Clear Sky Science · en
SIRT1 induces FOXO1/autophagy/NCOA4-induced ferroptosis and accelerates the progression of cerebral infarction by inhibiting the E2F1/NOTCH-1/YAP signaling pathway
Why this stroke study matters
Cerebral infarction, a common form of stroke, remains a leading cause of death and disability worldwide. Doctors can sometimes reopen blocked vessels, but many patients are still left with lasting brain damage. This study looks inside brain cells to uncover how a key control protein, SIRT1, may actually worsen injury after a stroke by pushing cells toward a destructive form of iron‑driven death. Understanding this hidden pathway could open the door to new treatments that protect the brain when every minute counts.
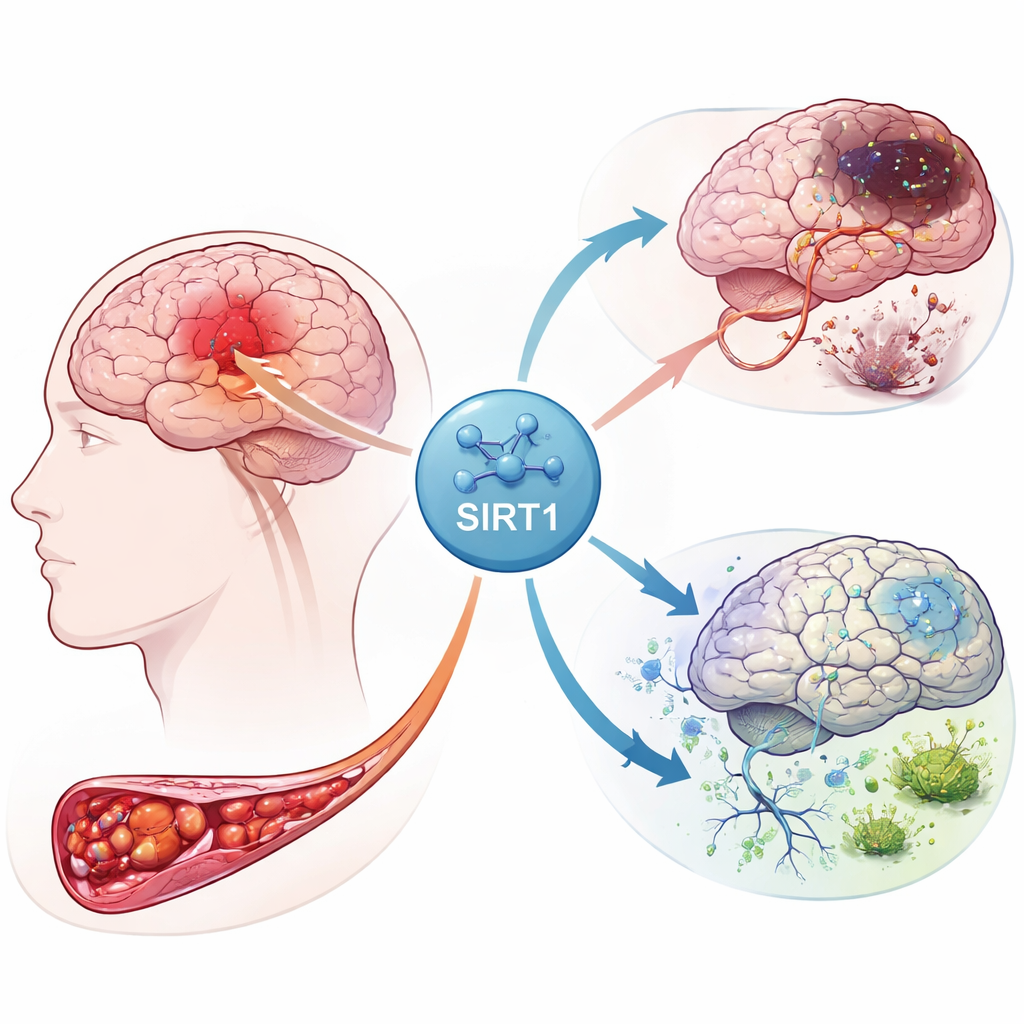
The chain reaction inside a damaged brain
When blood flow to part of the brain is cut off, nerve cells are starved of oxygen and energy. This crisis sets off a storm of signals inside many cell types, including supporting cells called astrocytes. The authors focused on a protein named SIRT1, a master regulator that responds to energy stress. Using gene‑expression data from stroke patients, they found that SIRT1 was closely linked to genes involved in iron handling, cell survival, and a newly recognized form of cell death called ferroptosis. Their analysis also highlighted connections between SIRT1 and several signaling routes already known to control cell growth and self‑cleanup processes.
From big data to animal brains and cell dishes
To see how these clues play out in living tissue, the team created a rat model of cerebral infarction by briefly blocking a major brain artery and then restoring blood flow. Some rats received a drug that activates SIRT1, while others were given a virus that boosted another pathway component, Notch‑1. Brain slices showed that SIRT1 activity rose sharply after stroke and that turning SIRT1 up made the area of dead tissue larger and neuron damage more severe. In contrast, raising Notch‑1 levels pulled the damage back. Protein tests confirmed this pattern: when SIRT1 was high, protective factors associated with Notch‑1 and a partner protein, YAP, fell, while the stroke injury expanded.
