Clear Sky Science · en
Fecal microbiota transplantation plus immunotherapy in metastatic renal cell carcinoma: the phase 1 PERFORM trial
Why Your Gut Might Matter in Kidney Cancer Treatment
Immunotherapy has changed how doctors treat advanced kidney cancer, giving some patients years of extra life. But these powerful drugs can also unleash the immune system on healthy organs, causing serious, sometimes life-threatening side effects. This study asks a deceptively simple question with big implications: if we first "reset" the gut microbiome using carefully screened donor stool in a capsule, can we make immunotherapy both safer and more effective?
A New Use for a Strange-Sounding Treatment
The PERFORM trial tested an approach called fecal microbiota transplantation, or FMT, in people with metastatic renal cell carcinoma, the most common form of advanced kidney cancer. Instead of traditional colon-delivered FMT, patients swallowed frozen capsules made from stool donated by rigorously screened healthy volunteers. Twenty patients who had not yet received systemic treatment took three rounds of these capsules and then started standard immunotherapy-based regimens, such as dual immune checkpoint inhibitors or immunotherapy combined with targeted drugs. The main goal was safety—would adding donor microbes on top of powerful immune drugs create unacceptable risks?
Safety, Side Effects, and Early Benefits
The good news is that FMT itself appeared safe. Only one patient had a mild gut symptom clearly linked to the capsules. The more serious immune-related side effects—such as colitis, rash, arthritis, or hormone problems—matched what would normally be expected from the immunotherapy drugs alone. Half of the patients experienced severe (grade 3) immune toxicities, but none had the most dangerous grade 4 or 5 events, and no unexpected complications emerged. On the benefit side, among the 18 patients whose tumors could be measured, 9 showed meaningful shrinkage of their cancer and 2 had complete responses. Strikingly, most of the patients whose tumors responded did not suffer severe immune side effects, hinting that a well-tuned microbiome might help patients get the best of both worlds: strong anti-tumor activity with less collateral damage.
The Gut Community as a Balancing Act
To understand why some patients tolerated treatment better, the researchers tracked the microbes, molecules, and immune cells in blood and stool over ten weeks. Patients who avoided severe side effects developed more diverse gut communities after FMT and kept them over time. Their microbial profiles became more similar to those of their healthy donors, not just in which species were present but in what those microbes could do—especially their ability to produce anti-inflammatory compounds such