Clear Sky Science · en
MTHFR polymorphism is associated with increased adverse events and poor clinical outcomes in gastric cancer patients with adjuvant S-1 chemotherapy
Why some people struggle more with the same cancer pill
Two patients can take the very same chemotherapy pill and have very different experiences: one finishes treatment with only mild discomfort, while another faces serious side effects and a higher chance of the cancer returning. This study asks a simple but crucial question for people with stomach cancer: can common inherited differences in our DNA help explain who does well and who does poorly on a widely used oral drug called S-1?
A pill designed to make tough treatment easier
S-1 is an oral form of chemotherapy used after surgery for stomach cancer, especially in East Asia. Instead of requiring a continuous hospital infusion, S-1 is taken as pills for a year, making life easier for patients. The drug is built around an older anti-cancer agent, 5-fluorouracil, but adds two helper ingredients that change how it is processed in the body. Large clinical trials have already shown that a year of S-1 after surgery can cut the risk of the cancer coming back and help patients live longer. But not everyone can tolerate a full year of treatment, and doctors have noticed that side effects and benefits vary widely from person to person.
Looking at DNA clues in treated patients
The researchers studied 334 people with stage II or III stomach cancer who had their tumors surgically removed and then took S-1 as adjuvant (after-surgery) treatment. Everyone in the study had good organ function at the start, and their blood was drawn before chemotherapy to look for small, common DNA differences known as single nucleotide polymorphisms, or SNPs. The team focused on 46 SNPs in six genes that help process 5-fluorouracil and related drugs. They then followed patients for a median of more than seven years, carefully recording treatment side effects, how much drug each person actually received over time, how long they stayed free of cancer, and how long they lived overall.
One common DNA variant stands out
Among all the genetic markers, one particular change in a gene called MTHFR emerged as especially important. This variant, named rs1801133, affects an enzyme that manages folate, a vitamin closely tied to building and repairing DNA. People who inherited two copies of the risk version were about three times more likely to develop blood-related side effects such as anemia or low white blood cell counts. When the researchers looked at long-term outcomes, patients carrying at least one copy of the risk version had shorter periods without cancer returning and shorter overall survival than those with the typical version. Another gene involved in drug breakdown, CYP2A6, also showed a link to worse cancer-free survival, but its impact was weaker and less consistent.
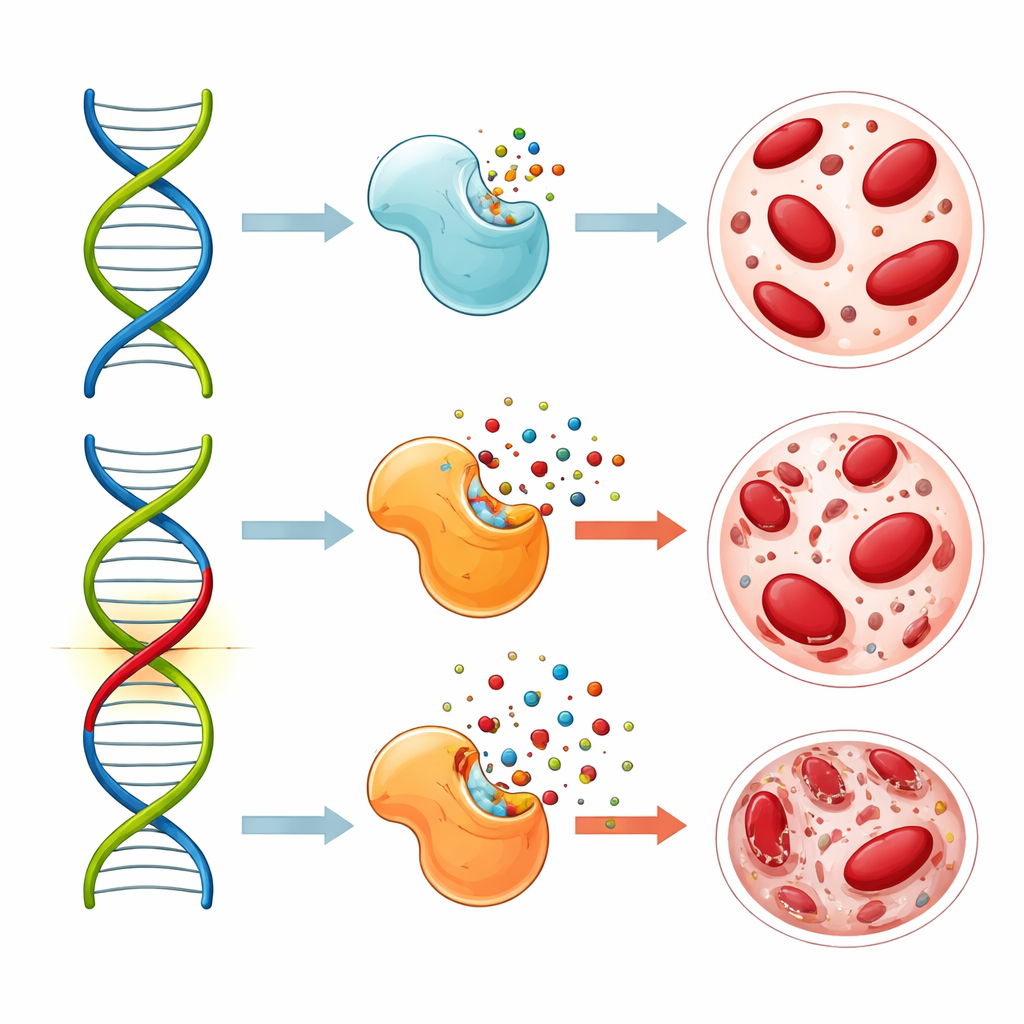
Why this DNA change might worsen side effects and outcomes
Laboratory work from other studies has shown that the MTHFR variant reduces the activity of its enzyme, tilting the folate balance inside cells. This shift can make 5-fluorouracil more potent at blocking the enzymes that cancer cells use to copy their DNA. In theory, that might sound like a good thing, because stronger drug action could mean stronger attack on cancer. But the same heightened effect also hits fast-growing healthy cells, especially in the bone marrow where blood cells are made. In this study, patients with the risk variant indeed had more serious blood problems and, unexpectedly, did not gain better cancer control; instead, they tended to do worse over the long term. The authors suggest that complex interactions between prolonged drug exposure from S-1 and the altered folate pathway may damage healthy tissues without delivering extra benefit against the tumor.
What this could mean for future cancer care
The findings point to the MTHFR rs1801133 variant as a potential biomarker—an inherited clue that could flag patients who are more likely to suffer from blood toxicities and have poorer results on standard S-1 therapy. The study does not yet prove that treatment should be changed based on this single DNA test, but it raises the possibility that, in the future, genetic screening could help doctors tailor doses, increase monitoring, or consider alternative regimens for higher-risk patients. In simple terms, the work moves cancer care one step closer to a world where a pill is not just chosen by tumor stage, but also by how each person’s genes handle the drug.
Citation: Kang, M., Kim, J.W., Lee, J.H. et al. MTHFR polymorphism is associated with increased adverse events and poor clinical outcomes in gastric cancer patients with adjuvant S-1 chemotherapy. Sci Rep 16, 14196 (2026). https://doi.org/10.1038/s41598-026-38429-3
Keywords: gastric cancer, S-1 chemotherapy, pharmacogenomics, MTHFR polymorphism, treatment toxicity