Clear Sky Science · en
Genomic analysis of a clinical Streptococcus suis ST1 isolate from CSF reveals antimicrobial resistance, virulence, and an evolutionary link to ST7
Why a Pig Germ Matters to People
Most of us think of farm infections as a problem for animals, not humans. But some germs can leap from pigs to people and cause life-threatening disease. This study focuses on one such bacterium, Streptococcus suis, which can trigger severe meningitis and lasting hearing loss. By reading the complete genetic blueprint of a strain taken directly from a patient’s spinal fluid, the researchers show how this germ resists key antibiotics, what makes it especially harmful, and how it is evolving alongside related strains that have caused deadly outbreaks.

A Patient, a Spinal Tap, and a Hidden Threat
The story begins with a person admitted to intensive care with purulent meningitis, a serious brain infection. Doctors isolated S. suis from the patient’s cerebrospinal fluid, confirming that a pig-associated germ had invaded the central nervous system. After treatment, the patient’s thinking improved, but they were left with severe hearing loss, a well-known complication of this infection. This real-world case gave scientists a rare opportunity: to examine the DNA of a human-derived strain, named 366, and ask what in its genome might explain its behavior in the body and its response to treatment.
Which Drugs Still Work—and Which Do Not
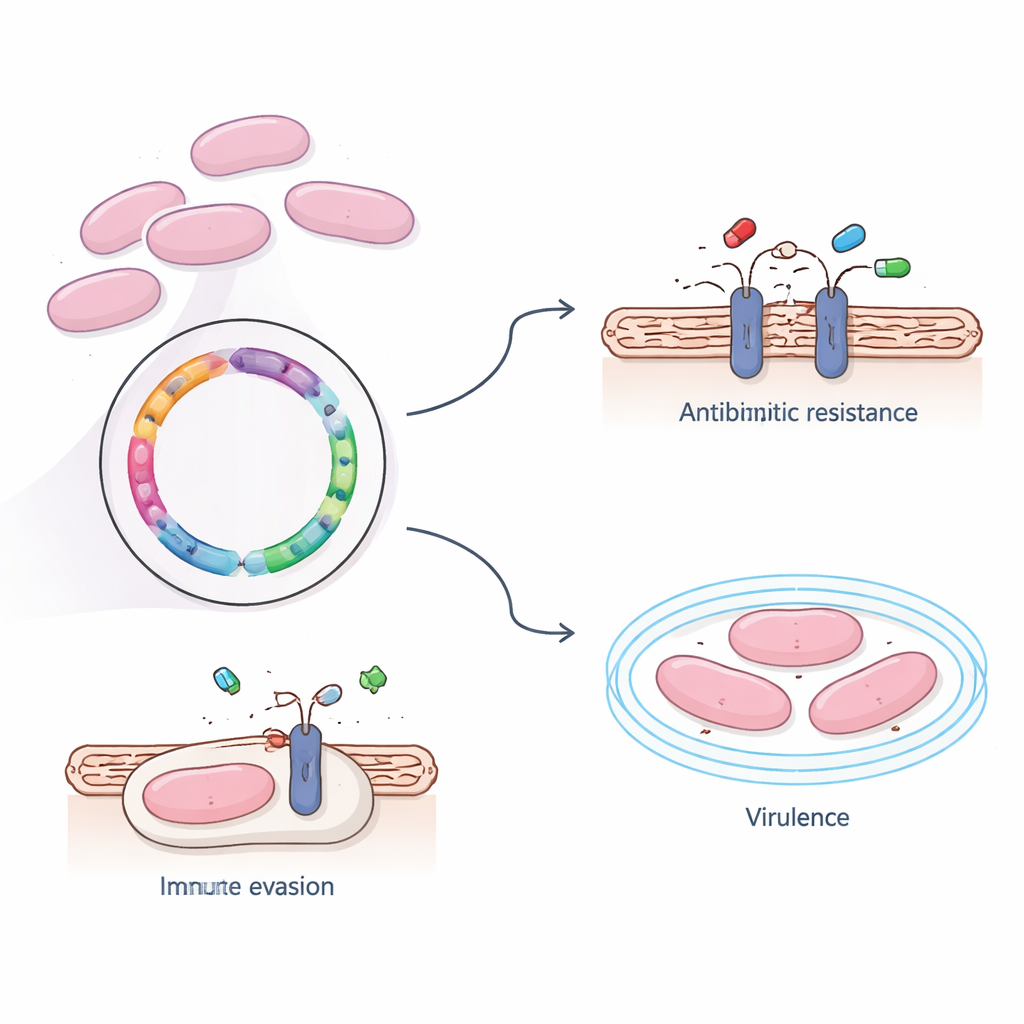
In the lab, the team tested how the bacterium responded to commonly used antibiotics. They found that strain 366 remained vulnerable to several frontline drugs, including penicillin, cephalosporins, and last-resort agents like carbapenems and vancomycin. However, it was strongly resistant to erythromycin, clindamycin, and tetracycline—medications widely used in both human and veterinary medicine. When the researchers scanned the genome, they discovered two resistance genes, ermB and tet(O), which alter the bacterium’s protein-making machinery so these drugs can no longer bind effectively. Interestingly, these resistance genes were embedded in the main chromosome rather than on easily shared mobile DNA elements, suggesting that this strain’s drug resistance spreads more through the expansion of successful bacterial lineages than by rapid gene swapping.
Genetic Clues to How the Germ Causes Disease
