Clear Sky Science · en
Capsular polysaccharides of Acinetobacter baumannii modulate antimicrobial resistance and innate immune response
Why hospital germs wear a protective coat
In modern hospitals, one stubborn germ has become a notorious troublemaker: Acinetobacter baumannii, a bacterium that shrugs off many antibiotics and lingers on medical equipment. This study looks at its slimy outer coat, made of sugar molecules called a capsule, and asks a simple question with big consequences: does this coat just hide the germ, or does it actively help it survive drugs and the body’s first line of defense?
A sugar shield around a dangerous germ
A. baumannii often infects people in intensive care units, causing pneumonia, bloodstream, and urinary infections. Many strains are resistant to multiple antibiotics, so doctors also rely on strong disinfectants, detergents, and even light-based methods to keep it in check. The researchers focused on the capsule, a thick, sugary layer that surrounds many of these bacteria. They compared a typical clinical strain with an almost identical version that had been genetically altered so it could no longer make this capsule. This allowed them to ask how much of the germ’s toughness comes specifically from that outer coat.
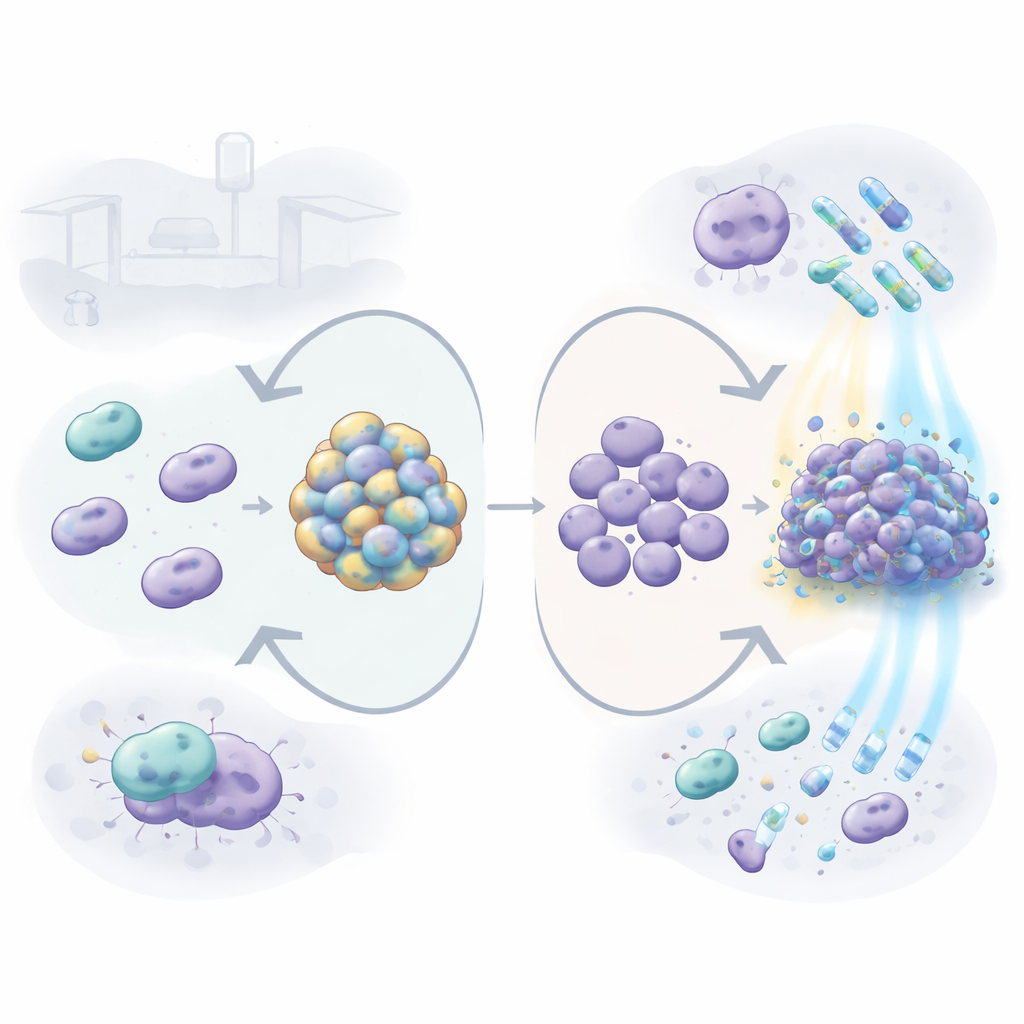
How the coat changes survival on hospital surfaces
When the capsule was removed, free-swimming (planktonic) bacteria became more vulnerable to several drugs and cleaners commonly used in hospitals, including the antibiotics gentamicin, tetracycline, and colistin, and the detergent SDS. In other words, without its coat the germ was easier to kill. But the story changed when the bacteria formed biofilms—sticky layers on plastic surfaces that resemble what they build on catheters or ventilator tubes. The capsule-free mutant responded by making a much thicker biofilm, rich in extracellular material such as DNA. These dense communities turned out to resist some treatments, especially colistin, chlorhexidine, and hydrogen peroxide, better than biofilms made by the normal, coated strain. This suggests that when the outer coat is missing, the bacterium can compensate by building a fortified communal shelter.
Light-based treatment and blood components
The team also tested a promising alternative approach called photodynamic therapy, which uses a harmless dye (chlorophyllin) and blue light to generate toxic oxygen molecules on demand. Here again, the capsule helped the germ: bacteria lacking the coat were 10 to 100 times more sensitive to this light-driven attack, both as free cells and as biofilms. The researchers then moved from surfaces to a more body-like setting, growing the bacteria in blood serum and exposing them to immune cells. In active serum, the capsule-free mutant