Clear Sky Science · en
Fc-free single-chain antibody mRNA therapy for airway infection of multidrug-resistant Pseudomonas aeruginosa
Why this research matters to everyday health
Antibiotic-resistant infections are already killing more than a million people each year, and the death toll is projected to rival or exceed cancer within a few decades. One of the worst culprits is Pseudomonas aeruginosa, a hospital-dwelling bacterium that attacks the lungs of patients on ventilators, people with cystic fibrosis, and those with weakened immune systems. This study explores a fresh way to fight these dangerous infections without relying on traditional antibiotics: using messenger RNA (mRNA) to turn the body into a factory for tiny, highly targeted antibacterial antibodies.
A new way to arm the body’s defenses
Instead of infusing patients with ready-made antibodies—which are expensive and slow to produce—the researchers packaged genetic instructions for a specialized antibody fragment into tiny lipid nanoparticles, similar to those used in mRNA COVID-19 vaccines. Once injected into the bloodstream, these particles travel mainly to the liver and spleen, where cells read the mRNA and start making a single-chain antibody fragment that recognizes a key structure on P. aeruginosa. This structure, called the type III secretion system, is a microscopic needle the bacterium uses to inject toxins into lung cells. By blocking this needle, the antibody neutralizes the bacteria’s most destructive weapon rather than trying to kill the microbe outright. 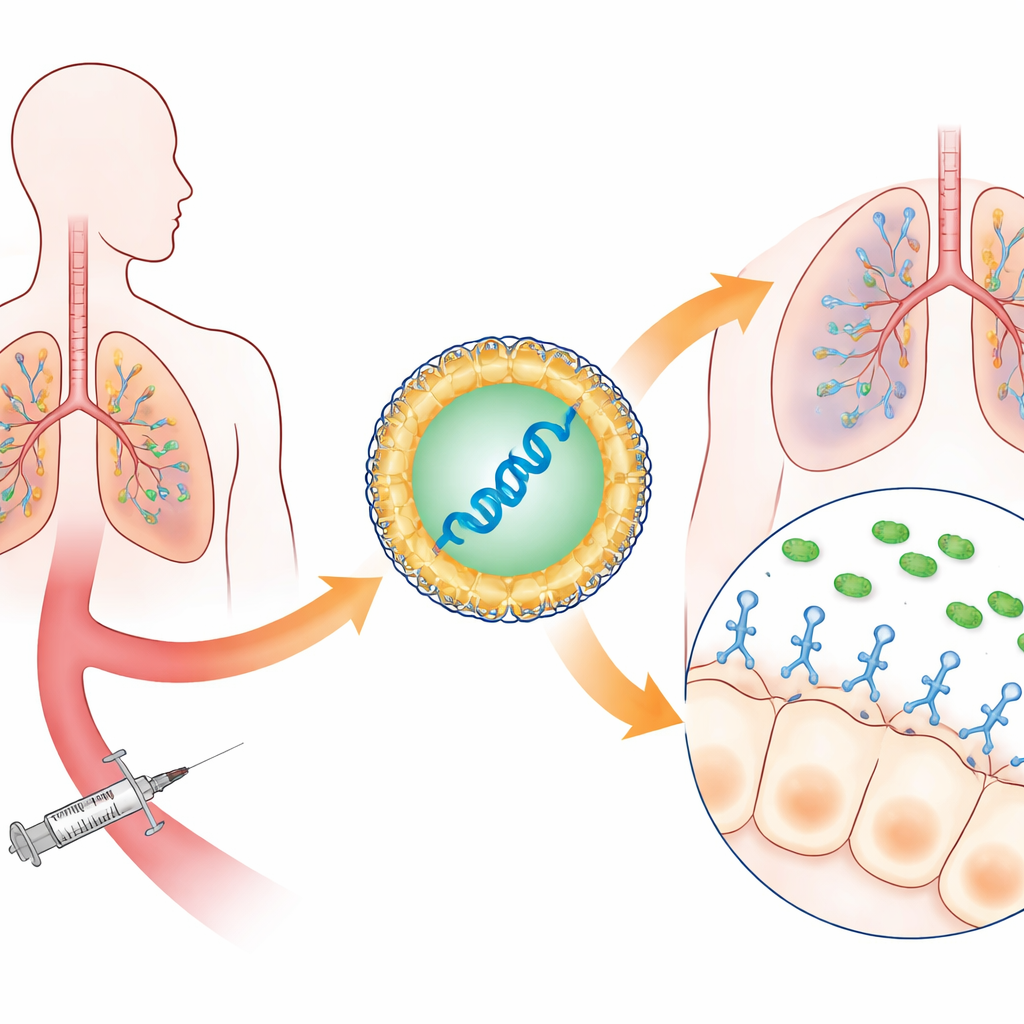
Protecting lungs before and after infection
The team tested their approach in mice exposed to lethal doses of P. aeruginosa delivered directly into the airways. When the mRNA therapy was given shortly before infection, mice that received the anti-bacterial antibody instructions survived almost completely, while most untreated animals died within a day. The treated