Clear Sky Science · en
Developing virtual physiology of human tumor tissue for malignancy assessment
Why squeezing tumors matters
When we think about cancer, we usually picture rogue cells growing out of control. But tumors are also physical objects that push, pull, and squeeze the tissues around them. Those invisible forces can help decide whether a tumor stays relatively contained or becomes life‑threatening and hard to treat. This study builds a detailed "virtual twin" of real human breast tumor samples to measure how stresses inside the tissue relate to how dangerous a tumor may be—without needing to experiment directly on patients.
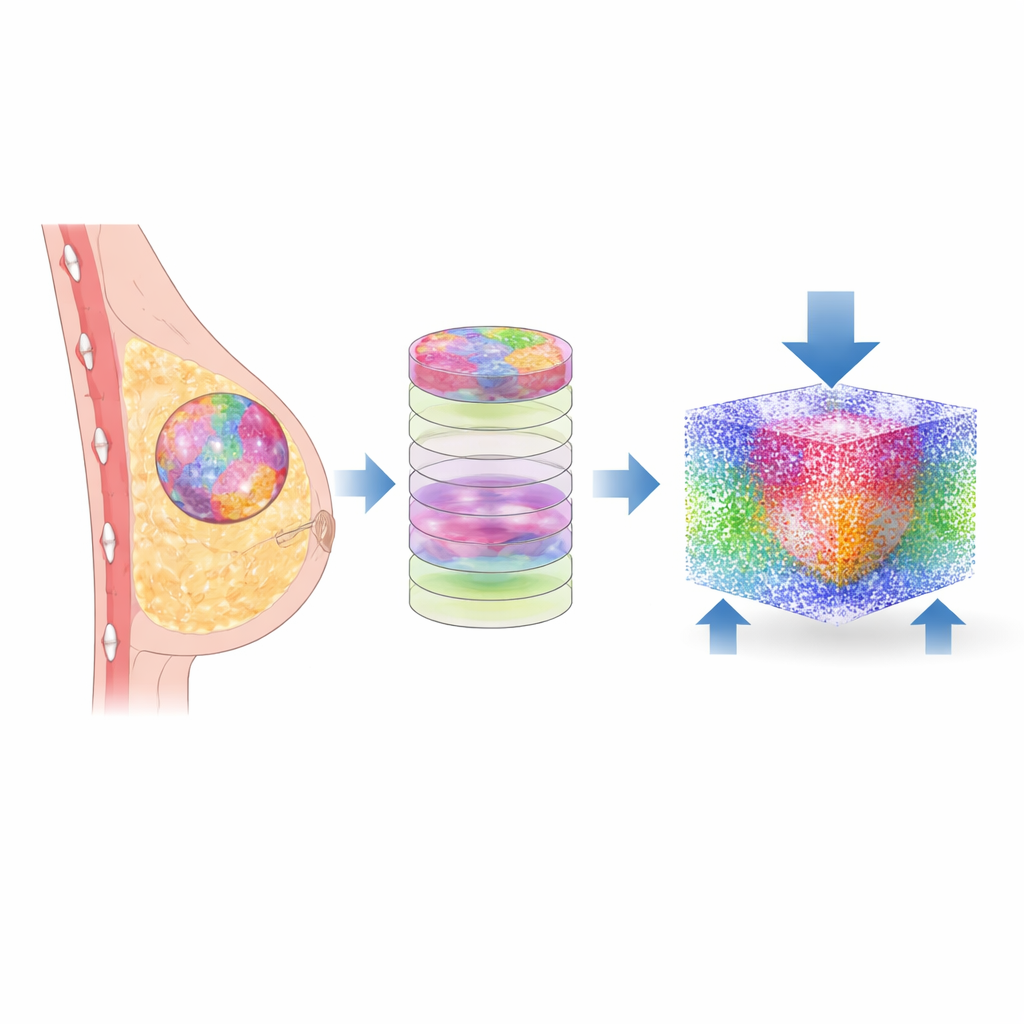
From a biopsy slide to a digital twin
The researchers started with tiny breast tissue biopsies similar to those examined by pathologists every day. Instead of traditional stains alone, they used Fourier Transform Infrared (FTIR) microscopy, which reads the chemical fingerprints of tissue. With machine learning, each pixel in the image was classified into one of six components: malignant cells, non‑cancerous cells, several types of supporting tissue (stroma), and other elements. Custom software then converted these flat images into stacked layers, building a three‑dimensional map of each patient’s tissue, preserving the complex patchwork of different regions exactly as seen in the real sample.
Turning tissue into physics
To explore how these structures behave under load, the team translated the 3D maps into a particle‑based computer model. Each tiny piece of tissue was represented by interacting particles with mechanical properties—such as stiffness and density—drawn from prior measurements of breast tissue. Using a method called smoothed particle hydrodynamics, they simulated what happens when a gentle, physiologically realistic pressure is applied from one side of the tissue, while the opposite side is held fixed. This allowed them to calculate how stress and strain—how much the tissue is squeezed and deformed—spread through every component of twelve different patient samples.
