Clear Sky Science · en
Reactivating exhausted tumor-infiltrating T cells by a bispecific DC-T cell engager in mice
Turning Tired Immune Cells Back On
Cancer often wins not because our immune system fails to see it, but because tumor-fighting cells become worn out and stop working well. This study in mice describes a clever new protein drug that physically links two key immune cells inside tumors so they can talk to each other again. By doing so, it reawakens “exhausted” killer T cells, shrinks tumors, and even leaves behind a memory of the cancer, potentially preventing it from coming back.
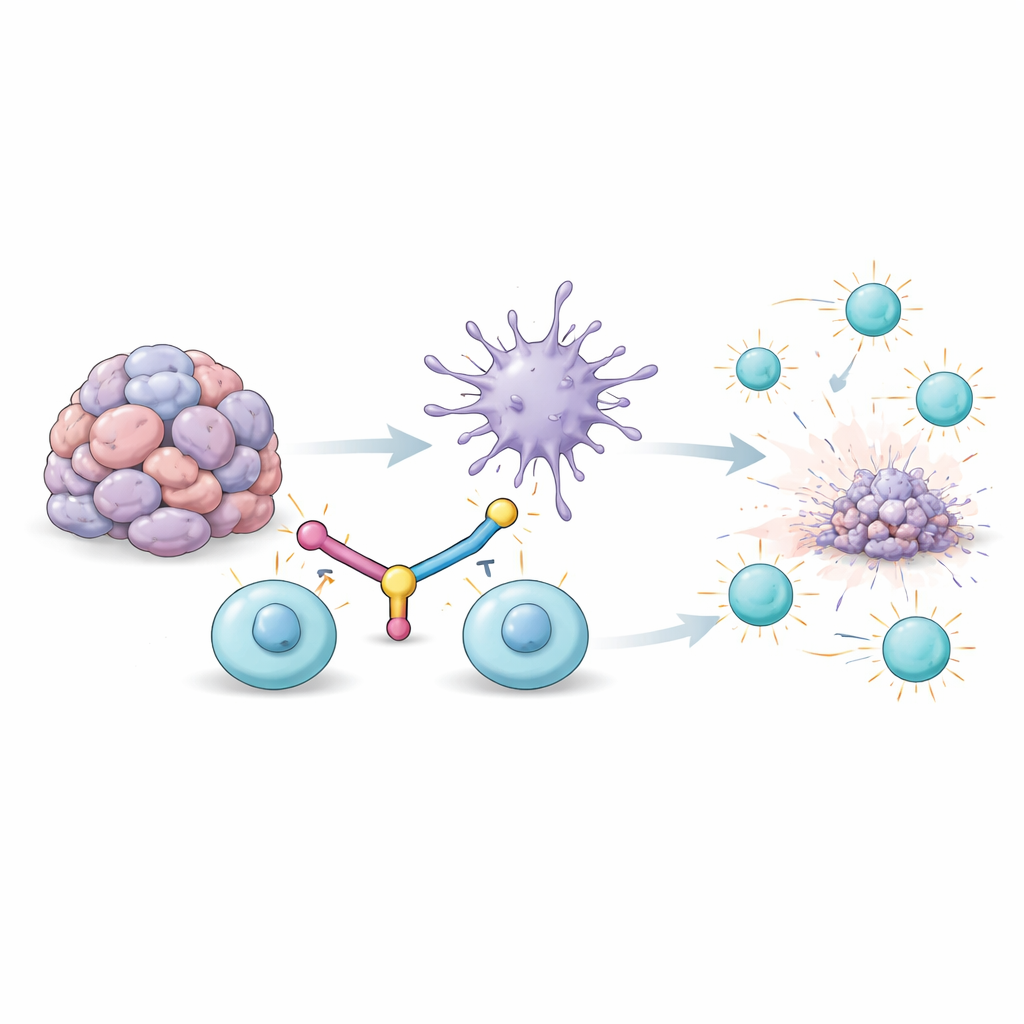
Why Tumor-Fighting Cells Run Out of Steam
Deep inside tumors, special white blood cells called T cells recognize and attack cancer. But when they are exposed to tumor signals for too long, they enter an exhausted state: they stop multiplying, make fewer attack molecules, and may even die off. One surface protein linked to this exhaustion is called TIM3. At the same time, another immune cell type, dendritic cells, is supposed to coach T cells by showing them tumor fragments and giving them strong “go” signals. Inside tumors, however, dendritic cells are often weakened by the tumor environment and receive too little of a powerful immune messenger called type I interferon. The result is a double failure: T cells are exhausted, and their main coaches cannot properly wake them up.
A Bridging Molecule to Reconnect Immune Allies
The researchers designed a single fusion protein, called a bispecific DC–T cell engager (BiDT), to fix both problems at once. One end of BiDT is an antibody that recognizes TIM3 on exhausted T cells. The other end is a modified version of interferon-alpha that strongly activates dendritic cells through their interferon receptor but is tuned to reduce unwanted effects elsewhere in the body. When BiDT is given to tumor-bearing mice, it accumulates inside tumors, where TIM3 is abundant on T cells. There, it literally bridges dendritic cells and exhausted T cells, forcing them into close contact while simultaneously switching dendritic cells into a more active, stimulatory state.
Reawakened Killers and Lasting Immune Memory
Once this bridge is in place, dendritic cells boost key “co-stimulatory” signals on their surface and help T cells turn back on survival and growth pathways. Exhausted T cells begin to look and behave more like effective fighters: they express more survival protein (so they are less likely to die), regain production of important molecules such as IL-2 and interferon-gamma, and expand in number. In mouse colon cancer and melanoma models, BiDT treatment often cleared established tumors and improved survival while causing less weight loss and systemic toxicity than giving interferon and TIM3 antibody separately. Remarkably, mice cured with BiDT resisted later tumor re-challenge, and their immune cells could transfer protection to other animals, showing that robust, tumor-specific memory had formed.