barely grew and formed weak biofilms, while the coated strain thrived, indicating that the capsule protects against molecules in the blood that normally help clear microbes. When added to macrophages—professional “eater” cells of the immune system—the mutant was swallowed and killed much more readily. At the same time, the gene needed for capsule production became more active under many stress conditions, including exposure to antibiotics, disinfectants, blue light, serum, and macrophages, showing that the bacterium actively boosts its coat when threatened.
Quieting and redirecting the immune response
To understand how the capsule influences inflammation, the authors infected mouse and human cells with either the coated or uncoated strain. Both triggered immune reactions, but the capsule-free mutant generally provoked stronger signals for inflammatory messengers such as IL-6 and IL-1β. In macrophages, the normal coated strain more strongly activated caspase-3, a protein linked to a controlled, relatively quiet form of cell death called apoptosis, while the mutant seemed to tilt responses toward more inflammatory pathways. When the team purified capsule material on its own and added it to immune cells without any bacteria present, it unexpectedly acted as a stimulant, driving the production of several inflammatory molecules and attracting human neutrophils across a membrane in a dose-dependent way. Tiny outer membrane bubbles released by the bacteria (vesicles) were also less inflammatory when they carried capsule on their surface, suggesting that the sugary coat can mask other, more alarming bacterial structures.
What this means for future treatments
Overall, the study shows that the capsule of A. baumannii is more than a passive cloak. It shields single cells from antibiotics, disinfectants, blood components, blue light therapy, and immune attack, while toning down some inflammatory signals and steering immune cells toward quieter forms of response. Yet when the capsule is removed, bacteria may build thicker biofilms that resist certain agents, and bits of shed capsule can themselves draw in immune cells and fan inflammation. For future therapies, this means that drugs or enzymes aimed at stripping away the capsule could help the body clear these infections—but only if they are combined wisely with other treatments that can penetrate the resulting biofilms and take advantage of the newly exposed bacteria.
Citation: Klimkaite, L., Kukanauskaite, G., Naujalis, J. et al. Capsular polysaccharides of Acinetobacter baumannii modulate antimicrobial resistance and innate immune response. Sci Rep 16, 14478 (2026). https://doi.org/10.1038/s41598-026-44001-w
Keywords: Acinetobacter baumannii, bacterial capsule, biofilms, antibiotic resistance, innate immunity