Clear Sky Science · en
Rare regulatory mutations disrupt mesenchymal molecular programs driving endocardial cushion formation in bicuspid aortic valve
When Heart Valves Form Differently
Bicuspid aortic valve is a common heart birth defect where the main heart valve to the body has only two flaps instead of three. Many people with this condition eventually need surgery, yet doctors still cannot fully explain why it happens or which family members are most at risk. This study digs deep into our DNA’s control switches to reveal how rare, hard‑to‑spot changes in gene regulation during early development can misdirect the building of heart valves and help produce a bicuspid aortic valve.
From Normal Valve to Hidden Risk
The aortic valve works like a door between the heart and the body’s largest artery. Most people are born with three leaflets that open and close smoothly with each heartbeat. In up to about 1.5% of the population, however, the valve has only two leaflets. This bicuspid shape can strain the heart, promote dangerous narrowing or leaking of the valve, and weaken the nearby aorta. Family studies show that the condition is strongly heritable, but known mutations in the protein‑coding parts of genes explain only a small fraction of cases. The authors suspected that the missing answers might lie in the non‑coding DNA that controls when and where genes turn on.
The DNA Control Map Behind Valve Development
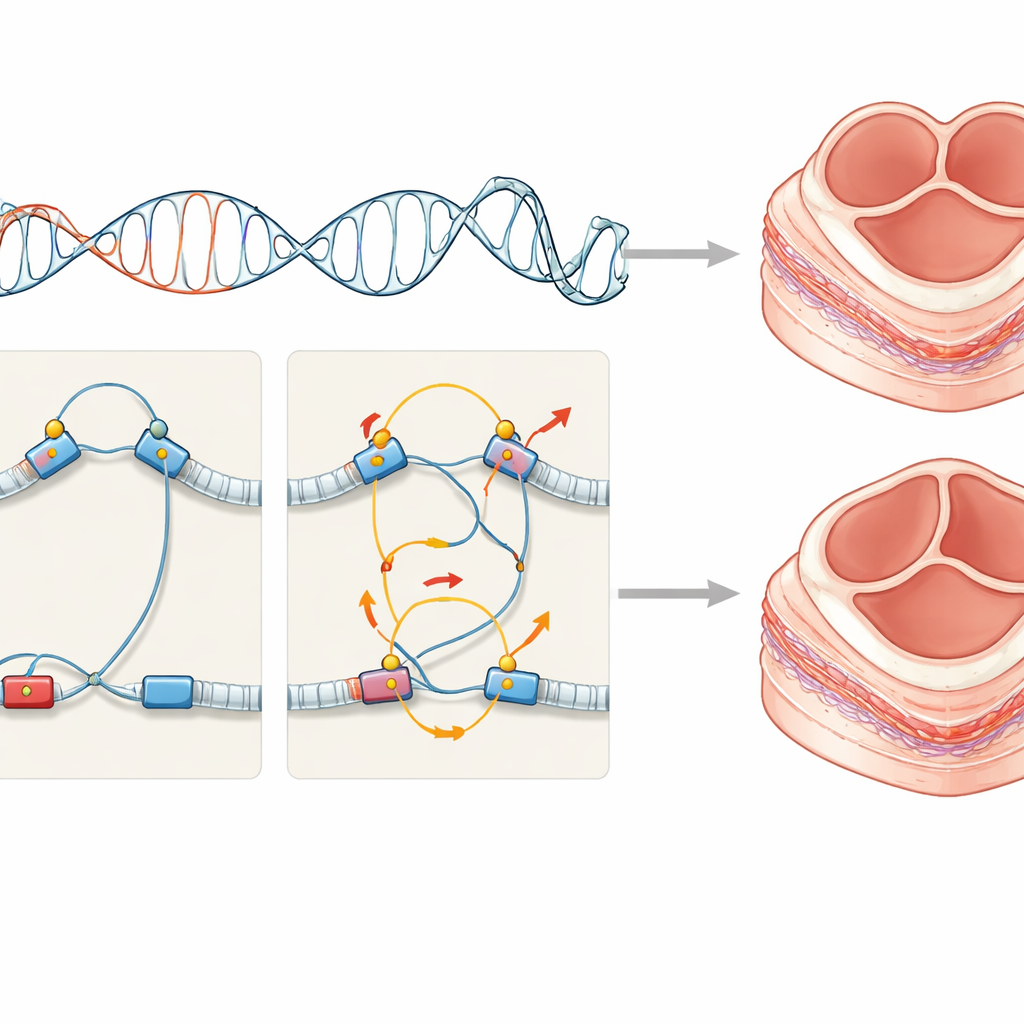
Most of our genome does not code for proteins. Instead, it contains regulatory elements, such as enhancers and promoters, that act like dimmer switches and wiring for genes. These regions can sit far from the genes they control, looping through three‑dimensional space to make contact. To see how this wiring differs between people with bicuspid and normal valves, the researchers collected cells from the inner lining of the ascending aorta from eight patients with bicuspid valves and eight with normal three‑leaflet valves, all undergoing surgery. They used a technique called promoter capture Hi‑C to map millions of long‑range DNA contacts and combined this with whole‑genome sequencing to locate rare mutations sitting inside these regulatory regions.
Rare Mutations that Rewire Developing Valve Cells
The team found that up to 95% of genetic differences between people occurred in non‑coding regions, and that bicuspid‑valve patients showed many more mutation‑linked changes in DNA looping around genes involved in valve development than controls. Rather than relying only on adult tissue signals, the scientists overlaid their wiring maps onto single‑cell and spatial gene‑expression data from human embryonic hearts. This allowed them to ask which early cell types would feel the impact of the altered regulatory switches. The strongest signal came from mesenchymal cells that contribute to structures called endocardial cushions—temporary pads of tissue in the fetal heart that later remodel into the mature heart valves and parts of the outflow tract.
