Clear Sky Science · en
Ex vivo and computational investigation of corneal iontophoresis to enhance penetration of high-molecular-weight compounds: a study using albumin as a model molecule
Why Putting Medicine into the Eye Is So Hard
Most people think of eye drops as a simple fix for eye diseases, but in reality very little of the drug in a drop actually reaches the inside of the eye. The clear front window of the eye—the cornea—is built to keep unwanted substances out, which is great for protection but a major obstacle for modern treatments such as protein drugs and other large biological molecules. This study explores a gentle electrical technique, called iontophoresis, designed to push big drug molecules through the cornea more effectively while still keeping the eye safe.
A Gentle Push from Electricity
Iontophoresis works by applying a low, controlled electric current at the surface of the eye to encourage drug molecules to move through the cornea. The researchers used a common blood protein, albumin, as a stand-in for many high–molecular-weight medicines. They placed freshly removed rabbit corneas in special chambers, applied different levels of current for just ten seconds, and measured how much albumin crossed to the other side. At the same time, they built a computer model to predict how much the cornea would heat up during treatment, because excess heat can damage delicate eye tissue.
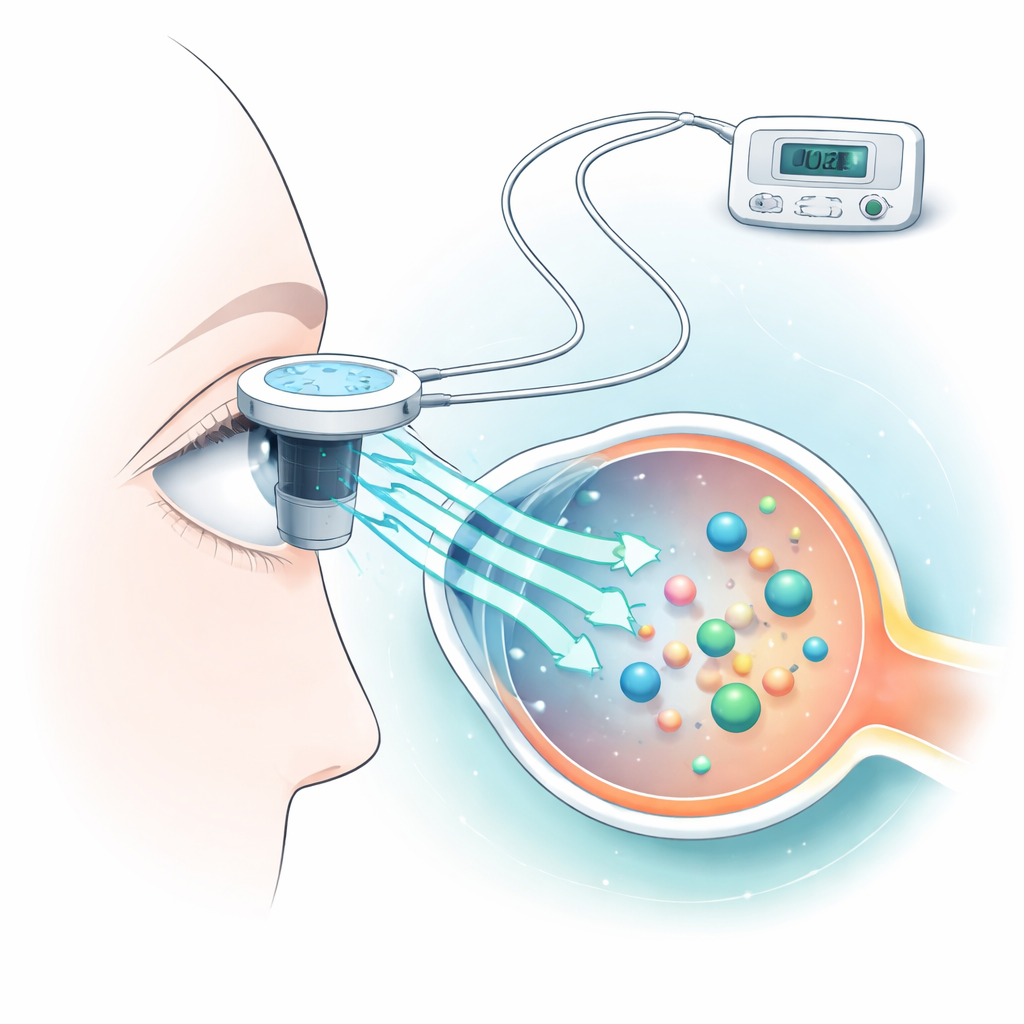
Finding the Sweet Spot Between Delivery and Damage
The team found that iontophoresis clearly boosted albumin transport compared with simple soaking, and that higher currents generally moved more protein across the cornea. Currents in the clinically relevant range, up to about 7 milliamps, increased albumin permeation, with the strongest gains at 6–7 milliamps. Their computer simulations showed that very low currents, up to roughly 2 milliamps, kept the corneal surface temperature within its usual range of about 32–36 °C, while currents of 3–7 milliamps warmed the surface further, sometimes approaching levels where heat stress becomes a concern if applied for too long. An extreme test current of 500 milliamps, used only as a damage model, produced intense heating and clear signs of tissue breakdown.