Beyond drug resistance, the investigators asked what makes this strain so capable of surviving in the bloodstream and invading the brain. Using several large reference databases, they identified four key genes linked to virulence—the ability to cause disease. Three of them help build a sugary outer capsule that shields the bacterium from being recognized and eaten by immune cells, while the fourth helps the germ latch onto host tissues. Large-scale analyses of all genes showed that many of them support basic life functions such as metabolism and protein production, but also highlighted transport systems that move nutrients, toxins, and possibly drugs across the cell membrane. One major pathway, known as the ABC transporter system, stood out as a central hub for importing useful molecules and exporting harmful ones, potentially including antibiotics.
Family Ties Between Dangerous Strains
To place this patient’s strain in a broader context, the team compared its genome with those of closely related S. suis strains from pigs and people in different regions. They found that strain 366 belonged to sequence type ST1, a genetic lineage already known to dominate human infections. When they built evolutionary trees using thousands of small DNA differences, they showed that ST1 is closely related to another lineage called ST7, which has been linked to large outbreaks. Pangenome analysis—looking at which genes are shared or unique across multiple strains—revealed a large core of common genes between ST1 and ST7, supporting the idea that epidemic ST7 strains evolved directly from an ST1 background by acquiring additional genetic material.
What This Means for Public Health
Put together, the findings paint a picture of a highly adapted pig-borne germ that has crossed into humans with a toolkit for both surviving treatment and causing severe disease. The patient’s strain carries stable, chromosome-encoded resistance to several widely used antibiotics and key genes that help it evade immune attack and cling to human tissues. At the same time, its close genetic relationship with outbreak-associated strains signals that dangerous variants can emerge from the same family tree. By dissecting the genome of a single, well-documented clinical isolate, this study deepens our understanding of how S. suis ST1 operates and evolves, providing a foundation for better surveillance, smarter antibiotic use, and more targeted strategies to protect both farmers and the wider public.
Citation: Jiang, J., Duan, W. & Liang, L. Genomic analysis of a clinical Streptococcus suis ST1 isolate from CSF reveals antimicrobial resistance, virulence, and an evolutionary link to ST7. Sci Rep 16, 11271 (2026). https://doi.org/10.1038/s41598-026-41475-6
Keywords: Streptococcus suis, zoonotic meningitis, antibiotic resistance, bacterial genomics, pig-to-human infection