Clear Sky Science · en
Understanding the underlying language code that governs the π–π non-covalent interactions between proteins and DNA
How Proteins Read DNA Without Touching It Permanently
Every second, countless proteins in our cells have to find and read specific words in the DNA "instruction book". They do this without permanently binding or cutting the DNA, using fleeting attractions between molecules. This paper explores one of the most important of these subtle forces: a special kind of stacking interaction between flat, ring-shaped parts of proteins and DNA bases. The authors argue that these contacts behave like a simple, repeatable code that helps proteins recognize DNA sequences, and they explain that code using ideas from quantum physics.
The Quiet Magnetism of Flat Rings
Both DNA bases and several amino acids in proteins contain flat, ring-shaped structures whose electrons are spread out over the whole ring. When two such rings line up face-to-face and slightly offset, they can attract each other through what chemists call π–π (pi–pi) interactions. Earlier work showed these contacts can be nearly as strong as hydrogen bonds and are common at protein–DNA interfaces. In this study, the authors focus on the benzene-like rings found in aromatic amino acids such as phenylalanine, tyrosine, and tryptophan, and how they stack against the rings of the four DNA bases. They argue that these stacks do more than simply glue molecules together: they form a structured, directional interaction that can carry information about which base is present.
A Consistent Grip on Many Different DNA Letters
By examining the geometry of these stacks, the authors find a recurring pattern. For the larger DNA bases (adenine and guanine, called purines), the most stable configuration puts the protein’s benzene ring parallel to the base so that two specific positions on the base (called N3 and C2) lie under two specific carbons (C1 and C2) of benzene. For the smaller bases (thymine and cytosine, called pyrimidines), the same benzene atoms instead align over two carbons called C5 and C6 in the base. In every case, the rings adopt a parallel but horizontally shifted arrangement, similar to two overlapping coins slightly slid past each other. This repeated alignment suggests a kind of structural "alphabet": the benzene ring stays the same, while each base presents its own pattern of electron-rich spots to that fixed frame.
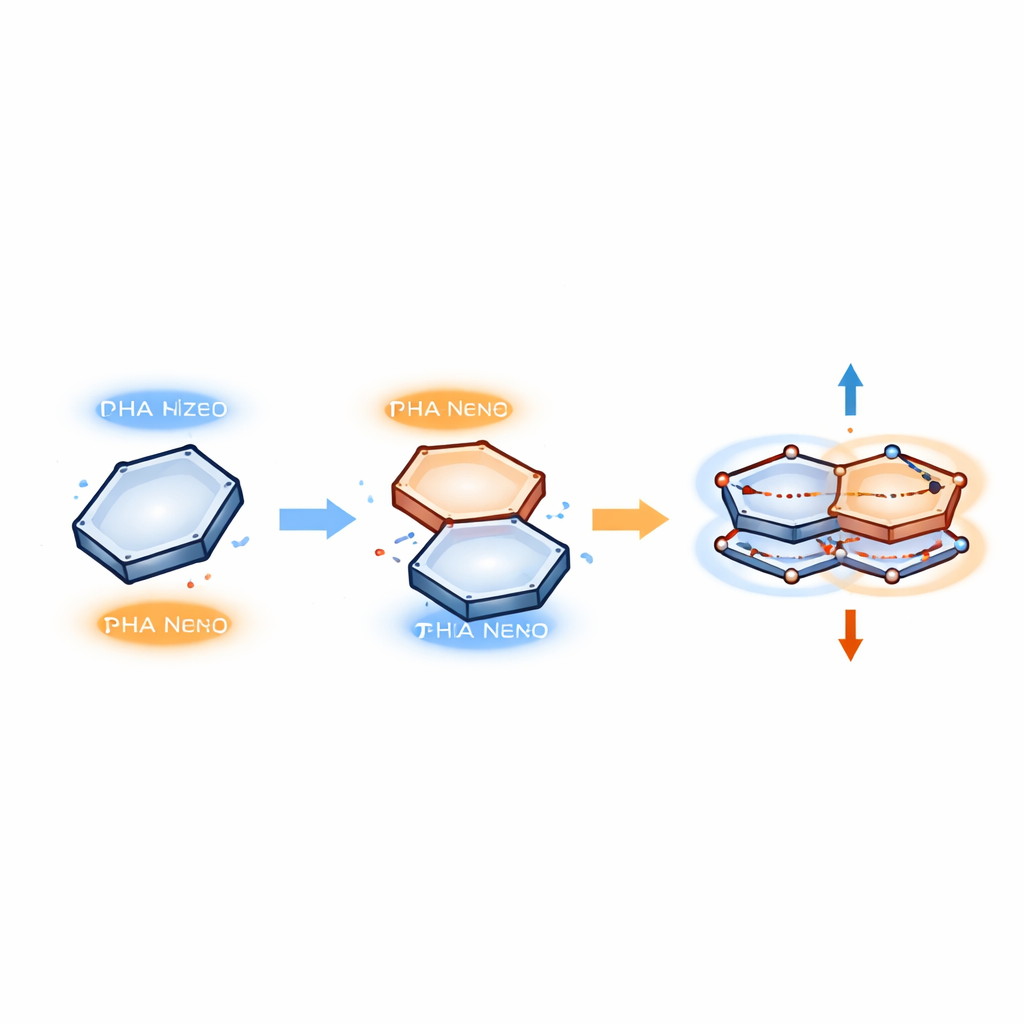
Electron Pairs that Oscillate Like Tiny Springs
To describe what happens inside these stacks, the authors use a model where electrons move in correlated pairs that oscillate between the two rings. Instead of forming a new chemical bond, one electron temporarily hops from an occupied region in one ring into an empty region (a "hole") in the other, and then back again. These paired motions are treated as resonant quantum states, somewhat like two masses connected by a spring that vibrate in step. In the model, only two key ingredients matter: the repulsion between electrons and their coupling to the molecule’s vibrations. When the benzene ring is displaced just right over a particular base, these ingredients combine to create a stable pattern of paired motion that links the two rings without breaking their original structures.
From Quantum Motion to a Measurable Force
Because these electron pairs move in a regular, oscillatory way, the authors can estimate the force holding the rings together using a tool from quantum mechanics known as the Hellmann–Feynman theorem. This theorem connects changes in energy to forces between particles, much as stretching a spring stores energy and creates a restoring pull. In the π–π stacks, the energy of the correlated electron pairs depends on how far the rings are from each other and how far the electrons "swing" during their oscillation. Differentiating this energy with respect to distance yields an effective force that keeps the rings at a preferred offset and separation—strong enough to stabilize protein–DNA complexes, but weak enough to be reversible when the complex needs to fall apart.
A Simple Code for Flexible DNA Recognition
Put together, the work suggests that proteins exploit a robust, reusable design: a constant benzene ring in the aromatic amino acid and a variable DNA base that offers different electron landscapes. The benzene ring provides the stable framework needed to form these oscillating electron pairs, while the base determines exactly where and how strongly they form. This gives proteins a non-permanent yet specific way to "feel" which base they are contacting, helping them recognize target sequences amid the vast genome. In everyday terms, π–π interactions let proteins press a standard-shaped reader head against different DNA "letters" and sense their identity through a finely tuned, quantum-scale touch.
Citation: Riera Aroche, R., Ortiz García, Y.M., Riera Leal, L. et al. Understanding the underlying language code that governs the π–π non-covalent interactions between proteins and DNA. Sci Rep 16, 14361 (2026). https://doi.org/10.1038/s41598-026-44532-2
Keywords: protein–DNA interactions, aromatic amino acids, pi–pi stacking, DNA recognition, quantum biology