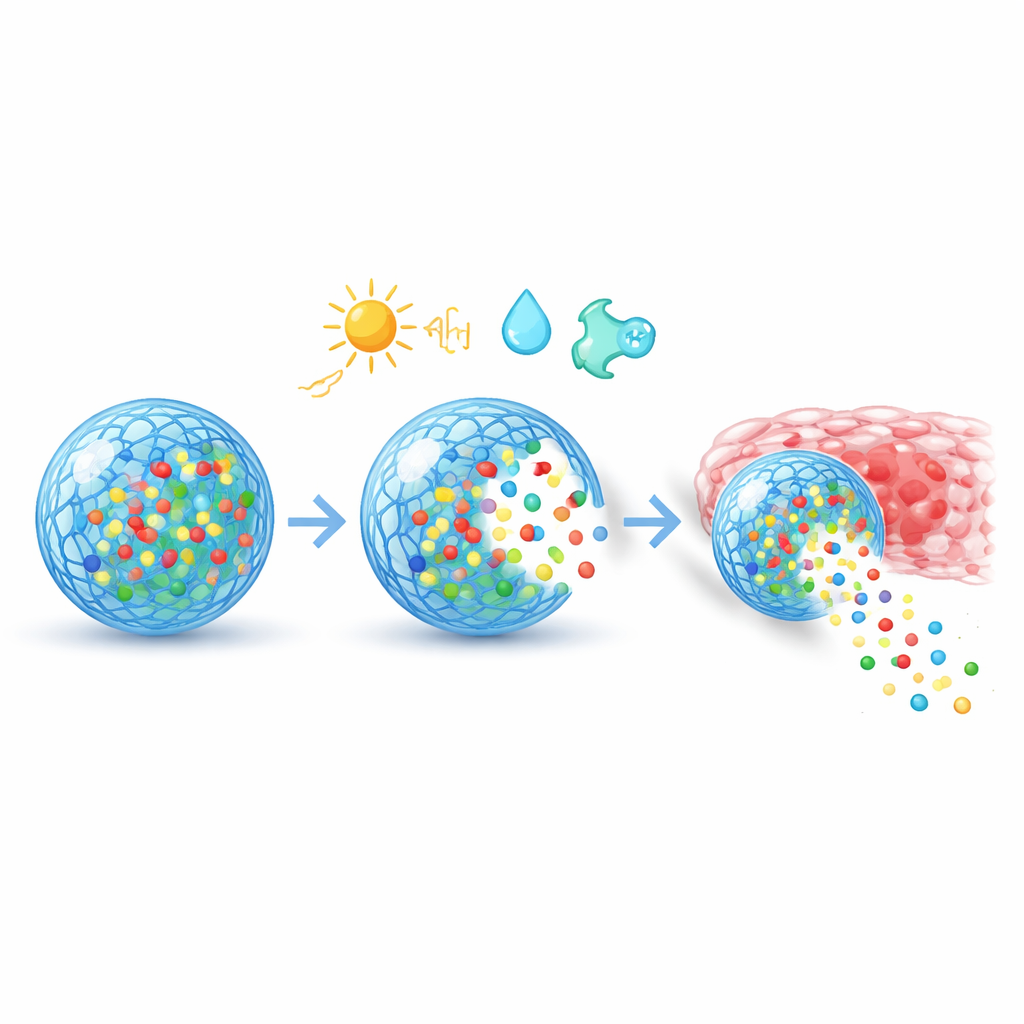
The core idea is to give a material the ability to “sense” its surroundings and change its behavior accordingly. The authors describe three broad families of triggers. Physical cues such as heat, light, magnetic fields, and ultrasound can be applied from outside the body to turn release on or off at chosen moments. Chemical cues such as acidity, redox balance, and salt levels differ between healthy and diseased tissues, allowing carriers to open only in, for example, the acidic microenvironment of tumors. Biological cues include enzymes, glucose, and reactive oxygen species that are elevated in specific diseases; these can cut special links in a material, soften a gel, or change its solubility, freeing the drug only in the problem area. Some advanced systems even combine several triggers to gain more precise control.
How Smart Carriers Steer and Release Treatments
Beyond sensing, these materials are engineered to navigate the body and interact with cells. Their surfaces can be decorated with short peptides, antibodies, or other binding agents that recognize markers on cancer cells, inflamed tissues, or specific organs, guiding the carriers to their targets. Once taken up by cells, clever chemistry helps them escape internal “waste bins” and deliver genetic cargo such as mRNA or CRISPR components to the right compartment. By adjusting porosity, charge, and degradability, designers can create slow, steady release over weeks, sharp bursts triggered by a light pulse or temperature change, or multi-step patterns that follow the phases of healing. At the tissue level, special coatings help particles slip through mucus, stick to wounds, or avoid being cleared too quickly by the immune system.
Real-World Uses From Insulin to Cancer Vaccines
The review surveys a rapidly expanding list of practical applications. For protein drugs and peptides, heat- or light-responsive implants and hydrogels can provide on-demand dosing without frequent injections. pH-sensitive particles that briefly open the gut’s tight junctions offer a route to swallow large biologic drugs that normally must be injected. Gene therapies benefit from responsive nanoparticles and gels that protect delicate DNA and RNA, then release them only inside target cells. Small-molecule drugs, such as chemotherapy agents, are loaded into carriers that respond to enzymes or oxidative stress found in tumors, concentrating treatment at the cancer site. In cell and gene therapy, engineered scaffolds and protective coatings improve how transplanted cells home to injured organs and survive there. For vaccines and immunotherapies, pH-triggered microneedle patches and 3D-printed immune-cell scaffolds can time the appearance of antigens and immune signals for stronger, longer-lasting protection.
Promise, Hurdles, and the Road Ahead
To a non-specialist, the takeaway is that these smart materials act like programmable delivery trucks for medicine, opening their cargo doors only under the right conditions. Early animal studies and some clinical examples show that this can boost effectiveness and cut side effects across cancer care, diabetes, wound healing, and regenerative medicine. However, the authors stress that important questions remain about long-term safety, manufacturing at large scale, and how these complex materials behave in the diverse, ever-changing environment of real patients. They foresee rapid progress as machine learning, advanced 3D and 4D printing, and tiny embedded sensors are woven into material design. If these challenges are met, stimuli-responsive biomaterials could become a foundation for future therapies that adapt to each person’s biology and deliver treatment exactly where and when it is needed.
Citation: Singh, H., Darban, Z., Ebrahimi, A. et al. Delivering biomedicines with stimuli‑responsive biomaterials. Commun Mater 7, 122 (2026). https://doi.org/10.1038/s43246-026-01163-4
Keywords: smart drug delivery, stimuli-responsive biomaterials, targeted therapy, nanomedicine, regenerative medicine