Clear Sky Science · en
Single-cell and multi-omics analysis identifies mitophagy-related biomarkers and therapeutic targets in ischemic stroke
Why damaged cell batteries matter in stroke
Stroke is often described as a problem of clogged blood vessels, but what happens inside brain cells after the blood flow stops can be just as important. This study looks at the cell’s “power plants” – mitochondria – and a built‑in cleanup process called mitophagy that removes damaged ones. By tracking how this cleanup system behaves in stroke and which genes control it, the researchers aim to find new ways to predict who is at risk for worse outcomes and to design treatments that protect brain cells when every minute counts.
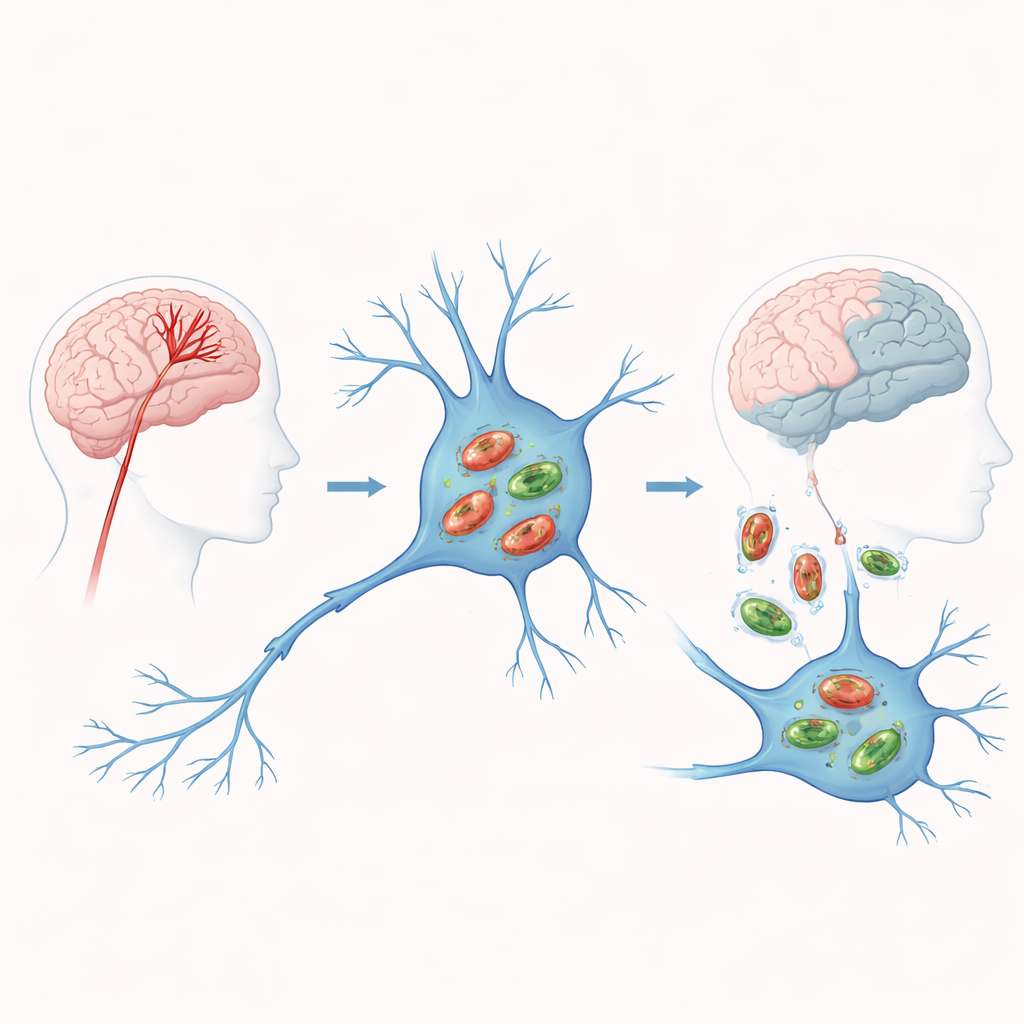
When blood stops, power fails in brain cells
In an ischemic stroke, a blocked artery cuts off oxygen and nutrients to part of the brain. Without fuel, mitochondria falter: they make less energy and spew harmful by‑products that injure cells and push them toward death. Mitophagy is the cell’s emergency response, tagging defective mitochondria and sending them for breakdown and recycling. In theory, timely cleanup prevents toxic buildup and limits damage. But if this process is too weak or too strong, it can either allow malfunctioning mitochondria to linger or strip cells of the energy makers they need to survive. Understanding where stroke patients sit on this knife‑edge is central to the study.
Mining big data to find warning signals in blood
The team combined several large gene‑expression datasets from stroke patients and healthy people, then focused on a panel of genes already linked to mitophagy. Using advanced statistics and multiple machine‑learning models, they searched for patterns that best distinguished stroke from non‑stroke samples. They found nineteen mitophagy‑related genes that changed in stroke and formed two major patient groups with different molecular and immune features. A deeper network analysis highlighted one dense cluster of genes tied to mitochondrial function and protein production, suggesting that energy supply and cellular “factory” activity are tightly intertwined in stroke.
