Clear Sky Science · en
Adapalene, an RAR agonist, exerts anti-inflammatory effects by regulating macrophage polarization through RAR $$\upbeta$$ -mediated signaling pathways
Why a skin medicine matters for whole-body health
Adapalene is best known as a topical acne gel, but this study suggests it may quietly have much broader powers. The researchers show that, when given by mouth in mice, adapalene can calm overactive immune cells, protect the liver from severe infection and fatty diet damage, and even improve survival in a sepsis model. For readers interested in how an everyday dermatology drug might be repurposed to fight life‑threatening inflammation and metabolic disease, this work opens an intriguing window.
Front-line immune cells that can harm or heal
Our bodies rely on macrophages—roving “big eaters” of the immune system—to detect germs, clear debris, and coordinate inflammation. These cells can adopt two broad modes. In an M1, or pro‑inflammatory mode, they pump out aggressive molecules that help kill microbes but can also damage our own tissues if left unchecked. In an M2, or anti‑inflammatory and repair mode, they help resolve inflammation and support healing. In obesity, sepsis, and fatty liver disease, macrophages become skewed toward the M1 state, fueling chronic low‑grade inflammation that contributes to insulin resistance and tissue scarring. Finding ways to nudge macrophages back toward a balanced or M2‑leaning state is therefore a major therapeutic goal.
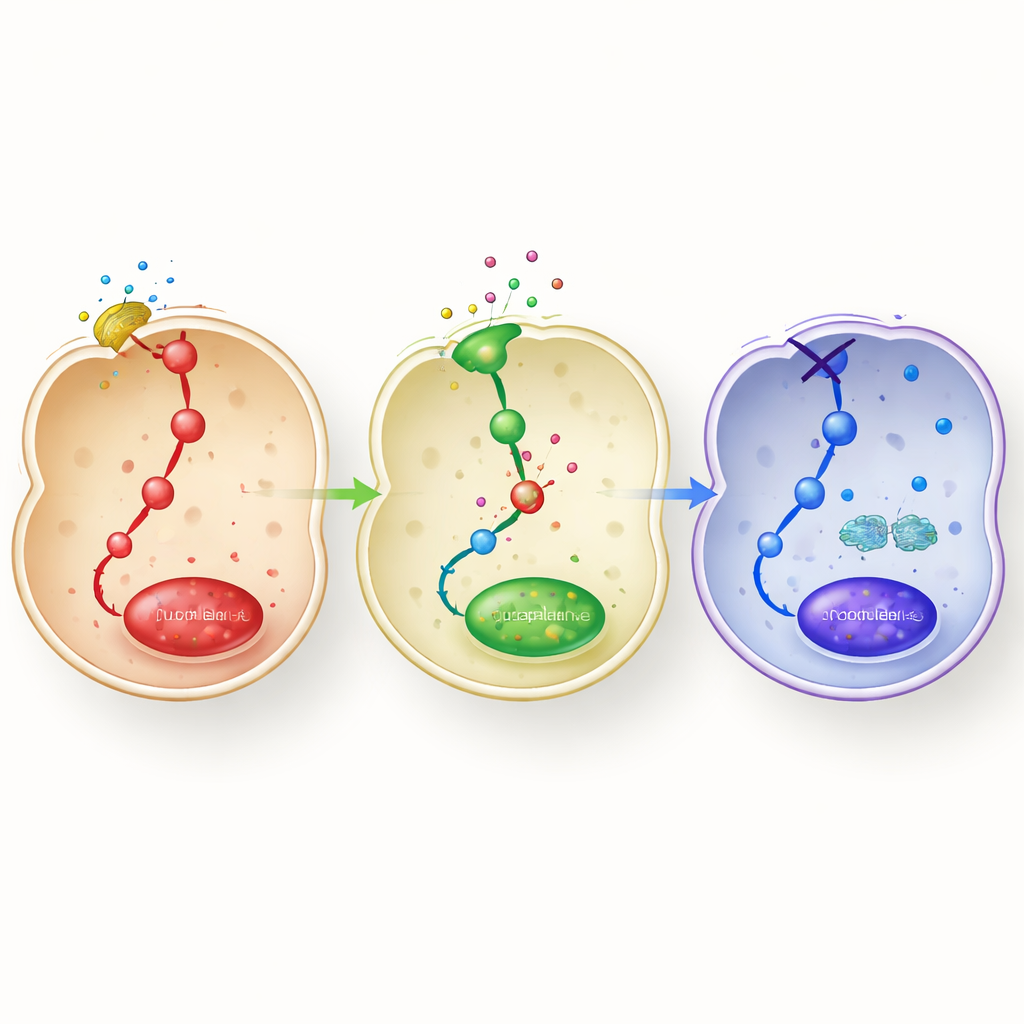
A vitamin A–related switch inside immune cells
Vitamin A and its relatives, called retinoids, control many genes by binding to retinoic acid receptors (RARs) inside cells. Adapalene is a synthetic retinoid that strongly targets one of these receptors, RARβ. The authors asked whether activating RARβ with adapalene could re‑tune macrophages exposed to lipopolysaccharide (LPS), a bacterial component that triggers a powerful inflammatory response. In mouse macrophage cells, LPS sharply increased classical inflammatory messengers and enzymes. Adding tiny amounts of adapalene dialed these signals down in a concentration‑dependent fashion, while at the same time boosting markers associated with the gentler, tissue‑repairing M2 state. Flow‑cytometry analyses confirmed that the population of cells shifted away from an M1‑like surface pattern and toward an M2‑like one.
