Clear Sky Science · en
Evaluation of the anti-inflammatory potential of atorvastatin targeting TNF-α, IL-6, and IL-1β using integrated in vitro and in silico approaches
Why a Cholesterol Drug Matters for Inflammation
Millions of people take atorvastatin to lower their cholesterol and protect their hearts. This study asks a surprising question: could the same pill also calm harmful inflammation throughout the body? By combining computer modeling with cell experiments, the researchers explore whether atorvastatin can directly dial down powerful inflammatory messengers that drive diseases such as arthritis, diabetes, and heart disease.
How the Body’s Alarm System Can Turn Against Us
Inflammation is the body’s built-in alarm system, switched on when we face infection or injury. In the short term it is protective, calling in immune cells and helping repair tissue. Problems arise when the alarm never really shuts off. Then, key chemical messengers called cytokines—particularly TNF-alpha, IL-6, and IL-1 beta—stay elevated and keep the body in a constant state of low-level battle. This chronic inflammation is linked to a wide range of illnesses, from joint disease and clogged arteries to neurological disorders and some cancers. Because completely new drugs are slow and costly to develop, scientists are keen to see whether trusted medicines like statins can be repurposed to target these inflammatory signals.
Probing Drug–Protein Encounters in the Computer
The team first used advanced computer simulations to see how atorvastatin might physically interact with the three cytokines. They performed molecular docking, a method that predicts how snugly a small molecule might fit onto a protein’s surface, and followed this with long molecular dynamics simulations to watch the complex over time. Atorvastatin showed the tightest and most stable fit with TNF-alpha, settling into a surface region used to latch onto its receptor on cells. The virtual complex remained compact and steady over a 200-nanosecond run, and energy calculations suggested that attractive forces between oily (hydrophobic) patches and ring-shaped regions of the drug and protein strongly favored binding.
Testing the Anti-Inflammatory Effect in Immune Cells
To see whether these computer predictions translate into real biology, the researchers turned to a standard laboratory model of inflammation. They exposed mouse macrophage cells—immune cells that help coordinate inflammatory responses—to a bacterial component that strongly activates them. Before this trigger, cells were treated with different doses of atorvastatin. The drug did not harm cell survival up to 20 micromolar, meaning its effects were not due to toxicity. As the dose increased, the cells released steadily less TNF-alpha, IL-6, and IL-1 beta into their surroundings. At the highest dose, levels of these inflammatory messengers dropped sharply. Atorvastatin also cut production of nitric oxide, another molecule that rises during inflammation and can damage tissues when overproduced.
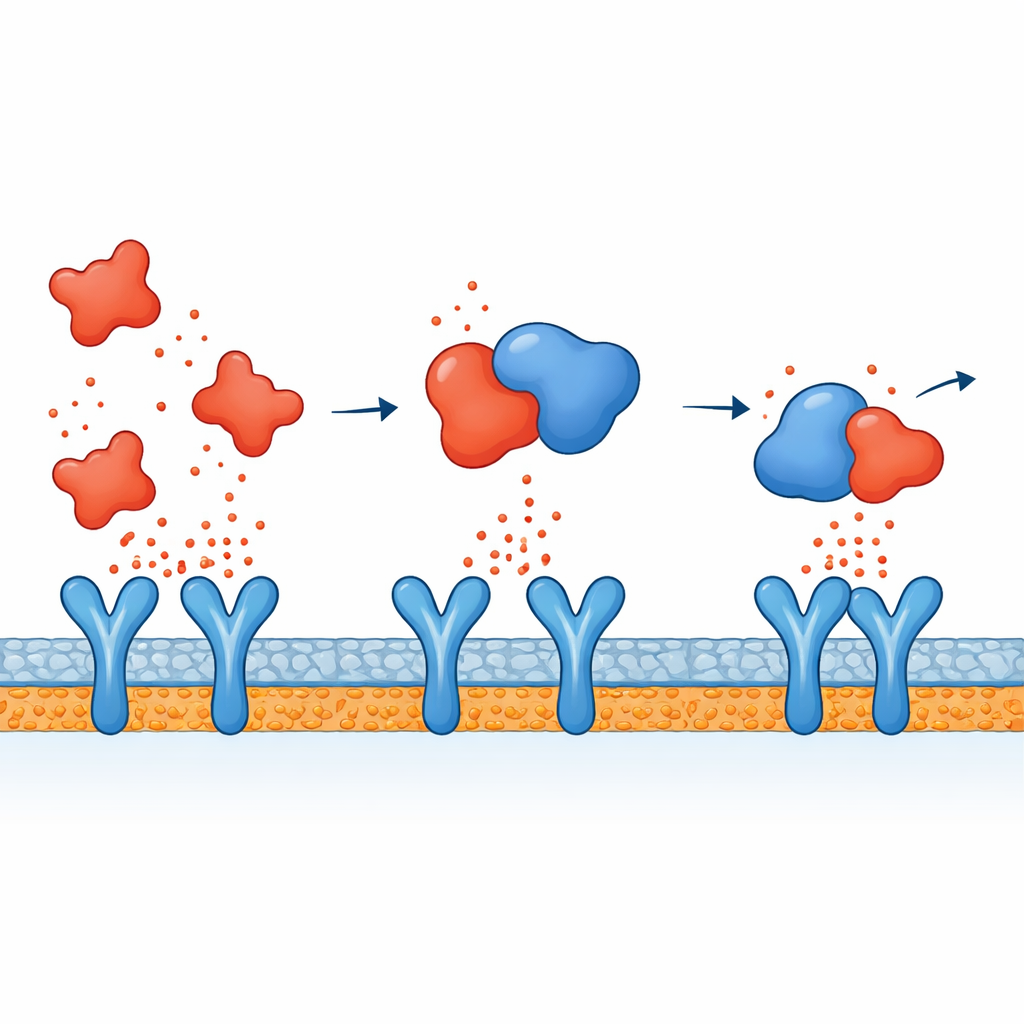
Linking Molecule Binding to Calmer Signaling
Putting the pieces together, the authors propose a two-pronged action for atorvastatin. Inside immune cells, it seems to dampen the signaling pathways that normally turn on inflammatory genes after a microbial threat, so fewer cytokines are made in the first place. At the same time, the simulations suggest that any cytokines that do escape into the bloodstream may be partially neutralized because atorvastatin binds directly to them, especially TNF-alpha, making it harder for these messengers to dock on their receptors and propagate the alarm. This combined effect could help explain why statins have been observed to lower inflammatory markers in patients, beyond their well-known impact on cholesterol.
What This Could Mean for Patients
For a general reader, the key takeaway is that a familiar cholesterol-lowering drug may also act as a multi-purpose inflammation modulator. While this work was done in computer models and cell cultures—not yet in patients—it offers a detailed, molecular-level picture of how atorvastatin might directly disarm some of the body’s most potent inflammatory signals without harming cells. The findings strengthen the case for testing atorvastatin, and possibly its close chemical cousins, as affordable and widely available treatments for chronic inflammatory conditions, alongside their established role in protecting the heart.
Citation: Swaroop, K., M., R., Shaik, S. et al. Evaluation of the anti-inflammatory potential of atorvastatin targeting TNF-α, IL-6, and IL-1β using integrated in vitro and in silico approaches. Sci Rep 16, 9914 (2026). https://doi.org/10.1038/s41598-026-39082-6
Keywords: atorvastatin, chronic inflammation, cytokines, drug repurposing, TNF-alpha