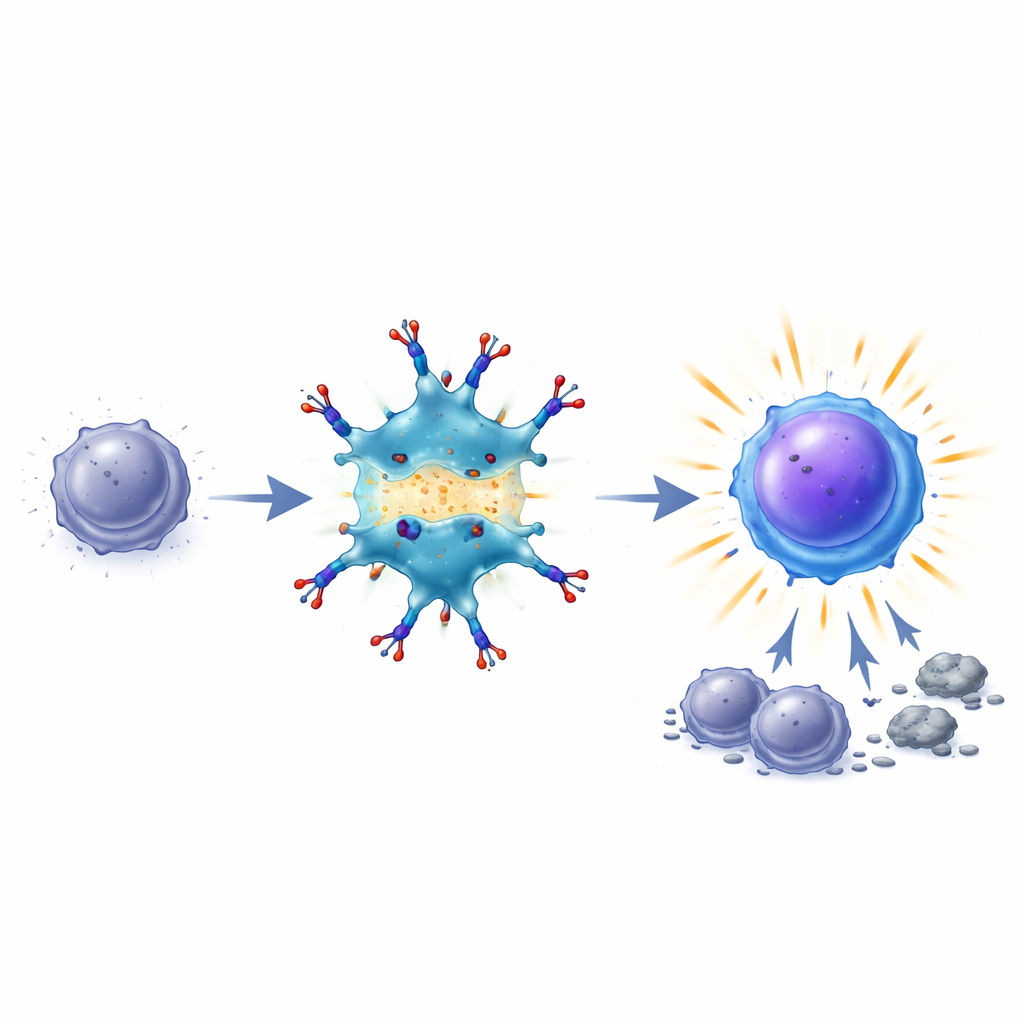
Working Inside the Tumor, Not Just in Lymph Nodes
Careful genetic and depletion experiments revealed that BiDT’s success depends on pre-existing killer T cells and dendritic cells already inside the tumor, rather than on fresh T cells arriving from lymph nodes. The interferon signal needs to act on dendritic cells, while engagement of TIM3 on T cells is crucial for their rescue. The reawakened T cells respond strongly to IL-2 signals and produce bursts of interferon-gamma, which in turn helps destroy tumor cells. In a “cold” melanoma model that normally resists standard checkpoint drugs, BiDT partly overcame this resistance. When combined with a PD-L1 blocking antibody, which prevents a separate brake on T cells, the therapy eradicated tumors in a substantial fraction of mice and generated long-lasting protection.
Safer Versions and Future Hopes
Because interferon-based treatments can be quite toxic in patients, the team also created a “prodrug” version, Pro-BiDT, in which the interferon part is kept inactive until it is cut free by enzymes that are abundant in tumors but scarce in healthy tissues. In mice, Pro-BiDT preserved the powerful antitumor effects of the original BiDT while greatly reducing signs of inflammation and blood toxicity. Together, these findings suggest a new way to treat cancer: rather than only releasing brakes on T cells from the outside, precisely reconnect and energize immune circuits within the tumor itself. If similar results can be achieved in humans, such drugs could help rescue tired immune responses, overcome resistance to current immunotherapies, and provide longer-lasting protection against cancer.
Citation: Zhang, X., Gao, Y., Hu, W. et al. Reactivating exhausted tumor-infiltrating T cells by a bispecific DC-T cell engager in mice. Nat Commun 17, 4081 (2026). https://doi.org/10.1038/s41467-026-70876-4
Keywords: cancer immunotherapy, T cell exhaustion, dendritic cells, bispecific antibodies, interferon-alpha