Five key genetic clues and the immune connection
From this network, the researchers trained and compared eight prediction models, ultimately homing in on five standout genes—SRPRB, ATP5J, LSM7, DEGS1, and TGDS—as core markers. A combined five‑gene test accurately separated stroke from control samples in their data. Lab experiments using a cell model of oxygen‑glucose deprivation and blood from stroke patients confirmed that these genes truly shift in real biological systems, not just in computer analysis. Several of them are linked to how mitochondria make energy, how cells handle fats, and how genetic messages are processed. The study also showed that these markers are strongly tied to certain immune cells and pathways, hinting that the way the immune system reacts after stroke is closely coupled to mitochondrial health and blood clotting processes.
Zooming in on brain defenders called microglia
Because blood samples cannot fully capture what happens inside the brain, the team turned to single‑cell data from a mouse stroke model. They examined thousands of individual brain cells and identified distinct groups, focusing on microglia, the resident immune cells of the brain. After stroke, microglia shifted from a calm, watchful state into inflammatory and disease‑associated states. These “activated” microglia showed much higher mitophagy activity, consistent with a strong push to clear damaged mitochondria. One of the five key genes, ATP5J, showed a dynamic rise along the progression from resting to disease‑linked microglia, underscoring its potential role as both a marker and a regulator of how these cells respond to injury.
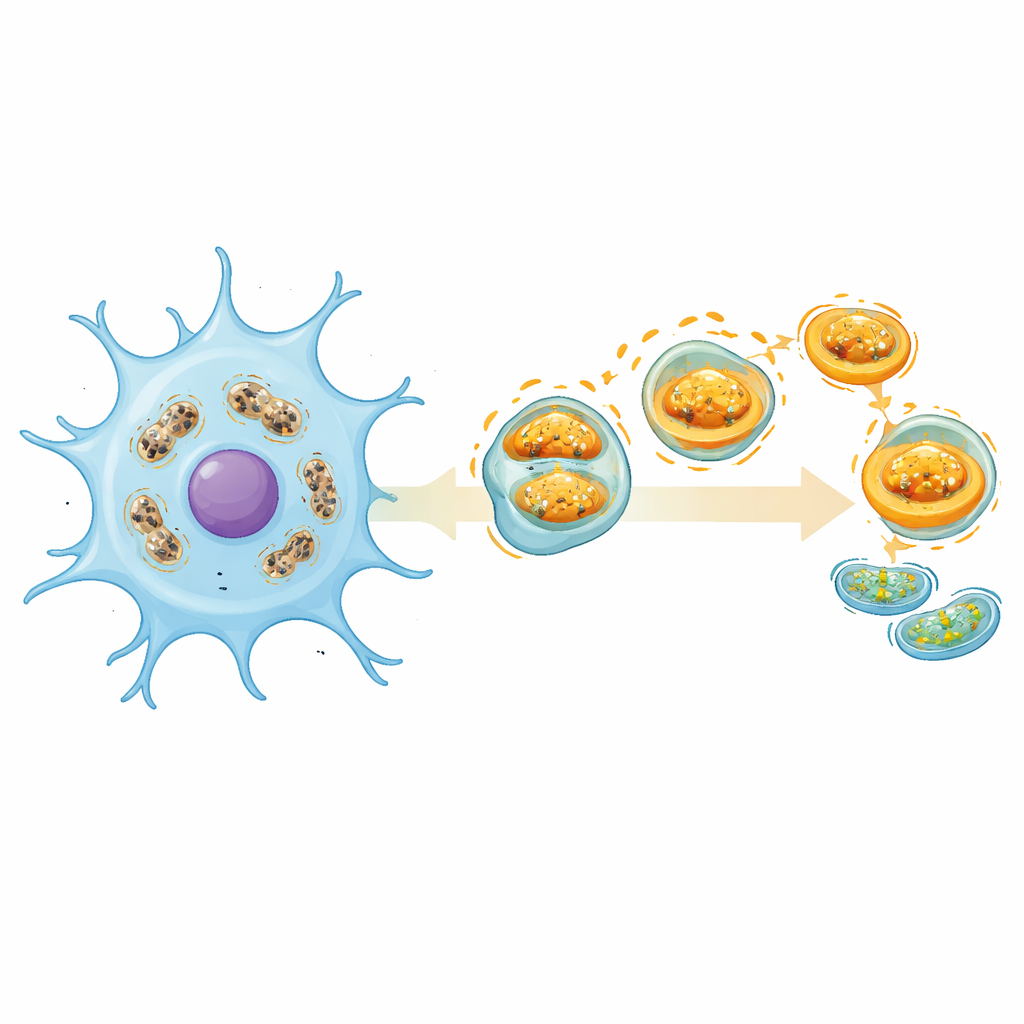
What this could mean for future stroke care
Taken together, the work portrays mitophagy not as simply good or bad, but as a finely tuned response that differs between cell types, disease stages, and body compartments like blood versus brain tissue. The five highlighted genes—especially ATP5J—offer promising starting points for blood‑based tests that could better classify stroke patients and, eventually, guide therapies that nudge mitochondrial cleanup toward a protective “sweet spot.” While more experiments in appropriate brain cell models and larger patient groups are needed, the study suggests that rescuing the brain in stroke may depend as much on repairing its microscopic power systems as on reopening blocked arteries.
Citation: Cao, Z., Wang, Y., Sun, M. et al. Single-cell and multi-omics analysis identifies mitophagy-related biomarkers and therapeutic targets in ischemic stroke. Sci Rep 16, 12433 (2026). https://doi.org/10.1038/s41598-026-43377-z
Keywords: ischemic stroke, mitophagy, mitochondria, microglia, biomarkers