How iron and self‑eating drive cell death
The researchers then moved to cultured astrocytes exposed to low oxygen, low nutrients, and added iron to mimic stroke conditions. By carefully adding inhibitors or boosters of SIRT1, E2F1, Notch‑1, YAP, and ferroptosis, they mapped a detailed chain of events. High SIRT1 turned down nuclear E2F1, Notch‑1, and YAP, but turned up another regulator called FOXO1, along with proteins that mark increased self‑digestion (autophagy) and a cargo adaptor named NCOA4. This combination favored a special type of autophagy that breaks open iron‑storage complexes, flooding cells with free iron. As this happened, markers of antioxidant defense shifted and cell death soared, matching the biochemical signature of ferroptosis. Blocking ferroptosis or restoring parts of the Notch‑1/YAP axis reduced these damaging changes.
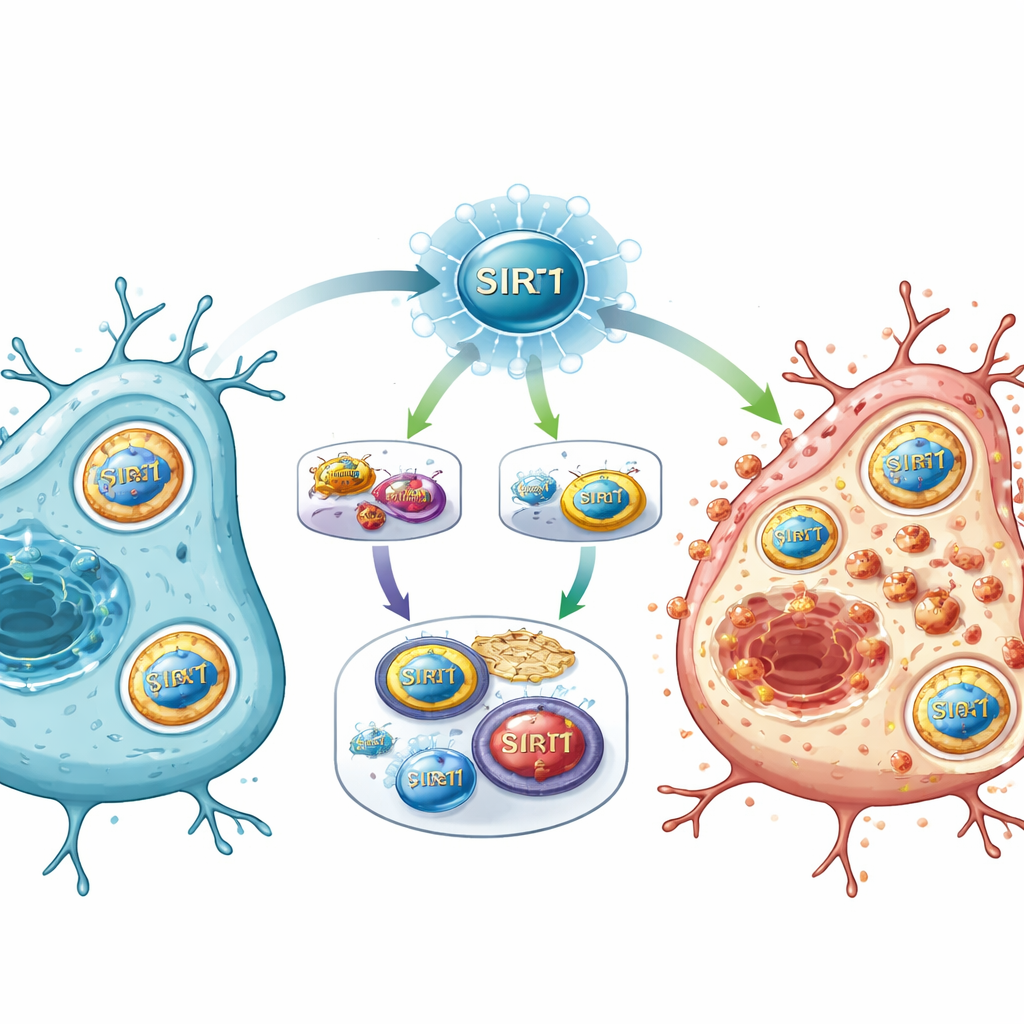
A new master switch for injury pathways
Putting the pieces together, the study proposes that SIRT1 acts as a master switch after stroke, sitting at the crossroads of energy sensing, gene regulation, and iron metabolism. When SIRT1 is strongly activated, it suppresses the E2F1/Notch‑1/YAP route, which normally helps keep certain survival genes and antioxidant systems running. At the same time, SIRT1 boosts FOXO1 and an autophagy program that recruits NCOA4 to dismantle iron‑storage proteins. The resulting iron overload, combined with weakened chemical defenses, drives lipid damage in cell membranes and pushes cells into ferroptosis.
What this means for future stroke care
For a non‑specialist, the key message is that not all cell death after stroke is the same, and some of it may be preventable. This work suggests that, in the stressed brain, SIRT1 can tip the balance toward a destructive iron‑dependent death pathway rather than protection. Targeting SIRT1 itself, or the downstream steps involving Notch‑1, YAP, FOXO1, autophagy, and NCOA4, could offer new ways to limit brain damage beyond simply reopening blocked vessels. While more studies in human tissue are needed, the pathway outlined here provides a roadmap for designing drugs that cool down this iron‑driven fire inside brain cells and potentially improve recovery after cerebral infarction.
Citation: Wang, G., Ma, S., Qi, L. et al. SIRT1 induces FOXO1/autophagy/NCOA4-induced ferroptosis and accelerates the progression of cerebral infarction by inhibiting the E2F1/NOTCH-1/YAP signaling pathway. Sci Rep 16, 13946 (2026). https://doi.org/10.1038/s41598-026-44355-1
Keywords: cerebral infarction, ferroptosis, SIRT1, autophagy, stroke mechanisms