Clear Sky Science · en
PGA-TMC/PTMC/nHA composite membrane with synergistic barrier and osteogenic functions for enhanced bone defect regeneration
Helping Broken Bones Heal Better
When a piece of bone is missing—after a tooth extraction, an injury, or surgery—the body struggles to rebuild the gap before soft tissue grows in and shuts the space. Dentists and surgeons often place thin barrier sheets over these defects to hold the space open so new bone can form. This paper introduces a newly engineered, dissolvable membrane designed to be stronger, last longer, and actively encourage bone to grow, potentially improving outcomes for patients who need bone rebuilding in the jaw or skull.
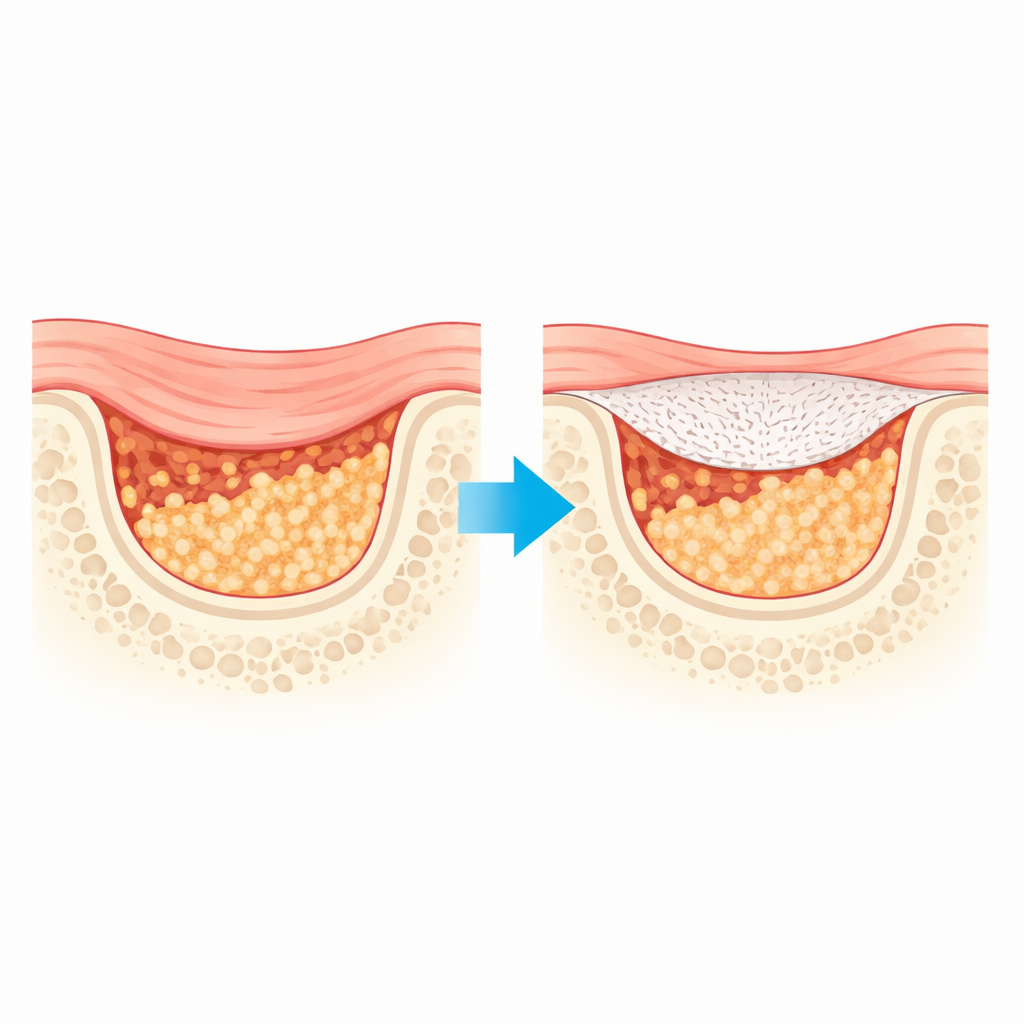
Why Current Bone Covers Fall Short
Standard barrier membranes used in guided bone regeneration act like temporary roofs over a construction site: they keep fast-growing soft tissue from invading while slower-growing bone fills in underneath. Many of today’s clinically used sheets are made from animal tissues. While widely accepted, they can degrade too quickly and may not be strong enough to hold delicate bone grafts in place over the months needed for full healing. Existing synthetic plastic-like materials solve some of these problems but often become weak in an unpredictable way as they break down and do little to actively stimulate new bone growth.
Designing a Smarter Support Scaffold
To tackle these issues, the researchers created a composite membrane called PGTTH using a fine-fiber spinning technique. The material combines three key ingredients: a stiff yet biodegradable backbone that gives the sheet its core strength; a softer, elastic component that breaks down slowly and steadily to preserve the membrane’s shape; and tiny particles of a bone-like mineral (nano-hydroxyapatite) that resemble the natural mineral in human bone. Spun together into a web of nanofibers, the resulting sheet mimics the structure of the body’s own supporting matrix around cells, offering both mechanical support and a hospitable surface for cells to cling to.
Testing Strength, Stability, and Cell Friendliness
The team first checked whether the new material could physically perform as needed. Compared with simpler versions lacking one or more components, the full PGTTH membrane was stronger, more flexible, and better matched to the stiffness of spongy bone, reducing the risk that it would either tear or overly shield the healing area from natural forces. In liquid conditions similar to the body, it swelled modestly but kept its thickness and shape. Over eight weeks in a lab solution, PGTTH and a related control membrane broke down slowly and evenly, while a simpler material collapsed earlier, suggesting it would fail too soon as a barrier. The surface of PGTTH also struck a balance between attracting water and resisting it, which is important for how proteins and cells first land on the material.
