Clear Sky Science · en
Cholesterol-rich diet exacerbates steatohepatitis in the STAM mouse model
Why this matters for everyday health
Fatty liver disease is no longer just a concern for people who drink heavily; it increasingly affects those living with obesity and type 2 diabetes. This study asks a surprisingly practical question: does the cholesterol in our diet simply raise blood levels, or can it actively drive the liver from a relatively silent fat buildup into a much more dangerous, scarred, and inflamed state? By fine‑tuning a widely used mouse model of advanced diabetes, the researchers show that adding extra dietary cholesterol sharply worsens liver injury, helping scientists build better test beds for future drugs.
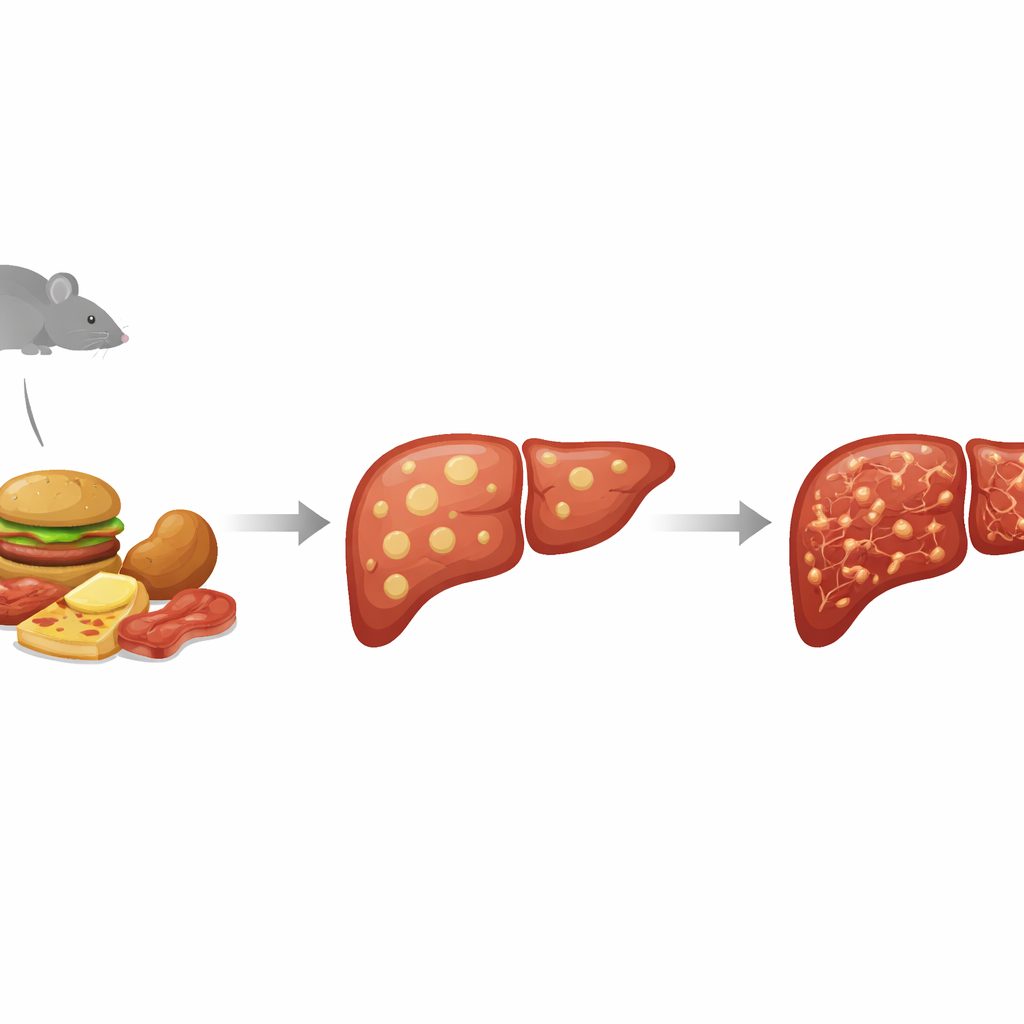
From simple fat to a sick, scarred liver
Doctors now group a wide range of fatty liver problems under the umbrella of metabolic dysfunction–associated steatotic liver disease (MASLD). At the mild end, liver cells simply store too much fat. At the severe end, the organ becomes inflamed and scarred, a condition called metabolic dysfunction–associated steatohepatitis (MASH), which can progress to cirrhosis and liver cancer. Because it is difficult and risky to study this whole progression in people, researchers rely on animal models that mimic both the metabolic problems of diabetes and the stepwise damage inside the liver. The STAM mouse model does this by chemically damaging the insulin‑producing cells in the pancreas and then feeding the animals a high‑fat diet, pushing them into a diabetes‑like state where liver disease develops rapidly.
Turning up the dial with extra cholesterol
The team asked what happens if the standard high‑fat diet in this model is replaced by one that is not only rich in fat but also in cholesterol and certain vegetable oils. Male mice were given the STAM treatment or left untreated, then fed either the classic high‑fat diet or the high‑fat, high‑cholesterol version and followed for up to 16 weeks. As expected, the STAM mice developed high blood sugar and lost body fat, reflecting severe diabetes. What changed with the cholesterol‑rich diet was the liver: these mice showed bigger, fattier livers earlier in life, with a mix of large and small fat droplets in their liver cells. Their overall disease scores—capturing fat buildup, inflammation, and injured, swollen liver cells—were highest in the high‑cholesterol STAM group.
