Clear Sky Science · en
TLR7/8 signaling balances cytokine responses in neonatal monocytes
Why newborn immunity matters
Newborn babies face a perfect storm: their first encounters with germs happen just as their immune systems are still learning the rules. This study asks a crucial question for parents and doctors alike—why are newborns so vulnerable to hospital-acquired infections, and can early immune signals from viruses change how babies handle dangerous bacteria like E. coli? By zooming in on a specific type of white blood cell called a monocyte, the researchers reveal how tiny sensing devices inside these cells can tilt the balance between helpful defense and harmful, lingering inflammation.
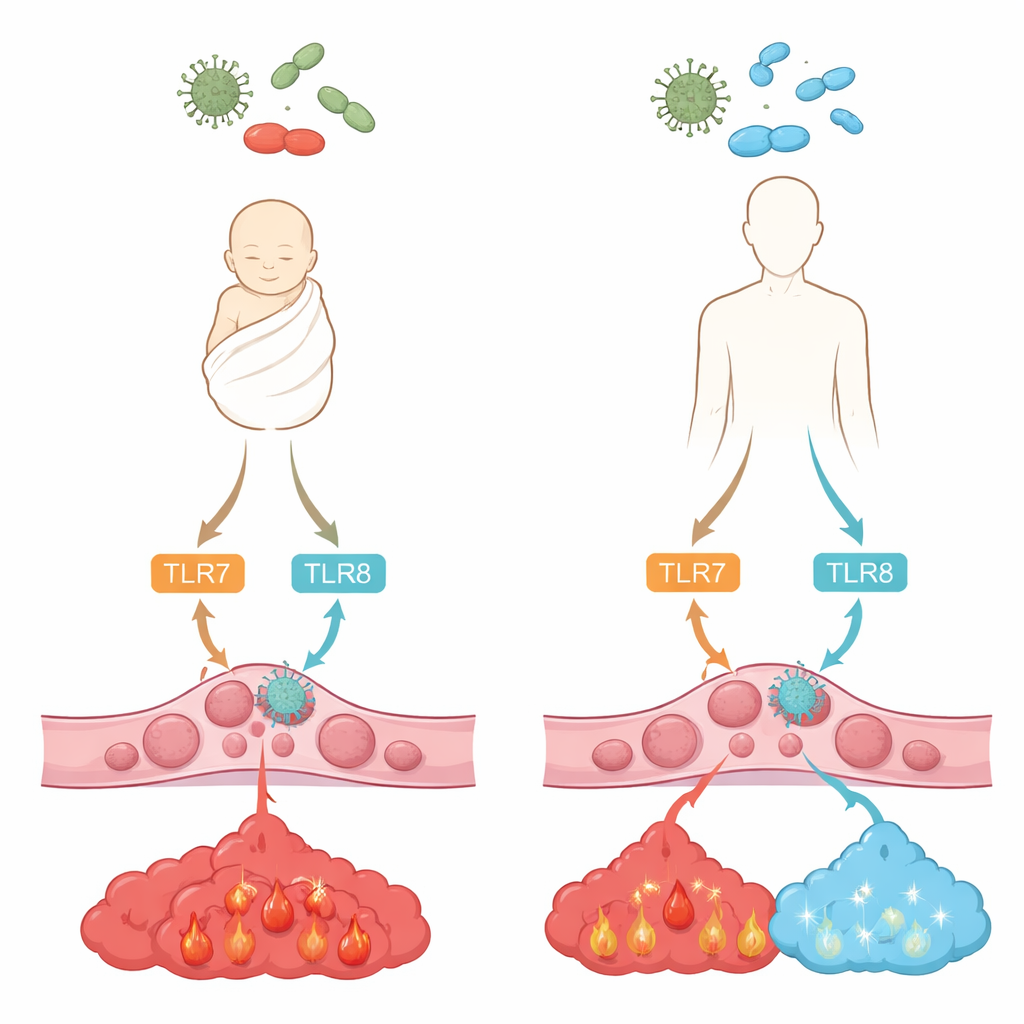
How cells sense invading germs
Our immune cells use molecular “alarm bells” known as Toll-like receptors to detect pieces of viruses and bacteria. Two of these, called TLR7 and TLR8, sit inside cells and recognize single-stranded RNA, a genetic material common in many viruses and some bacteria. When triggered, they set off internal signal chains that lead to the release of chemical messengers, or cytokines, that shape inflammation and antiviral defense. The team compared monocytes from adult blood with those from umbilical cord blood of healthy newborns, asking how strongly TLR7 and TLR8 are present and how they respond when activated by two drug-like compounds that mimic viral RNA.
Different wiring in newborn and adult cells
The researchers found that newborn monocytes carry similar amounts of TLR7 as adults but noticeably less TLR8. Despite this, when they switched on these receptors with the test compounds, both adult and newborn cells released comparable levels of the strong inflammatory messenger TNF-alpha and the versatile cytokine IL-6. The big difference appeared in IL-10, a calming signal that helps shut down inflammation before it becomes damaging. Adult cells produced plenty of IL-10 after TLR7/8 stimulation, but newborn cells produced almost none, even though they could make IL-10 when another pathway was engaged. This pattern suggests that newborn monocytes are wired to mount robust inflammation but lack an important “brake pedal.”
