Clear Sky Science · en
Pericytes are organ-specific regulators of tissue morphogenesis
How Support Cells Quietly Shape Growing Organs
Every organ in the body is laced with tiny blood vessels that deliver oxygen and nutrients. But these vessels do more than carry blood. This study reveals how a lesser-known group of support cells wrapped around capillaries, called pericytes, send local chemical signals that help lungs and brains build the right structures as they mature after birth.
The Hidden Helpers Along Blood Vessels
Blood vessels are lined by endothelial cells, which not only form tubes for blood flow but also release signals that guide nearby cells during organ growth and repair. Pericytes sit closely attached to small vessels and are known to keep vessel walls stable. Until now, their broader role in shaping whole tissues has been unclear. Using advanced genetic tools in mice, the researchers selectively turned off specific signaling molecules produced by pericytes, then tracked how lung and brain development changed. Their work shows that pericytes are not generic helpers; instead, they act in organ-specific ways and talk to different neighboring cells in each tissue.
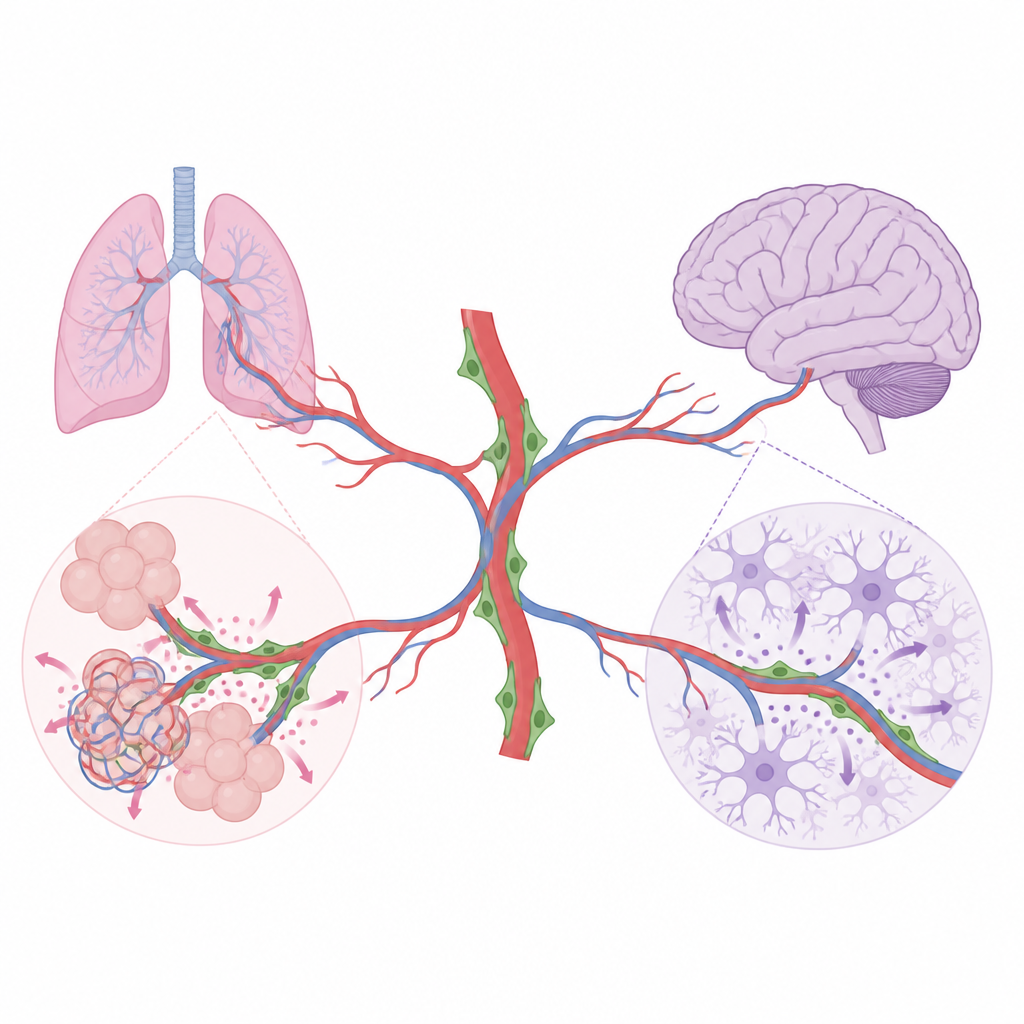
Pericyte Messages That Build Healthy Lungs
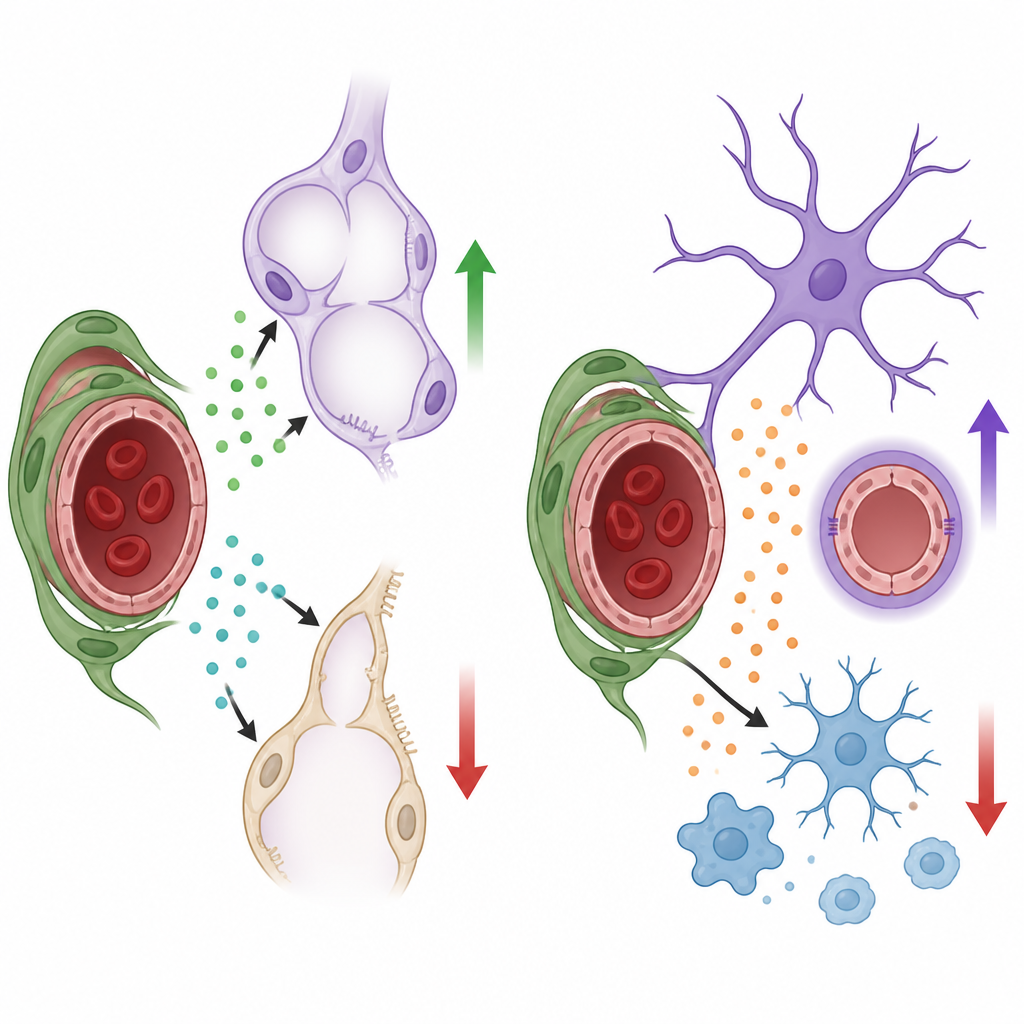
In the developing lung, air sacs must form in great numbers and become closely intertwined with capillaries so gas exchange can work efficiently. The team found that lung pericytes produce two important growth factors, HGF and BDNF. When HGF was removed only from pericytes, the small “stem-like” type 2 alveolar cells that line air sacs multiplied less and produced fewer mature surface-lining cells. The lungs had larger, simplified air spaces and a smaller overall volume, even though basic breathing remained adequate in young mice. When BDNF was removed from pericytes, lung capillaries grew poorly, endothelial cells divided less, and the air spaces again became enlarged. Follow-up experiments showed that BDNF from pericytes activates its partner receptor on lung endothelial cells, helping drive the expansion of the vascular network that is needed for normal air sac formation.
Different Rules for Brain Growth