mice had less lung swelling, fewer bacteria in their lungs and blood, and dramatically lower levels of inflammatory molecules linked to acute lung injury. The therapy also worked when given after infection had already begun, a more realistic stand-in for treating hospitalized patients. In these treatment-style experiments, survival rates with the mRNA-produced antibody matched or surpassed those achieved with the best available antibody protein therapy.
Outperforming antibiotics against tough hospital strains
P. aeruginosa found in real hospitals are often far more diverse and drug-resistant than standard laboratory strains. To mimic this clinical reality, the researchers collected ten resistant strains from patients and tested them, along with two lab strains, in mice whose immune systems had been deliberately weakened. Many of these isolates carried a particularly destructive toxin gene called exoU and resisted multiple antibiotic classes, including carbapenems, aminoglycosides, and fluoroquinolones. Even in this harsh setting—and at bacterial doses that reliably killed control animals—the mRNA-produced antibody substantially improved survival, especially against the most toxic, multidrug-resistant strains. In some cases, it matched or outperformed colistin, a last-resort antibiotic with significant side effects. When combined with colistin, the mRNA therapy rescued all treated mice, suggesting that such biological drugs could extend the useful life of existing antibiotics rather than replace them outright.
Small, Fc-free antibodies reach the infected surface better
A key innovation in this work is the use of an “Fc-free” single-chain antibody fragment, which contains only the business end that recognizes the bacterial target and lacks the bulky tail that full antibodies use to engage immune cells. Conventional wisdom favors keeping the Fc tail because it prolongs circulation time in the blood. Here, the researchers directly