as short-chain fatty acids. These patients also maintained higher levels of certain circulating metabolites, including vitamin A, specific amino acids, and energy-related molecules, all of which have been linked to healthier immune regulation and stress responses. Together, this pattern suggests that a stable, functionally rich gut ecosystem can help cushion the body against the inflammatory shocks of immunotherapy while still supporting its cancer-fighting effects.
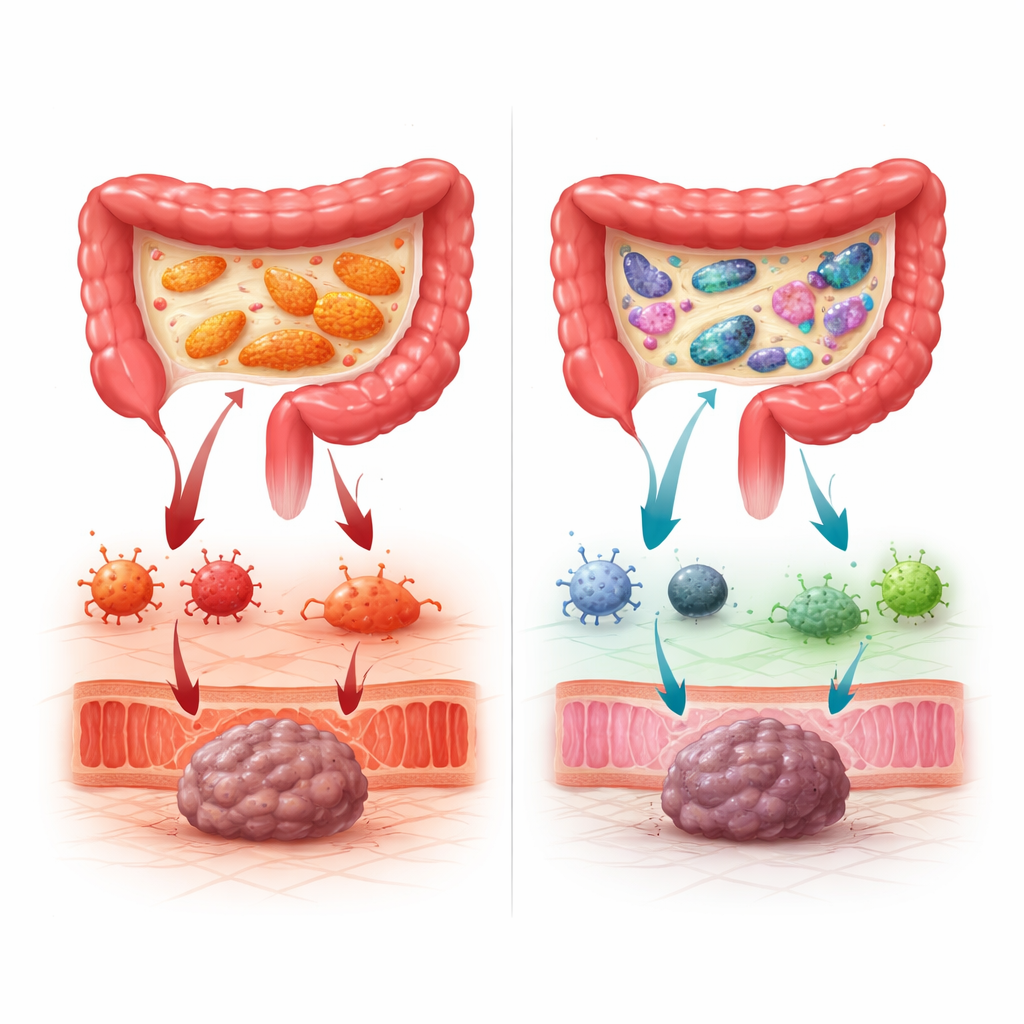
When One Bacterium Tips the Scales
Not all bacteria were helpful. The team identified one species in particular, Segatella copri, that became a troublemaker when it grew beyond a certain threshold. Patients who ended up with high levels of this microbe after FMT were much more likely to develop severe immune-related colitis and other toxicities, especially when treated with the combination of ipilimumab and nivolumab. Many of these patients also failed to respond to treatment. Importantly, it did not matter whether S. copri came from the donor or was already present at low levels in the patient: if conditions allowed it to bloom, the risk of toxicity and resistance increased. High S. copri levels were linked to a blood “fingerprint” of inflammation and to immune cell patterns showing overactive killer T cells but fewer regulatory cells and protective natural killer cells, signaling a loss of internal checks and balances.
What This Means for Future Cancer Care
In simple terms, this study suggests that shaping the gut microbiome before and during immunotherapy can influence whether the immune system fights cancer effectively without turning on the body itself. Introducing a carefully chosen mix of healthy microbes via FMT capsules was safe in this small trial and was associated with better responses and fewer severe side effects when the transplanted community remained diverse, anti-inflammatory, and functionally robust. At the same time, the findings warn that certain bacteria like S. copri, and specific microbial traits that promote inflammation, may tip patients toward dangerous toxicity and poor outcomes. While larger trials are needed, PERFORM points toward a future in which oncologists might screen both donors and patients by their microbes, selectively boost protective species, and avoid risky ones—using the gut as a powerful lever to fine-tune cancer immunotherapy.
Citation: Fernandes, R., Jabbarizadeh, B., Rajeh, A. et al. Fecal microbiota transplantation plus immunotherapy in metastatic renal cell carcinoma: the phase 1 PERFORM trial. Nat Med 32, 1325–1336 (2026). https://doi.org/10.1038/s41591-025-04183-8
Keywords: metastatic renal cell carcinoma, fecal microbiota transplantation, gut microbiome, immune checkpoint inhibitors, treatment toxicity