Clear Sky Science · en
The Caspase-1-EGR4 axis regulates macrophage repolarization in acute myeloid leukemia cells
Rebalancing the body’s cancer-fighting cells
Acute myeloid leukemia (AML) is an aggressive blood cancer that does more than grow out of control—it also quietly reshapes the immune system so the tumor can thrive. This study uncovers how leukemia cells can push nearby immune cells called macrophages into a tumor-supporting mode, and identifies a molecular "switch" that may be turned to push those same cells back into a tumor-fighting state. Understanding this switch could open new ways to make existing immune-based treatments more effective.
Friendly and unfriendly immune helpers
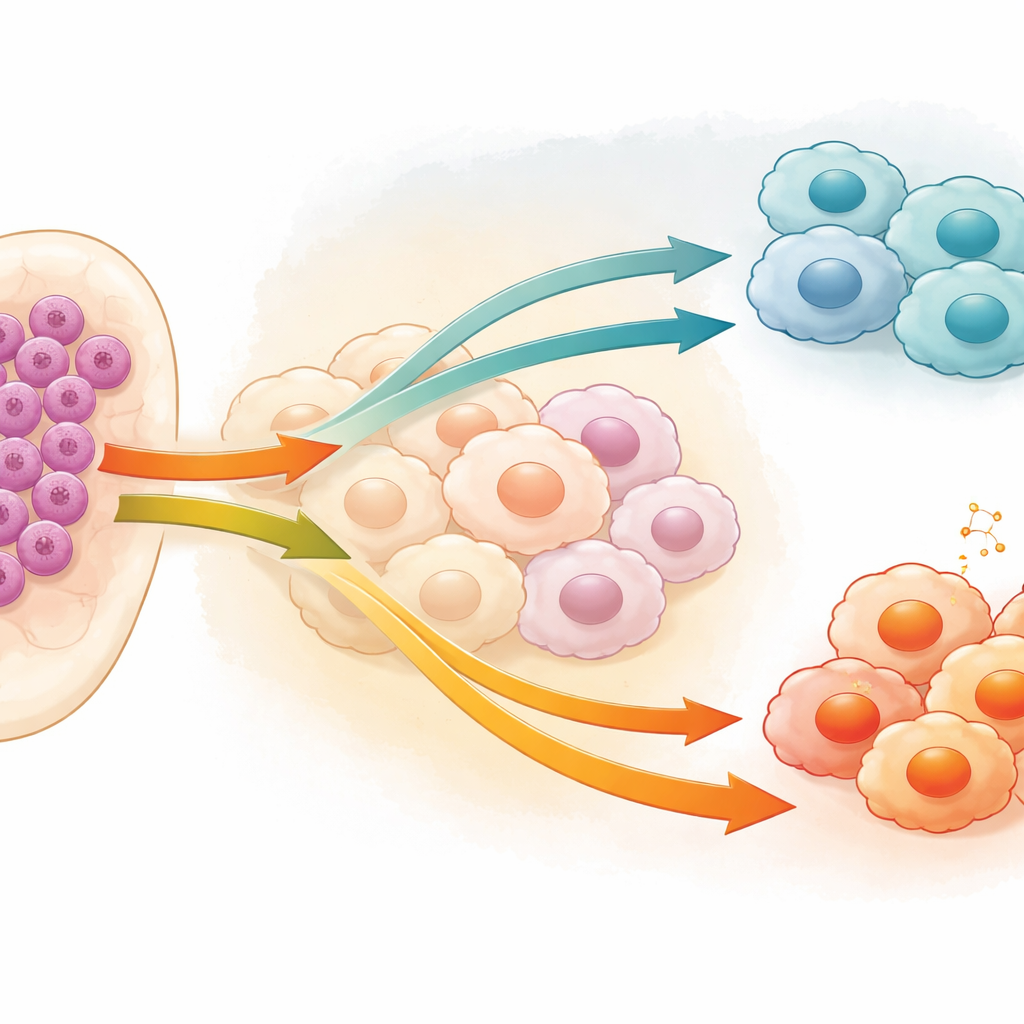
Macrophages are white blood cells that normally help defend the body and clean up damaged tissue. In many cancers, including AML, they can be “re-educated” into a nurturing role that protects tumor cells instead of attacking them. These tumor-associated macrophages tend to adopt an M2-like state, which calms inflammation and supports cancer cell survival, while the M1 state is more aggressive and hostile to tumors. The authors focused on how leukemia cells influence this balance, asking which signals coming from the cancer cells themselves tell macrophages to become either harmful helpers or helpful fighters.
A hidden signaling route inside leukemia cells
The team zeroed in on a protein called caspase-1 (CASP1), best known for its role in inflammatory cell death, but whose behavior in cancer is complex and sometimes contradictory. They found that AML cells often have high levels of CASP1, and that this increase is linked to a greater presence of tumor-supporting M2 macrophages. Using two human AML cell lines, they reduced CASP1 activity and collected the cocktail of molecules these altered leukemia cells released into their surroundings. When this conditioned medium was applied to macrophages grown in the lab, the macrophages displayed fewer M2 features and more characteristics associated with the M1, tumor-fighting state. This shift showed up at the gene level, in protein markers on the cell surface, and in the overall pattern of cell populations.