Shutting off inflammatory circuits and turning on repair
Digging deeper, the team mapped how adapalene rewires the molecular circuitry inside macrophages. LPS normally flips on several kinase pathways (MAPK and PI3K/Akt) and the master inflammatory switch NF‑κB, which then moves into the nucleus to activate inflammatory genes. Adapalene substantially reduced the activation of these pathways and limited NF‑κB entry into the nucleus. At the same time, it increased phosphorylation of STAT3, a signaling protein known to encourage M2‑type behavior. When the researchers reduced RARβ levels with genetic tools, or blocked it with a selective antagonist, adapalene largely lost these effects: inflammatory markers rose again, the calming M2 markers fell, and the signaling pathways re‑activated. This pinpointed RARβ as the key internal switch through which adapalene exerts its dual action—damping inflammatory circuits while promoting repair programs.
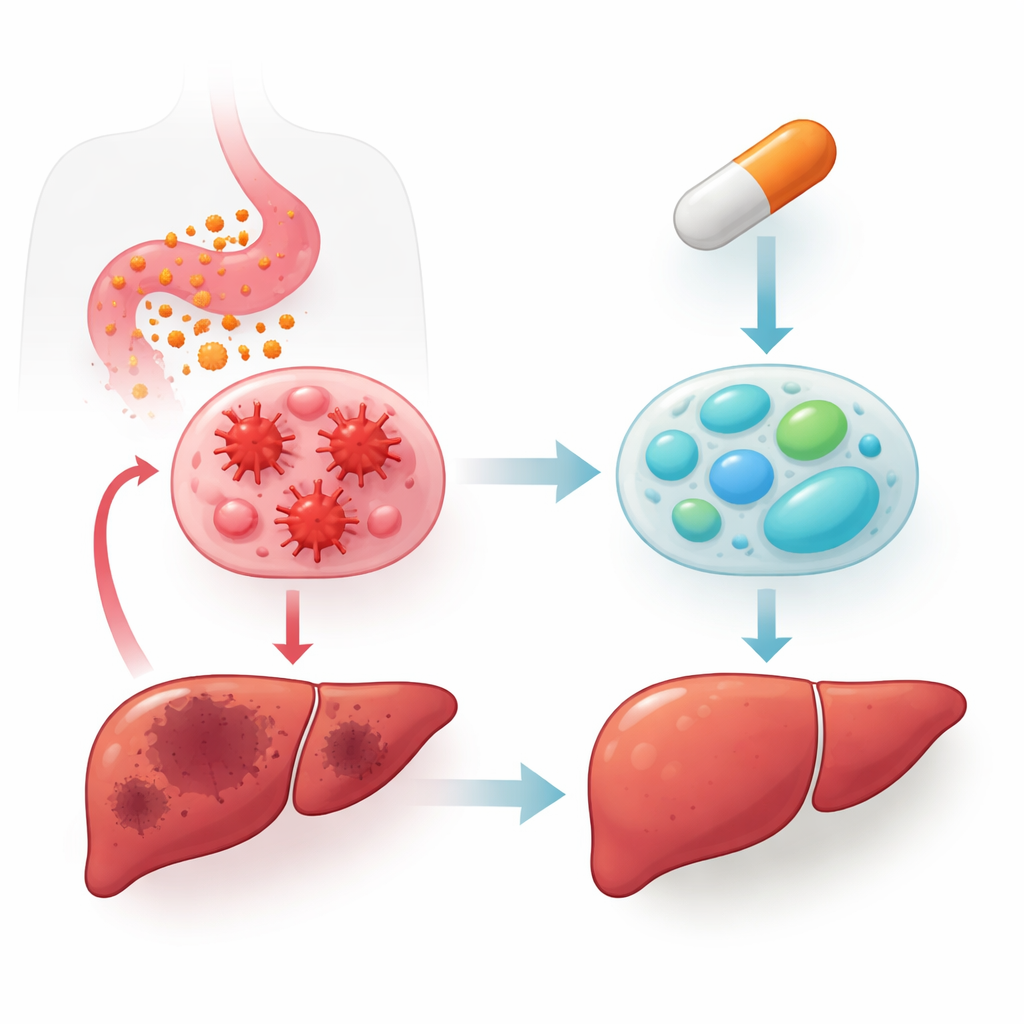
From dishes to diseased livers and whole animals
To test whether these cell‑culture findings matter in living organisms, the authors turned to mouse models of both acute and chronic inflammation. In a severe LPS‑induced sepsis model, oral adapalene lowered inflammatory cytokines in the blood and liver, reduced liver injury markers, improved liver tissue structure under the microscope, and dramatically improved survival from near‑certain death to high survival rates. In mice made obese with a high‑fat diet, several weeks of oral adapalene cut inflammatory markers in the liver, increased M2‑associated markers, reduced activation of the same kinase pathways, and lessened fat and collagen buildup linked to fatty liver disease and fibrosis. Across these models, liver levels of RARβ were selectively increased, reinforcing the central role of this receptor.
What this could mean for future treatments
Put simply, the study suggests that adapalene can act as an internal rheostat for macrophages: it turns down destructive inflammatory responses while turning up healing responses, largely through activating RARβ and quieting downstream signaling cascades. In mice, this translates into protection from lethal sepsis, reduced liver inflammation, and less scarring in obesity‑related fatty liver. Although adapalene is currently approved only as a skin treatment, these findings raise the possibility that carefully dosed systemic versions, if proven safe in humans, might one day help treat inflammatory and metabolic disorders by re‑educating the body’s own immune cells.
Citation: Lee, N.H., Choi, M.J., Ji, S.M. et al. Adapalene, an RAR agonist, exerts anti-inflammatory effects by regulating macrophage polarization through RAR\(\upbeta\)-mediated signaling pathways. Sci Rep 16, 11385 (2026). https://doi.org/10.1038/s41598-026-44454-z
Keywords: macrophage polarization, adapalene, inflammation, sepsis, fatty liver disease