Clear Sky Science · en
Tau phosphorylation impedes functionality of protective tau envelopes
Why tiny cell “sleeves” matter for brain health
Inside every nerve cell, long hollow rods called microtubules act like train tracks, carrying cargo that keeps the cell alive. These tracks are coated by a protein named tau, which forms a protective sleeve around them. In Alzheimer’s disease and related brain disorders, tau becomes heavily modified and clumps into tangles, while microtubules fall apart. This paper uncovers a missing piece of that puzzle: it shows that a common chemical change to tau, called phosphorylation, directly weakens these protective sleeves and leaves the cell’s internal tracks vulnerable to damage.
Protective coats on the cell’s inner rails
Microtubules are long protein tubes that give nerve cells their shape and provide highways for vital transport. Under healthy conditions, tau molecules don’t just float freely; they can gather tightly on the surface of microtubules to form dense, continuous layers the authors call “envelopes.” These envelopes act like insulating sleeves: they slow down or block certain other proteins, including molecular scissors known as severing enzymes that can cut microtubules into pieces. Earlier work showed that when tau is only loosely attached and moving individually, microtubules are easy targets for these scissors. When tau forms cohesive envelopes, the tracks are strongly protected. 
How chemical “tags” break the coat
The team asked what happens when tau is phosphorylated, a normal regulatory process that is greatly exaggerated in degenerating brains. Using purified proteins and high-resolution microscopy, they compared dephosphorylated tau (with few chemical tags) to tau bearing many phosphate groups. At very low concentrations, dephosphorylated tau readily assembled into large, continuous envelopes along microtubules, while phosphorylated tau mostly stayed as individual, mobile molecules. Even when both forms were present at higher levels and could form envelopes, the phosphorylated version covered less of the microtubule surface and its envelopes were thinner and more “gap-prone,” showing reduced cohesiveness between tau molecules.
Watching the sleeves fall apart in real time