Discovering a key brake: EGR4
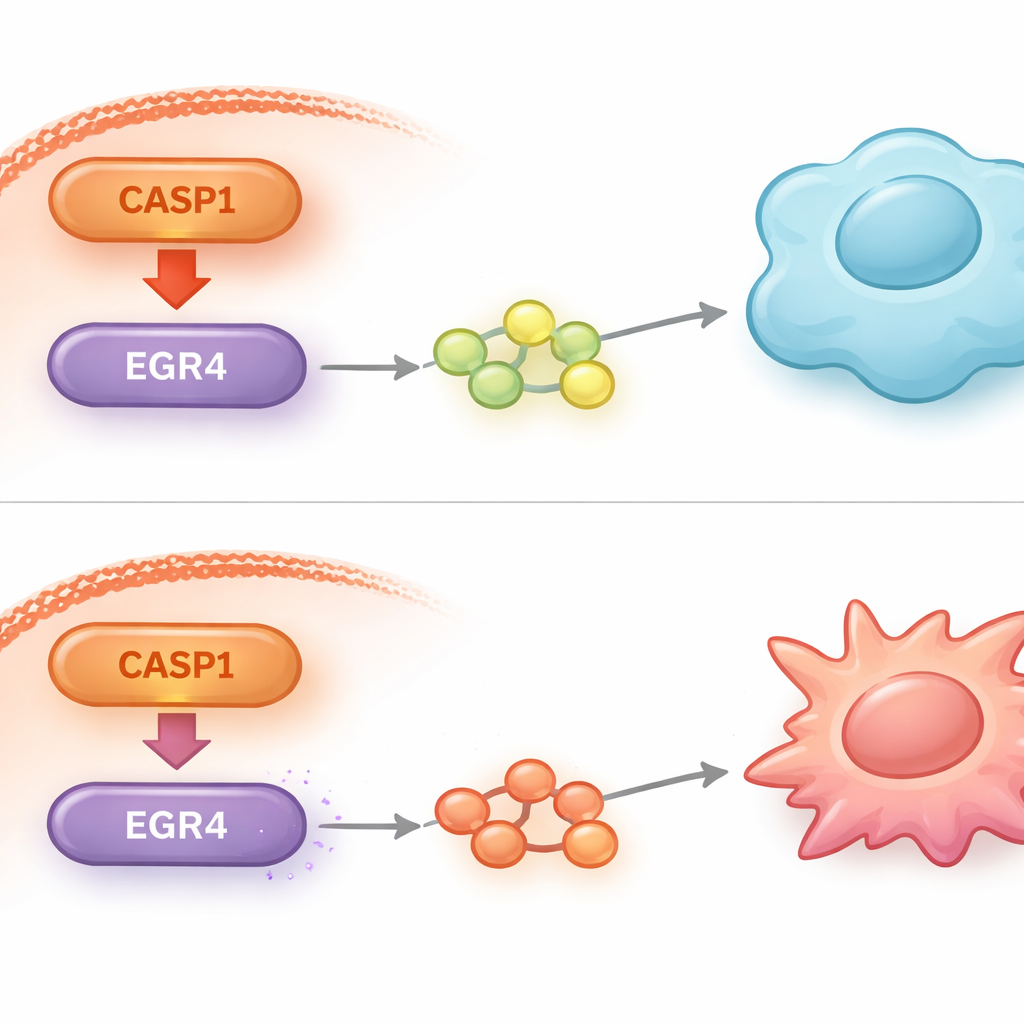
To understand how CASP1 inside leukemia cells could so strongly influence outside macrophages, the researchers examined global gene activity after CASP1 was reduced. They discovered that levels of a transcription factor called EGR4 rose sharply when CASP1 was knocked down. Further tests confirmed that EGR4 acts like a molecular brake on the leukemia cell’s ability to drive macrophages into the M2 state. When the scientists deliberately lowered EGR4 again in CASP1-deficient cells, the earlier beneficial shift in macrophage behavior was partially lost. This indicated that EGR4 is a crucial downstream player through which CASP1 exerts its effects on the tumor environment.
How one pathway tips the balance
Diving deeper, the study linked the CASP1–EGR4 pairing to another well-known signaling route, the IL-10/STAT3 pathway, which strongly favors M2-like, tumor-supporting macrophages. When CASP1 was reduced in leukemia cells, the signals that normally activate this pathway in nearby macrophages were dampened, particularly the anti-inflammatory molecule IL-10 and its partner, phosphorylated STAT3. Lowering EGR4 undid much of this dampening, again highlighting its central role. In mice implanted with human AML cells, blocking CASP1 not only slowed tumor growth but also remodeled the tumor microenvironment: macrophages shifted away from M2 traits, markers of cell division dropped, and EGR4 levels rose, all consistent with a less welcoming niche for leukemia cells.
What this could mean for future treatment
To a non-specialist, the main takeaway is that the leukemia cells are not acting alone; they are actively training nearby immune cells to protect them. This work identifies a specific chain of command—CASP1 controlling EGR4, which in turn shapes signals that push macrophages into a tumor-friendly role. Breaking this chain makes the tumor’s surroundings less supportive and slows cancer growth in animals. Because drugs that inhibit CASP1 already exist and have been tested in humans for other conditions, this CASP1–EGR4 pathway offers a promising new angle for therapies aimed at reawakening the body’s own immune defenses against AML.
Citation: Qian, Y., Chen, Y., Feng, ZX. et al. The Caspase-1-EGR4 axis regulates macrophage repolarization in acute myeloid leukemia cells. Sci Rep 16, 11319 (2026). https://doi.org/10.1038/s41598-026-41381-x
Keywords: acute myeloid leukemia, tumor microenvironment, macrophage polarization, caspase-1, STAT3 signaling