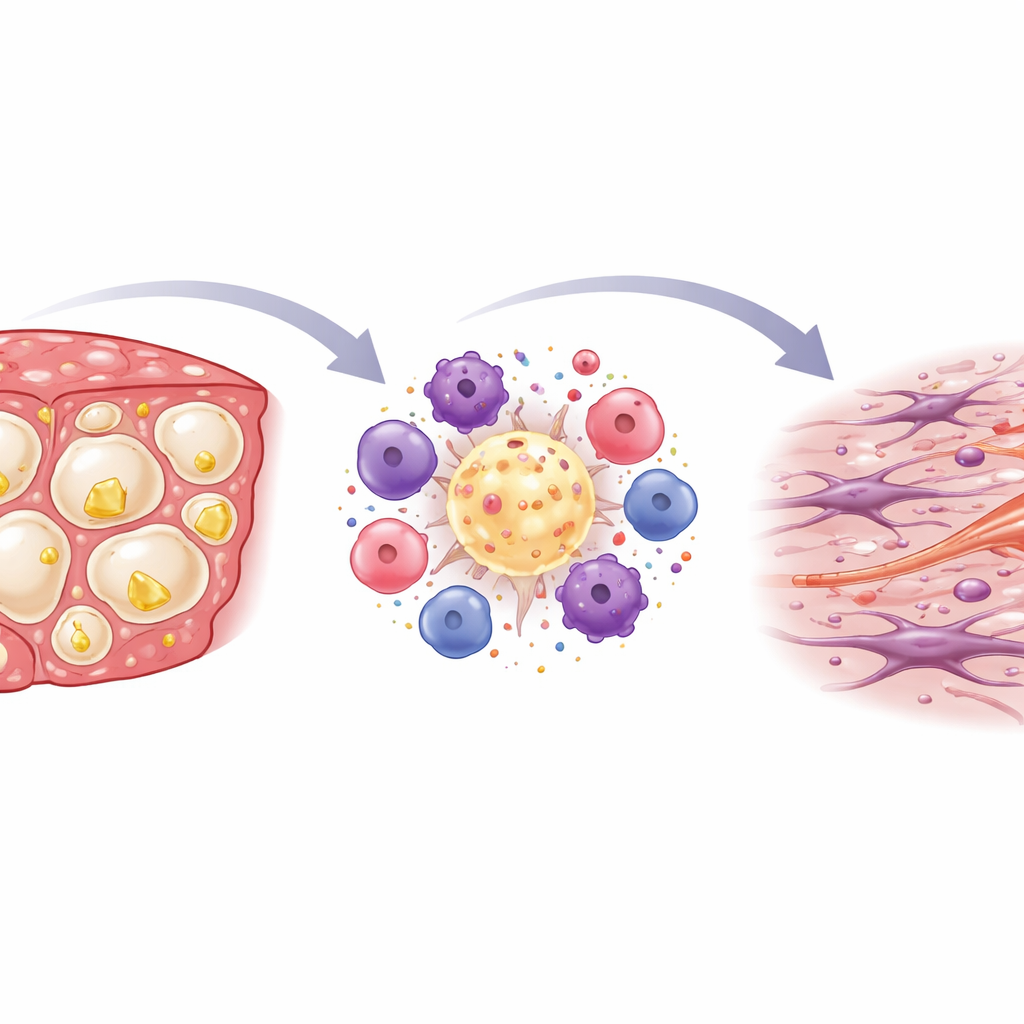
Inflammation and scarring inside the liver
When the researchers stained and examined liver slices under the microscope, they saw that the cholesterol‑rich diet greatly boosted immune activity. Clusters of immune cells formed crown‑like rings around dying, fat‑laden liver cells, a pattern previously linked to aggressive human MASH. Chemical readouts from liver tissue confirmed this picture: genes marking immune cells and classic inflammatory messengers were switched on most strongly in the high‑cholesterol STAM mice. Over time, this inflammation went hand in hand with the buildup of scar tissue. Specialized stains revealed increasing collagen fibers, especially around these immune clusters, and genes associated with fibrosis were markedly elevated, indicating that scar‑producing cells had been activated.
What cholesterol is doing at the molecular level
To understand the process more deeply, the team analyzed which genes in the liver changed their activity across the different diets. In STAM mice, the cholesterol‑rich diet shifted the balance of pathways controlling how the liver handles fats and cholesterol, in ways that likely favor the buildup of a toxic, unbuffered cholesterol pool inside cells. At the same time, networks of genes tied to immune responses, inflammatory signaling, and remodeling of the tissue framework were strongly activated, particularly as the animals aged from 8 to 12 weeks. Together, these patterns match a story in which extra cholesterol worsens stress on liver cells, summons immune cells, and then drives the transformation of support cells into fiber‑producing cells that lay down permanent scar tissue.
How this model can speed better treatments
Although no mouse can fully mirror human fatty liver disease—which often arises in obesity with high insulin levels—the refined STAM model captures a critical piece of the puzzle: cholesterol‑driven worsening of liver injury in the setting of advanced diabetes. By reliably producing strong inflammation and fibrosis over a defined time course, this model provides a more realistic testing ground for experimental drugs aimed at halting or reversing scarring. In plain terms, the study shows that, at least in this setting, dietary cholesterol is not an innocent bystander; it helps push a fatty liver toward lasting damage. That insight, and the improved model built around it, should help researchers more quickly identify which future therapies are most likely to protect human livers from the long‑term consequences of metabolic disease.
Citation: Jonas, W., Gottmann, P., Jähnert, M. et al. Cholesterol-rich diet exacerbates steatohepatitis in the STAM mouse model. Sci Rep 16, 11231 (2026). https://doi.org/10.1038/s41598-026-45979-z
Keywords: fatty liver disease, dietary cholesterol, liver fibrosis, type 2 diabetes, mouse disease models