Clear Sky Science · en
Integrated DFT, molecular docking, and molecular dynamics investigation of some novel 2-thiohydantoin analogues as potent CDK2 inhibitors for anticancer therapy
Why this research matters for future cancer treatment
Cancer drugs often work like blunt tools, harming healthy cells while attacking tumors. This paper explores a more precise strategy: designing small molecules that selectively shut down a key engine of cell division, a protein called CDK2. By using advanced computer simulations instead of trial-and-error chemistry, the researchers identify several promising candidate molecules that could one day become safer, more targeted anticancer medicines.
Stopping runaway cell division at its source
Many cancers grow because their cell cycle—the internal clock that controls when cells divide—has gone out of control. CDK2 is one of the main switches that drives cells through this cycle. Earlier drugs that tried to block CDK2 showed promise, but they often also hit closely related proteins that are essential for normal cells, leading to side effects. The authors focus on a little-explored chemical family called 2‑thiohydantoins, ring-shaped molecules that can be tuned electronically and structurally. Their goal is to see whether selected members of this family could latch onto CDK2 more strongly, and more selectively, than the cell’s own fuel molecule, ATP, while avoiding other kinases.
Using computers to “pre-test” drug candidates
Instead of going straight to the lab bench, the team first used several layers of quantum chemistry and molecular modeling to understand how their molecules behave. They calculated how easily each compound can move electrons around its structure, how polar it is, and which parts of the molecule are most likely to form attractive contacts with a protein. Compounds labeled 2b through 2e stood out: they had small energy gaps between their key electronic states and high “electrophilicity,” meaning they should interact readily with biological partners. Maps of the electrical surface of each molecule highlighted strongly negative regions around oxygen and sulfur atoms and positive regions around certain hydrogens—natural hotspots for hydrogen bonds when a drug nestles into a protein pocket.
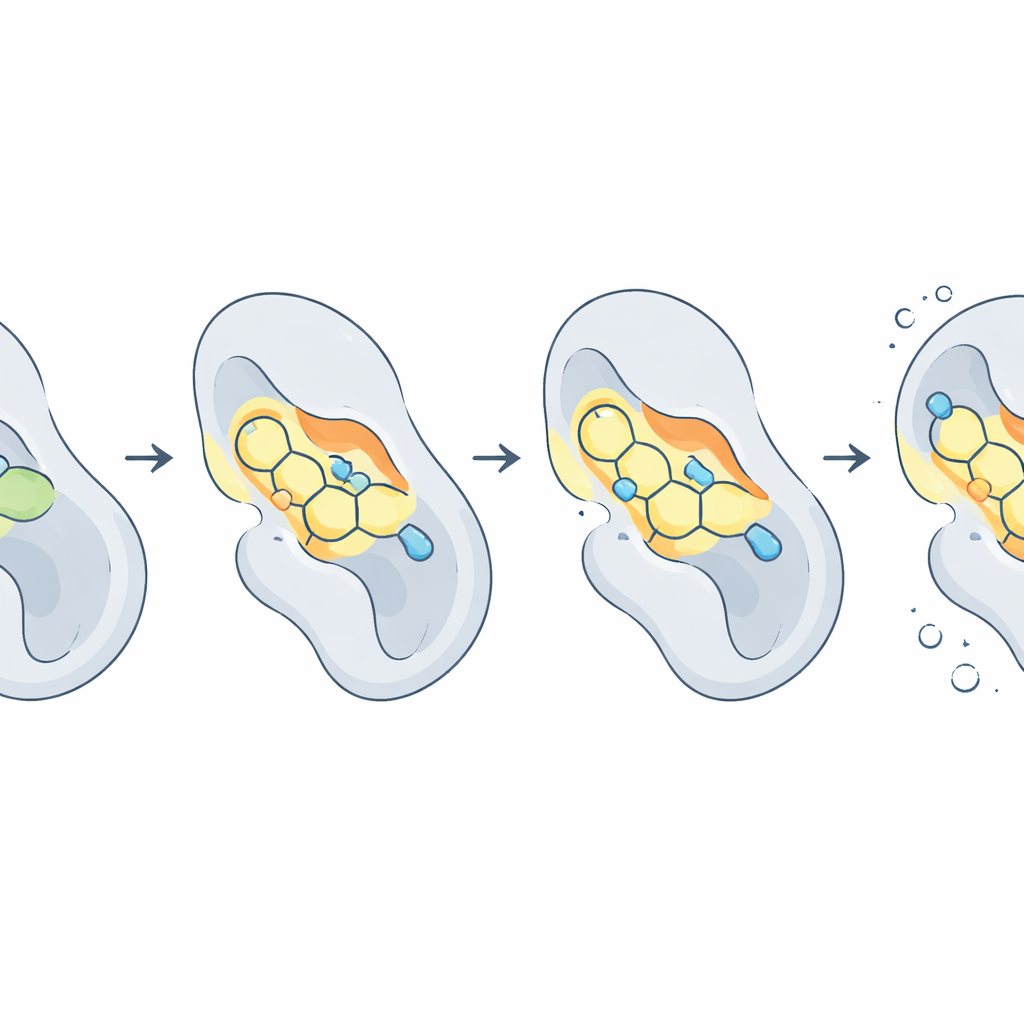
Testing the fit inside the CDK2 pocket