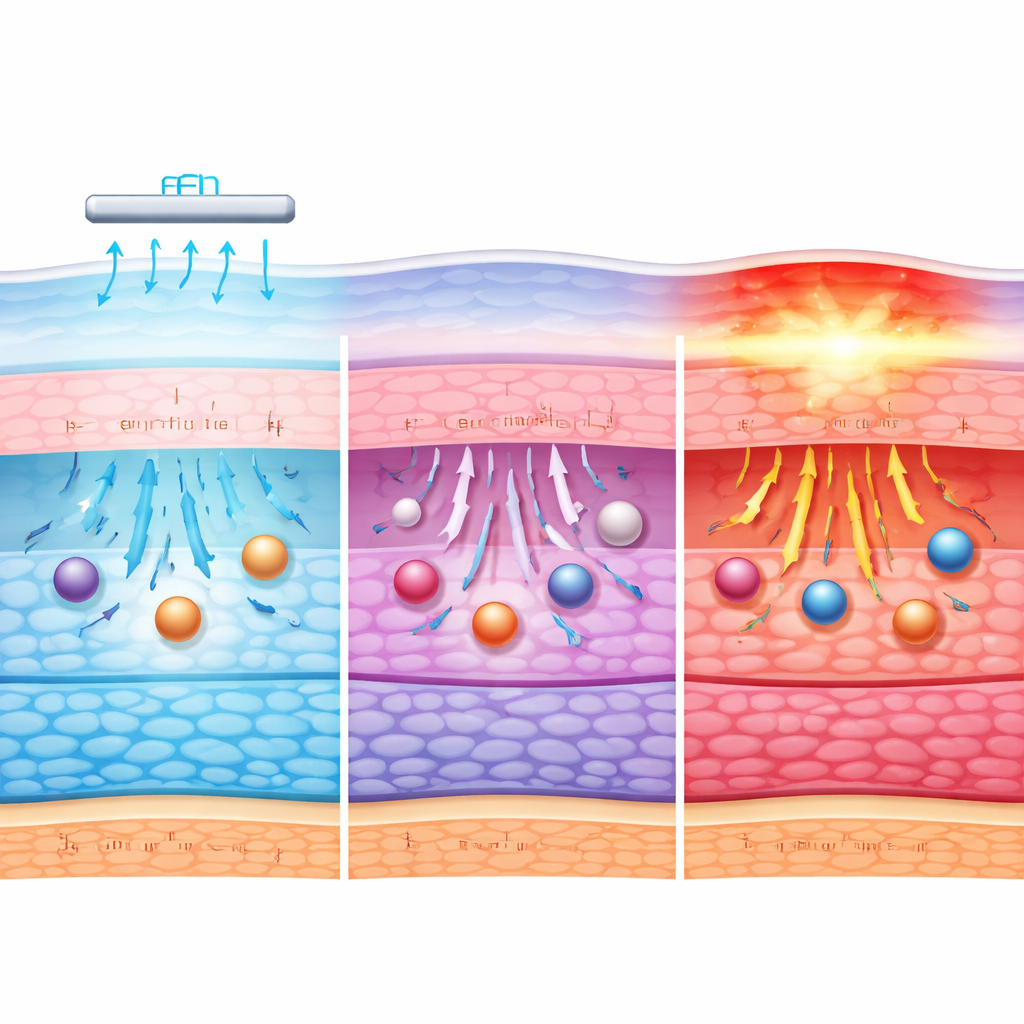
Looking Inside the Cornea’s Molecular Fabric
To move beyond simple temperature and transport measurements, the researchers turned to infrared spectroscopy, a technique that reveals how water, proteins, and fats inside the cornea respond at the molecular scale. At low to moderate currents, the spectral signatures indicated subtle shifts in water binding, protein shape, and lipid (fat) organization, consistent with the tissue adapting to the electrical and thermal load without losing its basic structure. These changes likely loosen pathways enough for large molecules to slip through more easily. At the highest, non-clinical current, however, the infrared patterns shifted dramatically, signaling disrupted proteins, altered water networks, and disturbed membranes—hallmarks of irreversible damage.
How the Process Balances Flow, Heat, and Structure
The study shows that multiple factors work together during corneal iontophoresis. The electric field itself helps drive albumin through the tissue by pushing fluid and charged particles; mild warming further encourages movement without immediately harming cells. At low currents, this combined effect is modest, and the cornea’s barrier largely remains intact. As the current increases into the mid-range, more albumin penetrates, helped by both stronger electrical forces and small, reversible relaxations in the cornea’s molecular packing. Only when the current becomes extreme do heat and structural disruption dominate, destroying the barrier rather than gently opening it.
What This Means for Future Eye Treatments
For a layperson, the key message is that a carefully tuned dose of electricity may one day let doctors deliver big, powerful drug molecules into the eye without needles. This work maps out a practical window of current and time that boosts drug penetration while keeping corneal temperature and molecular structure within safe limits in an ex vivo model. Although further studies in living eyes and with real therapeutic proteins are needed, the findings support iontophoresis as a promising, non-invasive way to treat serious eye diseases that currently require injections, potentially making care both safer and more comfortable for patients.
Citation: Mohamed, A.K., Mahmoud, S.S., Elshibly, S.M. et al. Ex vivo and computational investigation of corneal iontophoresis to enhance penetration of high-molecular-weight compounds: a study using albumin as a model molecule. Sci Rep 16, 10990 (2026). https://doi.org/10.1038/s41598-026-43580-y
Keywords: ocular drug delivery, corneal iontophoresis, albumin transport, thermal safety, biologic therapeutics