compared mRNA encoding the small fragment alone with a larger version fused to an Fc domain. They found that while the Fc-bearing form stayed longer in the bloodstream and organs, the smaller fragment crossed more efficiently from blood into the thin fluid layer coating the lung’s air sacs—the actual battleground where the bacteria attack. Measurements of fluid washed from the airways showed several-fold higher levels of the small fragment than of the Fc-fused antibody, even though its blood levels were lower. This superior access to the infection site likely explains why the Fc-free version gave better survival in multiple treatment models. 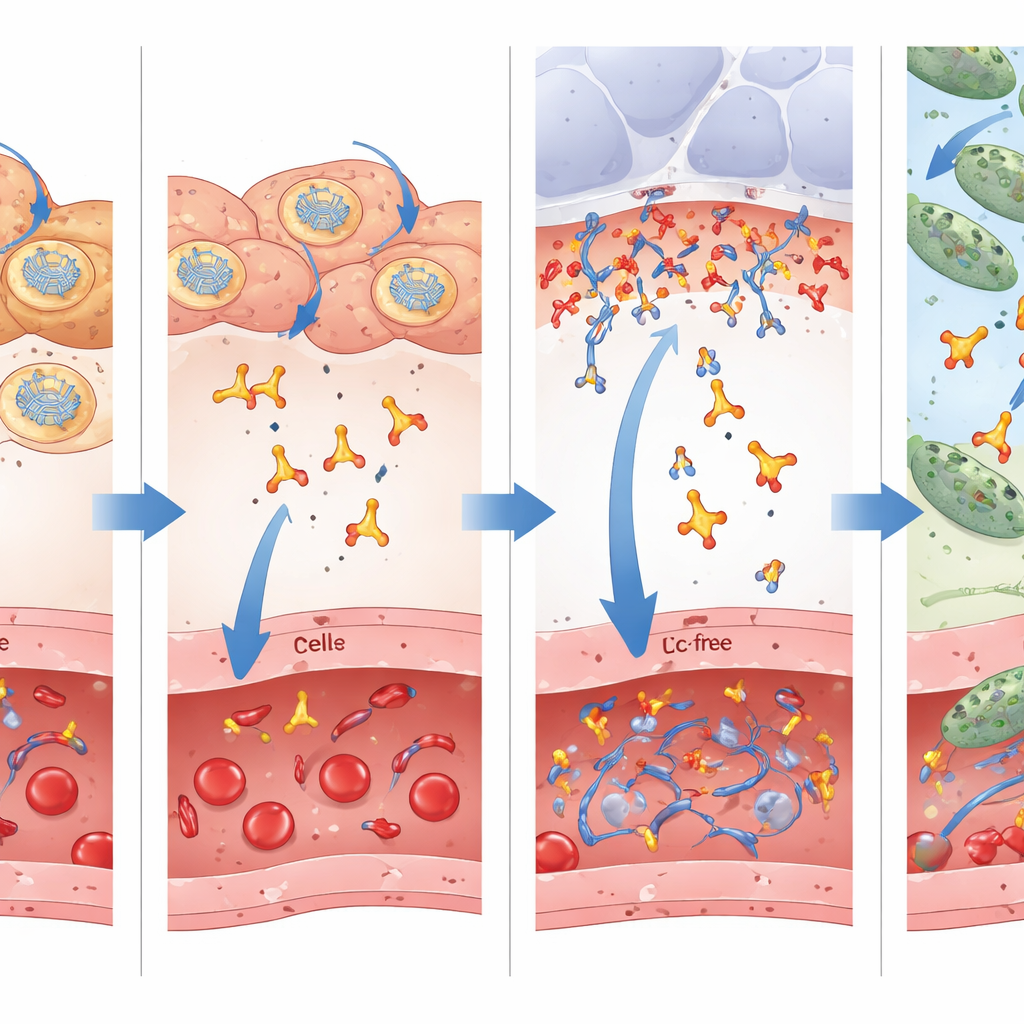
A versatile platform for future infections
In simple terms, this study shows that a single injection of mRNA can quickly flood vulnerable lungs with precisely targeted, small antibody fragments that blunt the most damaging tricks of a deadly, drug-resistant bacterium. The treatment reduced inflammation, limited bacterial spread, and saved animals even when their immune systems were suppressed and the infecting strains resisted standard antibiotics. Because mRNA manufacturing is rapid and modular, the same strategy could in principle be adapted to other dangerous bacteria that use similar toxin-delivery systems. While further work is needed to refine dosing, safety, and delivery for humans, these results point to a future in which doctors can rapidly “upload” new defensive molecules into patients to outpace evolving microbes, offering a powerful new tool against the growing crisis of antimicrobial resistance.
Citation: Kinoshita, M., Kawaguchi, K., Mochida, Y. et al. Fc-free single-chain antibody mRNA therapy for airway infection of multidrug-resistant Pseudomonas aeruginosa. Nat Commun 17, 2960 (2026). https://doi.org/10.1038/s41467-026-71040-8
Keywords: antimicrobial resistance, mRNA therapeutics, Pseudomonas aeruginosa, antibody fragments, lung infection