Disrupted Cushion Programs in the Fetal Heart
By tracing which genes were connected to mutated enhancers and promoters, the authors identified 198 candidate genes linked to bicuspid aortic valve, about thirty times more than previously known. These genes clustered in pathways that drive the transformation of flat lining cells into invasive cushion cells, a process known as epithelial‑to‑mesenchymal transition, and in signaling routes such as TGF‑beta and related networks. They also saw repeated hits in binding sites for a family of control proteins related to NFAT, known to be crucial for shaping cushions into mature valves. Computer models of regulatory activity supported the idea that many of these variants act specifically in fetal heart tissue and in the ascending aorta. Together, the results suggest that rare regulatory mutations subtly distort the gene programs of cushion‑forming mesenchymal cells, nudging development toward an abnormal two‑leaflet valve.
A Complex Genetic Puzzle with Many Small Pieces
Looking across patients, the researchers saw that each person carried their own mix of rare regulatory changes, often combined with protein‑altering mutations, and that only a minority of affected genes were shared between individuals. Yet these diverse changes connected into an interacting protein network, pointing to a common biological theme rather than random damage. The work also shows that some regulatory wiring patterns first established in the embryo can still be detected in adult cells, offering a practical window into prenatal events.
What This Means for Patients and Families
For non‑specialists, the main message is that bicuspid aortic valve is not usually caused by a single faulty gene, but by many subtle changes in how genes are switched on during early heart formation. These rare regulatory mutations help explain why the condition runs in families yet is so genetically diverse from one person to another. In the long term, mapping this hidden control layer of the genome could improve genetic risk assessment for relatives and guide research into new ways to monitor or treat people born with this common valve defect.
Citation: Zhigulev, A., Buyan, A., Lázár, E. et al. Rare regulatory mutations disrupt mesenchymal molecular programs driving endocardial cushion formation in bicuspid aortic valve. Nat Commun 17, 3587 (2026). https://doi.org/10.1038/s41467-026-71758-5
Keywords: bicuspid aortic valve, heart valve development, noncoding DNA, regulatory mutations, mesenchymal cells