Clear Sky Science · en
TBK1 restricts IRGQ-mediated autophagy
How Cells Decide What to Keep and What to Trash
Inside every cell, a busy recycling system constantly sorts out worn-out parts and misfolded proteins before they cause trouble. This study uncovers how a small protein hub helps start this cellular cleanup for specific cargo, and how another enzyme can flip that hub into an off state. Understanding this switch sheds light on how cells fine-tune waste disposal, with implications for aging, cancer, infections, and immune responses.
A Targeted Cleanup Crew Inside Cells
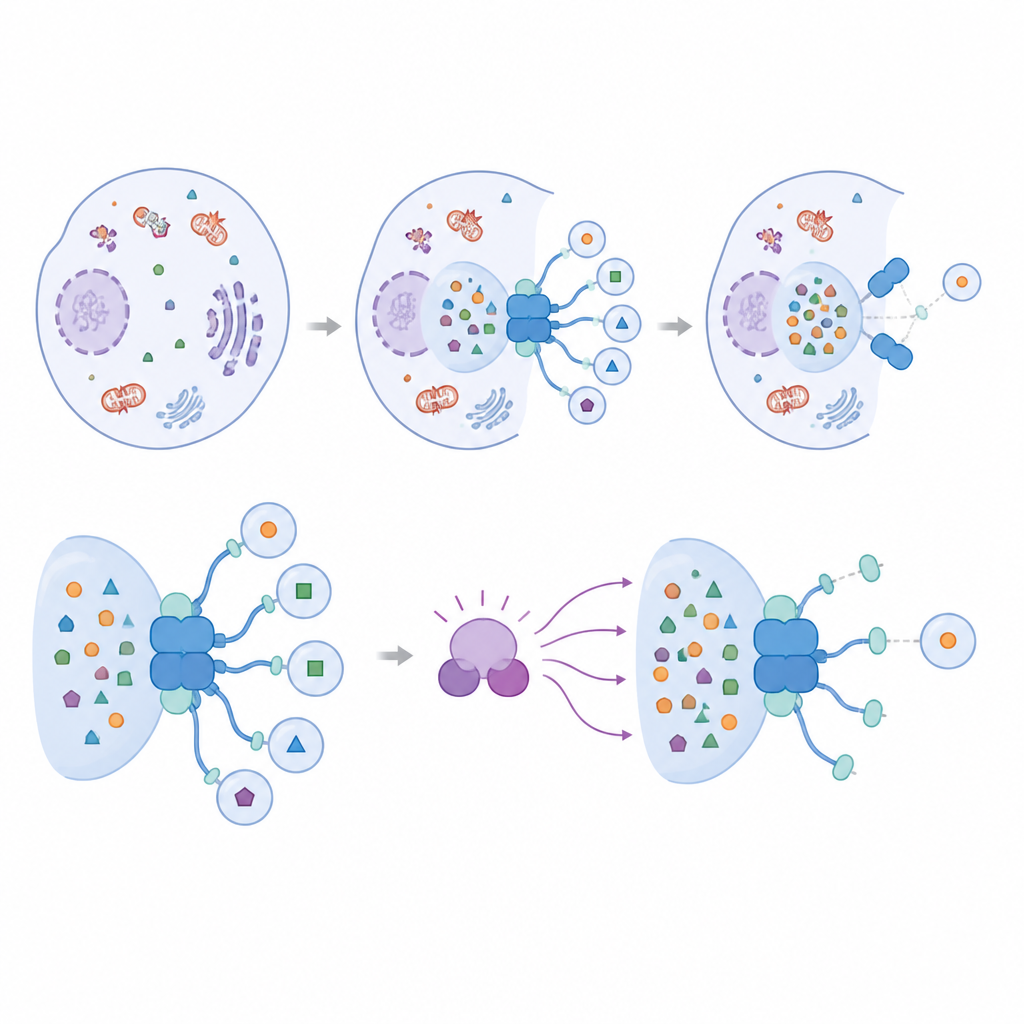
Cells do not just randomly digest their contents. Instead, they use a process called autophagy to wrap selected material in tiny membrane sacs and deliver it to acidic compartments for breakdown. Specialized receptor proteins help pick which cargo gets packed and help recruit the core cleanup machinery. The protein IRGQ was recently identified as one such receptor that recognizes certain versions of immune display molecules on the cell surface. In this work, the authors show that IRGQ is more than a simple tagger of cargo; it also organizes the early steps of autophagosome formation.
A Protein Hub That Bridges Cargo and Machinery
IRGQ interacts with a family of small adaptor proteins known as ATG8s, which decorate the forming autophagosome membrane and are essential for building these recycling sacs. The team found that IRGQ carries two separate docking sites that can bind different ATG8 members. One of these sites shows a striking preference for a single ATG8 protein called GABARAPL2. Using structural predictions, protein pulldown assays, and microscopy, the researchers demonstrate that IRGQ and GABARAPL2 together form a hub that brings in key enzymes responsible for attaching ATG8 proteins to membranes. When this hub is present, cells show increased ATG8 attachment and more autophagic activity for specific cargos, confirming that IRGQ actively promotes the initiation of selective cleanup rather than just labeling material for disposal.