The same pericyte signals did not matter everywhere. In the brain, pericytes produced little or no HGF and BDNF, and turning off these molecules in mural cells caused no detectable changes in vessel density, barrier tightness, or the behavior of nearby nerve-supporting cells. Instead, brain pericytes relied on a different signal, a protein called Nodal. When Nodal production was blocked specifically in these pericytes after birth, brain capillaries failed to expand normally, and subtle leakage of blood components into brain tissue appeared. Lab tests with cultured mouse brain endothelial cells showed that Nodal could directly boost their growth and movement through a well-known signaling pathway, supporting its role as a pro-growth cue for the brain’s microvessels.
Keeping Brain Support Cells in Balance
Loss of pericyte-derived Nodal had another striking effect: it pushed key brain support cells into a stressed state. Astrocytes, the star-shaped cells that wrap vessels and help maintain the blood–brain barrier, became hypertrophic and expressed higher levels of markers associated with reactive responses to injury. Microglia, the brain’s resident immune cells, adopted more activated shapes and turned on genes linked to inflammation. Single-cell RNA sequencing confirmed that, without Nodal, microglia shifted toward a more pro-inflammatory profile, and astrocytes showed a molecular signature of reactivity. In culture, adding Nodal to isolated astrocytes and microglia activated the same internal signaling switch and dampened several markers of activation, suggesting that pericyte-derived Nodal normally helps keep these support cells and the brain environment in a calmer, more protective state.
What This Means for Organ Health
Together, the findings show that pericytes use organ-specific “vocabularies” of growth factors to steer development: in lungs, they promote air sac and vessel formation through HGF and BDNF, while in the brain they foster vessel growth and restrain inflammatory reactions through Nodal. Rather than acting as passive structural braces, pericytes act as local signaling hubs that tailor blood vessel behavior and nearby cell responses to the needs of each organ. Understanding these hidden conversations could inform future strategies to support lung maturation in premature infants, protect the brain’s barrier in disease, or fine-tune inflammation in neurological disorders without disturbing vital blood flow.
Citation: Rasouli, S.J., Kruse, K., Diéguez-Hurtado, R. et al. Pericytes are organ-specific regulators of tissue morphogenesis. Nat Commun 17, 4229 (2026). https://doi.org/10.1038/s41467-026-71643-1
Keywords: pericytes, lung development, brain vasculature, angiocrine signaling, neuroinflammation