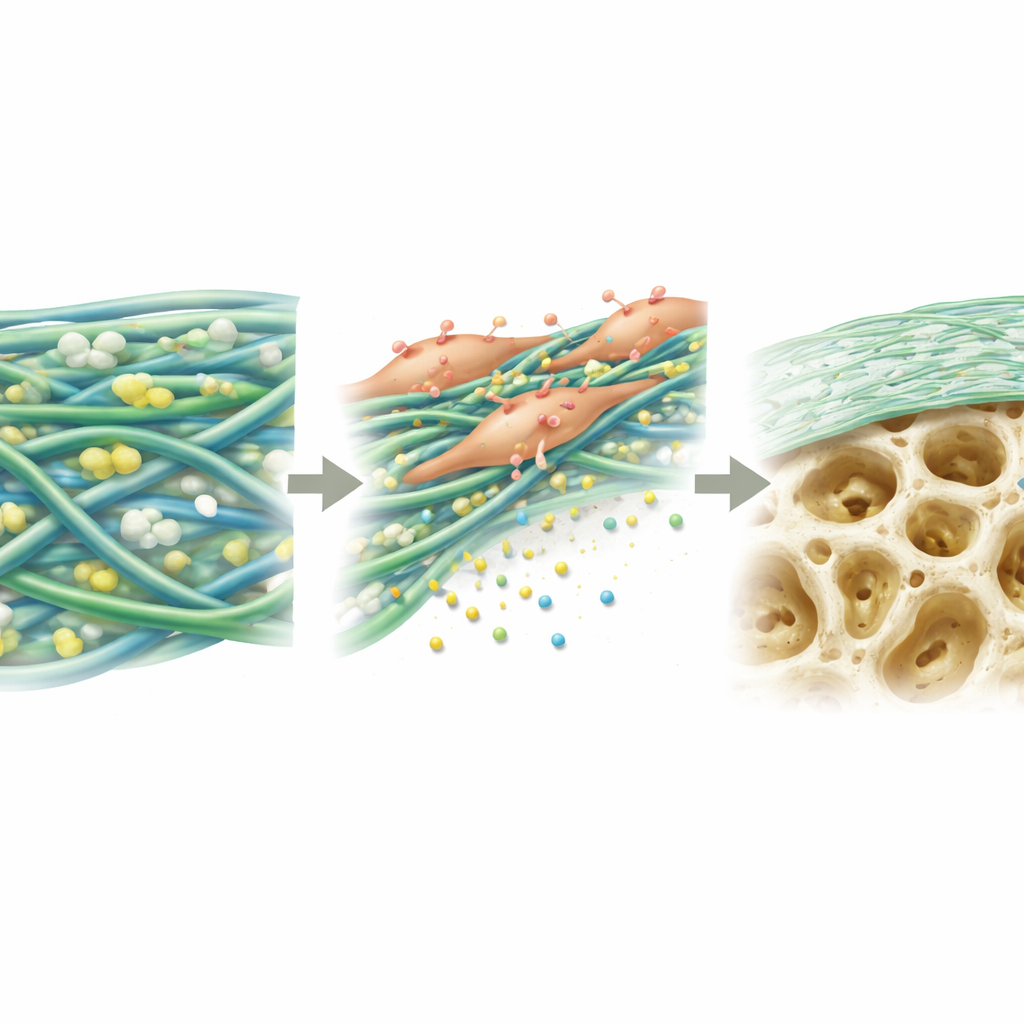
Encouraging Bone-Forming Cells to Do Their Job
Next, the scientists examined how bone marrow stem cells behaved on the different membranes. On PGTTH, cells attached more readily, spread out more fully, and multiplied better over several days than on other test materials. Over three weeks in bone-forming conditions, cells on PGTTH produced more mineral deposits—early building blocks of bone—than cells on the comparison sheets. At the molecular level, the cells ramped up the activity of several key genes and proteins involved in turning stem cells into bone-making cells. The authors attribute this to a combination of the membrane’s surface properties and the slow release of calcium and phosphate ions from the bone-like particles, which together send biochemical signals that nudge cells toward building new bone.
Proving Bone Repair in Living Animals
To see if these advantages translated into real healing, the team created sizable round defects in the skulls of rats—holes that would not naturally fill in on their own. Some defects were left uncovered, some were covered with a simpler membrane, and others with the new PGTTH sheet. After twelve weeks, high-resolution 3D imaging showed that the PGTTH-covered defects contained far more new bone with a denser, more interconnected structure than the other groups. Under the microscope, these areas looked strikingly like mature, well-organized bone, with layered structure and uniform mineral content, rather than the patchy, fibrous, or incomplete repair seen with the older material or no membrane at all.
What This Could Mean for Patients
Overall, the study shows that a carefully tuned blend of strong yet gentle plastics and bone-like mineral can do more than simply block soft tissue: it can actively guide and speed up the body’s own bone-building process. While the work has so far been done in rats and more long-term and mechanistic studies are needed, the PGTTH membrane appears to offer both the stable “roof” and the biological “nudge” that complex bone healing requires. If future research confirms its safety and effectiveness in people, this kind of material could improve treatments for jawbone repair, dental implants, and other procedures that depend on reliable, high-quality bone regeneration.
Citation: Wang, J., Wang, P., Wang, B. et al. PGA-TMC/PTMC/nHA composite membrane with synergistic barrier and osteogenic functions for enhanced bone defect regeneration. Sci Rep 16, 10815 (2026). https://doi.org/10.1038/s41598-026-45665-0
Keywords: guided bone regeneration, bioactive membranes, bone tissue engineering, nano-hydroxyapatite, electrospun scaffolds