What the virtual tumors revealed
The simulations showed that tumors are not stressed equally. Samples in which malignant areas were broken into many small, scattered islands experienced much higher local stresses than tumors where malignant regions formed larger, more continuous patches. The same pattern appeared for the surrounding tissue types: small, disconnected patches of any component tended to bear higher loads than broad, continuous regions. Neighborhood also mattered. When a stiff region sat next to a much softer one, both experienced elevated stresses, whereas clusters surrounded by tissue of similar stiffness were less strained. In some cases, even samples classified as normal but with highly mixed, mosaic‑like structure showed stress levels comparable to clearly malignant tissues.
Forces that may tip tissue toward cancer
These internal stresses were not just small ripples. In the most heterogeneous tissues, predicted compressive stresses reached levels that previous experiments have shown can nudge healthy cells toward malignant behavior by altering how they grow, move, or die. Stiffer, malignant regions tended to deform less than their softer neighbors, but they contributed to concentrating stress in nearby components. The model also tracked how the surface areas of different tissue regions changed over time, revealing that small, disconnected components not only felt higher stresses but also underwent larger shape changes, which could further disturb the local environment.
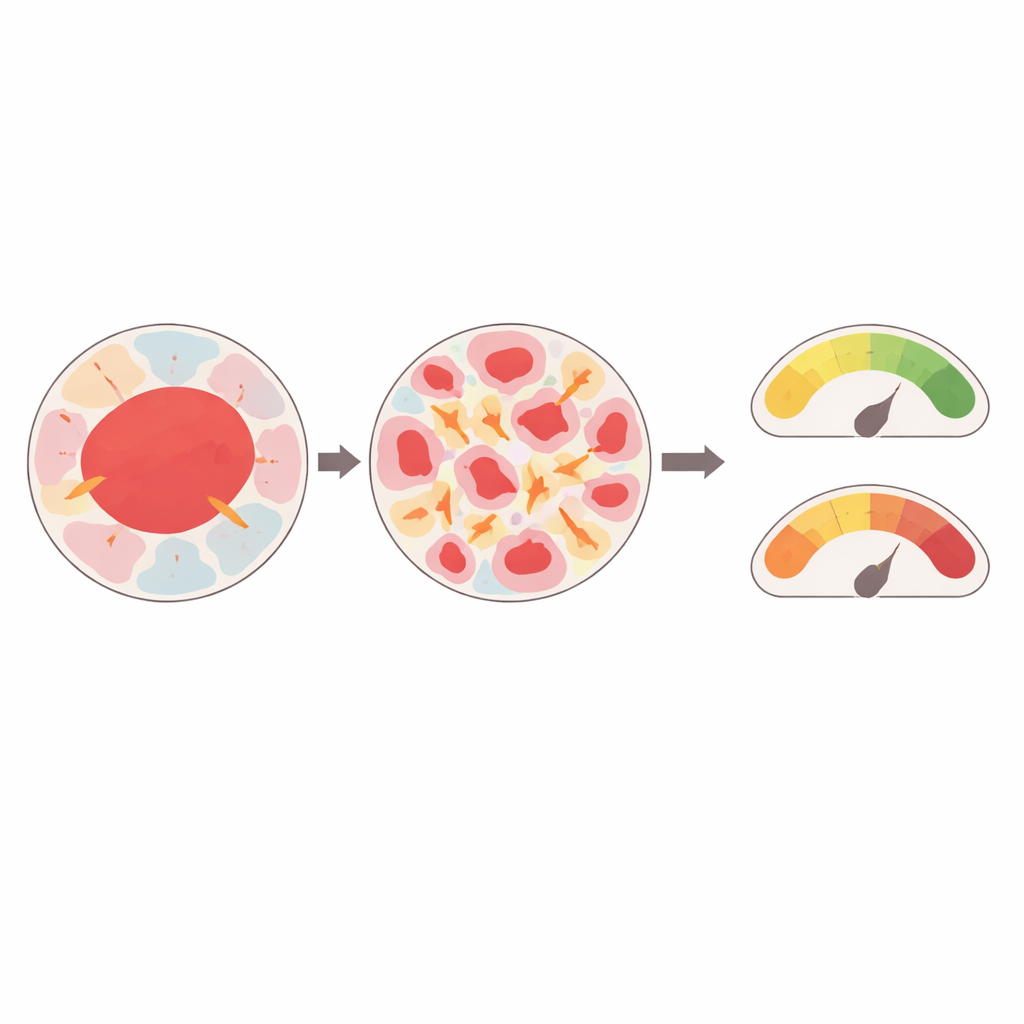
Toward personalized mechanical fingerprints of tumors
By linking real biopsy images to detailed physical simulations, the study delivers a proof‑of‑concept "virtual physiology" for breast tumors. It suggests that how a tumor is built—how patchy it is, how big each region is, and which tissues touch each other—strongly shapes the mechanical forces acting within it. Those forces, in turn, fall within ranges known to promote malignant transformation and therapy resistance. In the future, similar digital twins might help flag seemingly normal but mechanically stressed tissues that are at risk of becoming cancerous, or guide doctors toward treatment plans tailored to a tumor’s unique mechanical fingerprint as well as its genetics.
Citation: Arbabi, S., Vincent, H., Hansen, E. et al. Developing virtual physiology of human tumor tissue for malignancy assessment. npj Precis. Onc. 10, 136 (2026). https://doi.org/10.1038/s41698-026-01316-1
Keywords: breast cancer, tumor mechanics, digital twin, tumor microenvironment, computational oncology