Inside the signaling control room
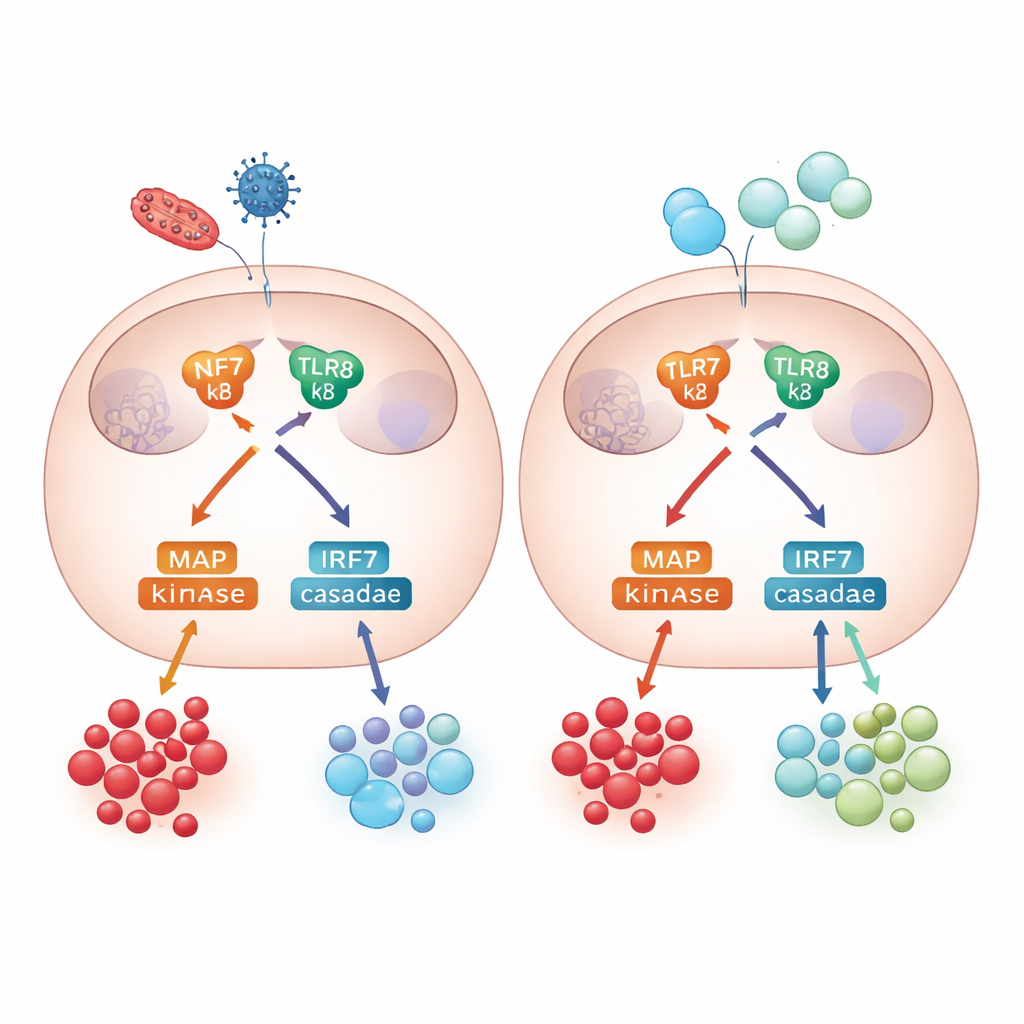
To understand how these differences arise, the team probed the internal signaling machinery that links TLR7/8 activation to cytokine release. Both adult and newborn monocytes engaged major signal routes, including NF-κB and a group of enzymes called MAP kinases. Blocking one of these enzymes, p38, had only modest effects on inflammatory signals like TNF-alpha and IL-6 but strongly reduced IL-10 production in adult cells, underscoring that anti-inflammatory control depends on this pathway. The study also examined antiviral responses: both adult and newborn monocytes activated the transcription factor IRF7 and produced type I interferons, key antiviral cytokines, showing that antiviral defenses were largely intact in newborns even though their regulatory feedback was weaker.
When a viral warning meets a bacterial threat
Because real-world infections often occur in sequence—such as a virus followed by a bacterial invader—the researchers mimicked this scenario in the lab. They first stimulated TLR7/8 in monocytes, then exposed the cells to fluorescent E. coli to track how well they engulfed and killed the bacteria. Prior TLR7/8 activation changed the levels of surface molecules involved in bacterial uptake and generally reduced the cells’ ability to swallow E. coli, especially in newborn monocytes. Yet, intriguingly, pre-treatment with the TLR7-focused compound imiquimod led to reduced survival of E. coli in newborn cells, hinting that while they take up fewer bacteria, they may kill them more effectively once inside. Cytokine release during these co-challenges did not show strong synergy, but adults again displayed a more balanced mix of inflammatory and anti-inflammatory signals compared with newborns.
What this means for fragile newborns
Taken together, the work paints a picture of newborn monocytes that can recognize viruses and bacteria and mount solid inflammatory and antiviral responses, but that lack the IL-10 “off switch” that helps adults avoid excessive, lingering inflammation. This skewed balance may contribute to newborns’ susceptibility to severe infections and complications such as sepsis. At the same time, the finding that a TLR7-targeting compound can decrease bacterial survival in newborn cells suggests that carefully tuned drugs might one day boost protection against opportunistic infections. Any such approach would need thorough testing, but this study offers a mechanistic roadmap for understanding—and potentially improving—early-life immune defenses.
Citation: Dreschers, S., Heiler, E., Oppermann, L. et al. TLR7/8 signaling balances cytokine responses in neonatal monocytes. Sci Rep 16, 12202 (2026). https://doi.org/10.1038/s41598-026-46534-6
Keywords: newborn immunity, Toll-like receptors, monocytes, neonatal sepsis, inflammation