Clear Sky Science · en
Histidine alleviates Hashimoto’s thyroiditis via the neutrophil extracellular traps-NF-κB signaling pathway
Why this research matters for everyday health
Hashimoto’s thyroiditis is a common cause of an underactive thyroid, especially in women, and can affect energy, mood, weight, and fertility. This study looks at whether a natural building block of protein, histidine, might help calm the immune attack on the thyroid by dialing down specific harmful immune processes.
A closer look at thyroid autoimmunity
In Hashimoto’s thyroiditis, the immune system mistakenly targets the thyroid gland, leading to antibodies against thyroid proteins and gradual destruction of thyroid tissue. People with this condition may develop hypothyroidism and face complications such as fatigue, feeling cold, and difficulty getting pregnant. Despite how common it is, doctors still lack tools to predict who will worsen and have few options beyond replacing missing thyroid hormone. The authors reasoned that small molecules circulating in the blood might reveal new clues about what drives the disease and how to protect the thyroid.
Finding a key missing nutrient signal
The researchers compared blood samples from 20 people with Hashimoto’s thyroiditis and 20 healthy volunteers using metabolomics, a technique that measures many small molecules at once. Among 192 measured molecules, they found 48 that differed between the two groups, with the amino acid histidine standing out as the most strongly reduced in patients. Because amino acids are basic components of diet and body proteins, a drop in histidine suggested it might be consumed or diverted during disease, hinting at a possible protective role that is lost in Hashimoto’s thyroiditis.
How immune nets can harm the thyroid
The team then turned to immune cells called neutrophils, which can release web like structures known as neutrophil extracellular traps, or NETs. These sticky nets help catch microbes but can also damage nearby tissues and have been linked to many autoimmune diseases. In lab grown human cells, the researchers used a high level of iodide, a form of iodine already suspected of aggravating thyroid autoimmunity, to trigger NET release. Under this stress, neutrophils produced more reactive oxygen species, more NETs, and higher levels of histamine related molecules, while histidine levels fell. This pattern matched what they had observed in patient blood and suggested that low histidine goes hand in hand with a harmful overproduction of NETs.
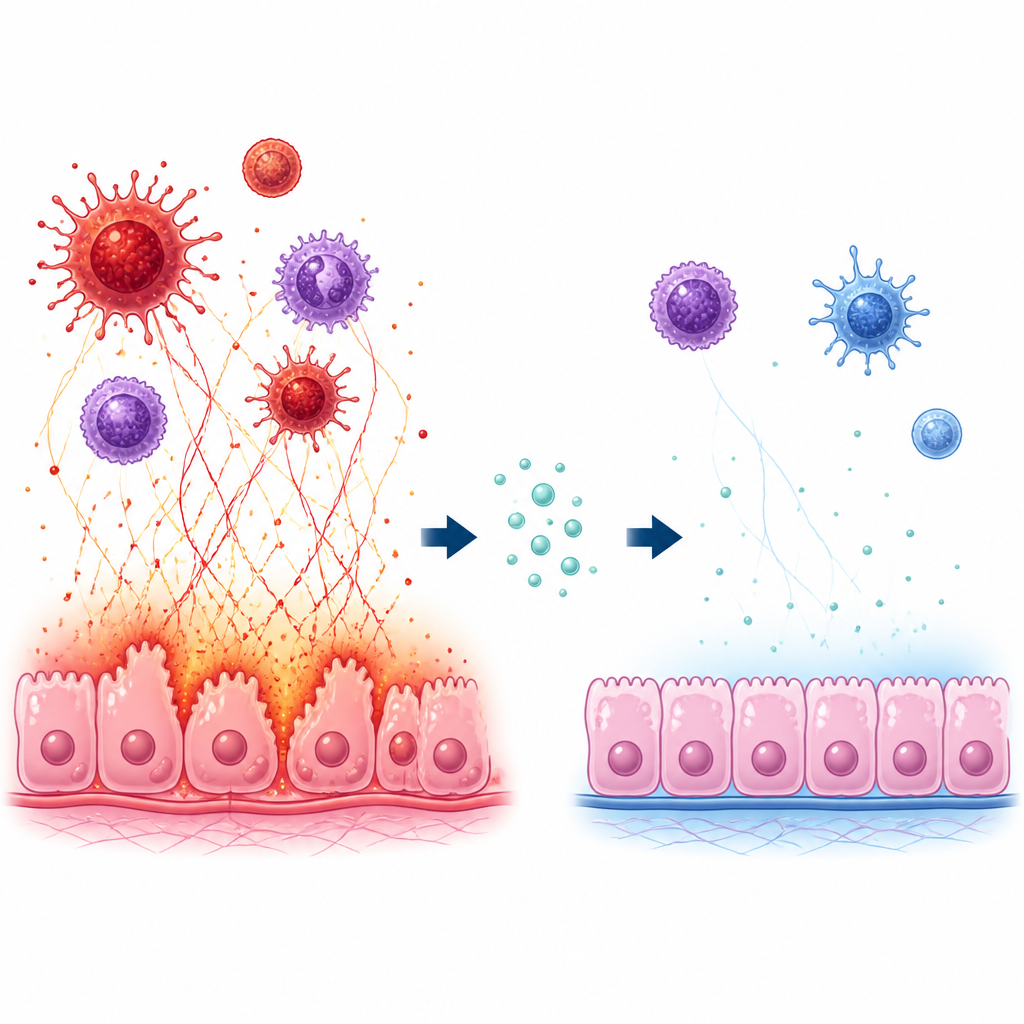
Histidine as a brake on immune damage
Next, the scientists added increasing amounts of histidine to the neutrophils exposed to high iodide. With more histidine present, the cells released fewer NETs and less oxidative stress, and the production of histamine and its related enzymes dropped. To see how this affected the thyroid itself, they co cultured these treated neutrophils with human thyroid follicular cells. Higher histidine protected thyroid cells from dying, boosted their survival, and shifted their chemical signals: pro inflammatory messengers such as TNF alpha and IL 6 went down, while an antioxidant enzyme called SOD went up. At the same time, histidine reduced activity in a major inflammatory control center inside cells, the NF kappa B pathway.
What this could mean for people with thyroid disease
Taken together, the results suggest that lower histidine is linked to more aggressive immune activity against the thyroid, while extra histidine can quiet damaging immune nets and inflammatory signals in cell models. The work does not show that histidine supplements will treat Hashimoto’s thyroiditis in people, but it highlights histidine and the NETs NF kappa B pathway as promising pieces of the puzzle. In the future, carefully designed clinical studies will be needed to test whether adjusting histidine levels, through diet or targeted therapies, can safely help protect the thyroid in patients living with this chronic autoimmune condition.
Citation: Ding, T., Wang, Y., Zhang, L. et al. Histidine alleviates Hashimoto’s thyroiditis via the neutrophil extracellular traps-NF-κB signaling pathway. Sci Rep 16, 15148 (2026). https://doi.org/10.1038/s41598-026-45671-2
Keywords: Hashimoto thyroiditis, histidine, neutrophil extracellular traps, thyroid inflammation, NF kappa B