The next step was virtual docking: placing each candidate into the three‑dimensional structure of CDK2 and letting the computer search for the best fit, much like trying keys in a lock. Several molecules, particularly 2b, 2c, and 2d, gripped CDK2 more tightly than ATP itself in these simulations. They formed crucial hydrogen bonds with two residues, Lys33 and Thr14, that sit in the heart of the ATP‑binding pocket, and they also packed snugly against hydrophobic amino acids that line the cavity. In contrast, the parent scaffold without tailored substitutions bound weakly, underscoring how subtle changes in the chemical decoration of the ring can dramatically alter performance.
Watching the protein–drug complex in motion
Because proteins and drugs are not rigid, the authors ran molecular dynamics simulations, effectively slow‑motion movies at the atomic scale, over ten billionths of a second. These showed that the overall shape of CDK2 stayed stable while some drug–protein pairs wobbled or even drifted away. One molecule, 2f, proved exceptionally steady, barely shifting its position within the pocket and maintaining on average nearly three hydrogen bonds to the protein. Others, like 2c and 2d, balanced strong binding with moderate flexibility. To translate these motions into an overall “stickiness,” the team used a technique called MM‑PBSA to estimate binding free energies. Here, compound 2d emerged as the best all‑around performer, with an especially favorable balance between attractive forces inside the pocket and the energetic cost of leaving the surrounding solvent.
What this could mean for patients
Taken together, the simulations highlight four 2‑thiohydantoin compounds—2b, 2c, 2d, and 2f—as especially promising starting points for anticancer drug development. They combine favorable electronic properties, a strong and well‑oriented grip on CDK2, and, in the case of 2d, a particularly advantageous overall binding energy. While these findings are purely computational and must still be confirmed experimentally, they provide a detailed blueprint for chemists: where to place halogen, acid, or ester groups on the core ring to strengthen binding and selectivity. If future lab tests bear out these predictions, the work could accelerate the creation of more precise CDK2‑targeted therapies that slow tumor growth while sparing healthy cells.
Citation: Khaled, N.A., Ahmed, S.A., Ibrahim, M.A. et al. Integrated DFT, molecular docking, and molecular dynamics investigation of some novel 2-thiohydantoin analogues as potent CDK2 inhibitors for anticancer therapy. Sci Rep 16, 10985 (2026). https://doi.org/10.1038/s41598-026-42330-4
Keywords: CDK2 inhibitors, targeted cancer therapy, molecular docking, 2-thiohydantoin, computational drug design