A Molecular Brake on the Cleanup Hub
The study then asks how this powerful initiation hub is controlled. The focus turns to TBK1, an enzyme already known to influence several forms of selective autophagy. The authors show that TBK1 directly modifies GABARAPL2 at a single amino acid close to its contact point with IRGQ. When this site is altered to mimic a permanently modified state, the bond between IRGQ and GABARAPL2 weakens sharply, and the complex falls apart. Advanced modeling and cell-based experiments agree that this change does not broadly disrupt GABARAPL2’s ability to interact with other partners, but rather specifically destabilizes its link to IRGQ and the associated initiation machinery.
Selective Effects on Cargo Rather Than Global Recycling
Breaking apart the IRGQ–GABARAPL2 hub has surprisingly focused consequences. Bulk autophagy, the cell’s general recycling response during starvation, continues largely unchanged even when the modification site on GABARAPL2 is altered. However, the trafficking of GABARAPL2 itself and of IRGQ’s cargo, a subset of major histocompatibility complex class I molecules, is clearly impaired. In cells carrying the phospho-mimicking version of GABARAPL2, these immune molecules accumulate at the cell surface instead of reaching lysosomes for breakdown, closely mirroring what happens when IRGQ is missing. Conversely, blocking TBK1 activity pushes more of this cargo toward lysosomal compartments, indicating that TBK1 normally acts to restrain this particular route of autophagic clearance.
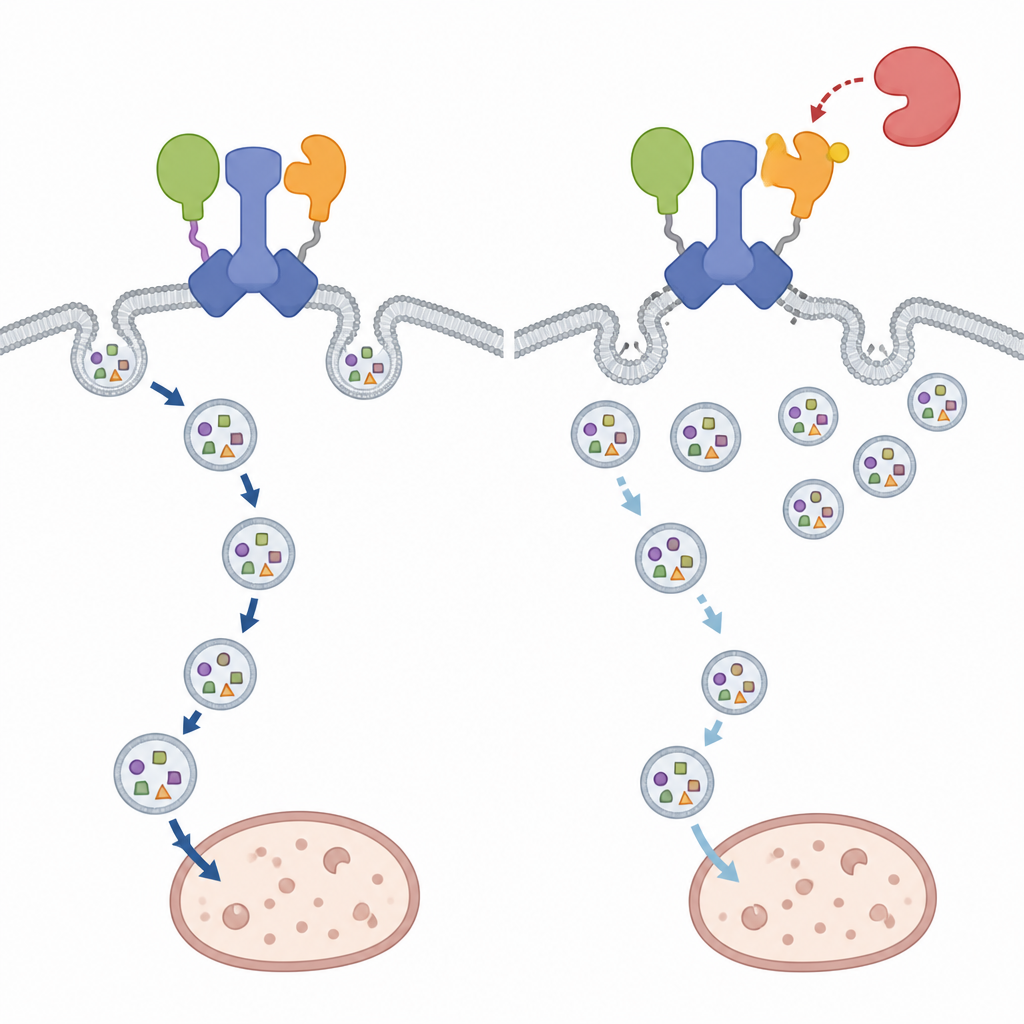
A Context-Dependent Stop Signal for Cellular Recycling
Taken together, the findings reveal IRGQ as a central organizer that links chosen cargo to the early steps of autophagosome formation, and TBK1 as a context-dependent brake on this pathway. By modifying GABARAPL2 at just the right moment, TBK1 can switch off the IRGQ hub without shutting down the cell’s global recycling system. For a lay reader, this means cells possess a finely tuned switch that can turn a specific cleanup route on or off, shaping how immune signals are displayed and how damaged components are removed in different situations.
Citation: Gestal-Mato, U., Lascaux, P., Poveda-Cuevas, S.A. et al. TBK1 restricts IRGQ-mediated autophagy. Nat Commun 17, 4335 (2026). https://doi.org/10.1038/s41467-026-73005-3
Keywords: selective autophagy, IRGQ, TBK1, GABARAPL2, MHC class I quality control