To probe stability, the researchers built envelopes from relatively unmodified tau and then added a brain-related enzyme, Cdk5, that adds phosphate groups to tau. They saw the envelopes retreat from their edges and even split into pieces, like a peeling coat. Further analysis showed that phosphorylation not only weakens tau’s grip on microtubules but also reduces how strongly tau molecules stick to one another, both of which are needed for a solid sleeve. In living cells, they used fluorescence techniques to monitor tau on microtubules. Normal tau behaved as a slow-turnover, almost glued-on layer, consistent with an envelope. Tau mutants unable to form envelopes, or tau exposed to Cdk5, exchanged much faster, indicating a looser, more fragile attachment that matches the in‑vitro results. 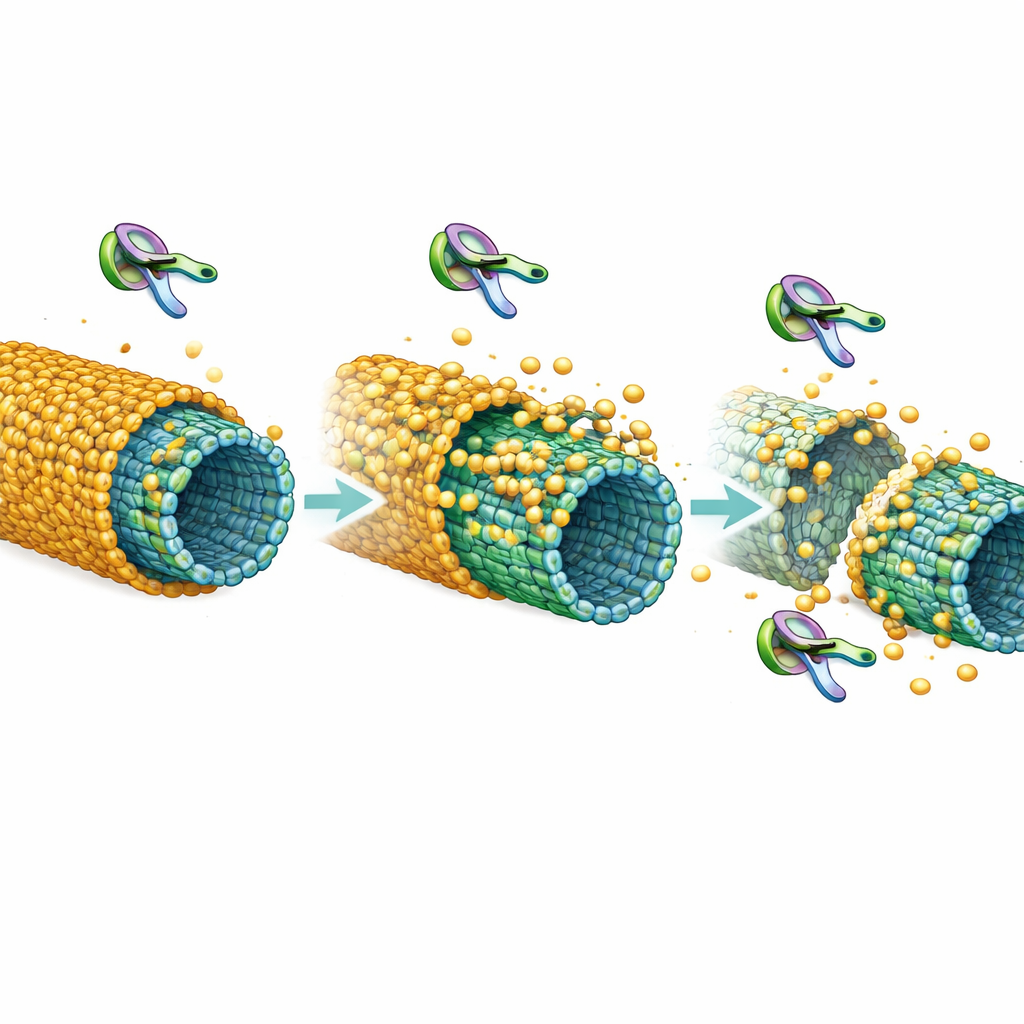
From fragile sleeves to broken tracks
The crucial question was whether these changes matter for microtubule survival. The authors turned to katanin, a powerful severing enzyme. On bare or loosely coated microtubules, katanin rapidly chopped the tubes. When microtubules were wrapped in robust tau envelopes, they remained largely intact, confirming the protective role of the coat. However, when envelopes were made from phosphorylated tau, protection was noticeably weaker: more cuts appeared within the coated regions, and microtubules disassembled faster even though tau density looked similar. In cultured cells, overproducing katanin alone led to a sharp loss of microtubules, but co‑expressing full-length tau rescued the lattice. Co‑expressing tau that cannot form envelopes, or driving extra phosphorylation of tau via Cdk5, largely removed this protection and allowed microtubules to erode.
What this means for diseases like Alzheimer’s
Overall, the study shows that heavy phosphorylation of tau does more than nudge it off microtubules: it disrupts the formation, stability and shielding power of tau envelopes. As tau becomes more phosphorylated, envelopes disassemble, tau is released into the cell where it may aggregate, and the exposed microtubules become easy targets for severing enzymes. This combination can gradually strip axons and dendrites of their internal tracks, undermining transport and structure long before large tangles appear. For a layperson, the message is that Alzheimer’s‑linked chemical changes to tau weaken the microscopic “sleeves” that normally guard nerve-cell highways, helping explain how the brain’s wiring starts to fray at the molecular level.
Citation: Siahaan, V., Weissova, R., Karhanova, A. et al. Tau phosphorylation impedes functionality of protective tau envelopes. Nat Chem Biol 22, 759–769 (2026). https://doi.org/10.1038/s41589-025-02122-9
Keywords: tau protein, microtubules, phosphorylation, Alzheimer’s disease, neurodegeneration