Clear Sky Science · en
Carbene-catalyzed double esterification enables enantioselective conformational self-locking of pillar[5]arenes
Shaping tiny rings for smart materials
Chemists are learning to sculpt molecules almost like watchmakers shape gears. This study shows how to “lock” a small ring-shaped molecule into one hand‑like form, using a simple catalyst and common ingredients. Because such rings can cradle other molecules, this new control over shape could influence future sensors, materials and medical tools.
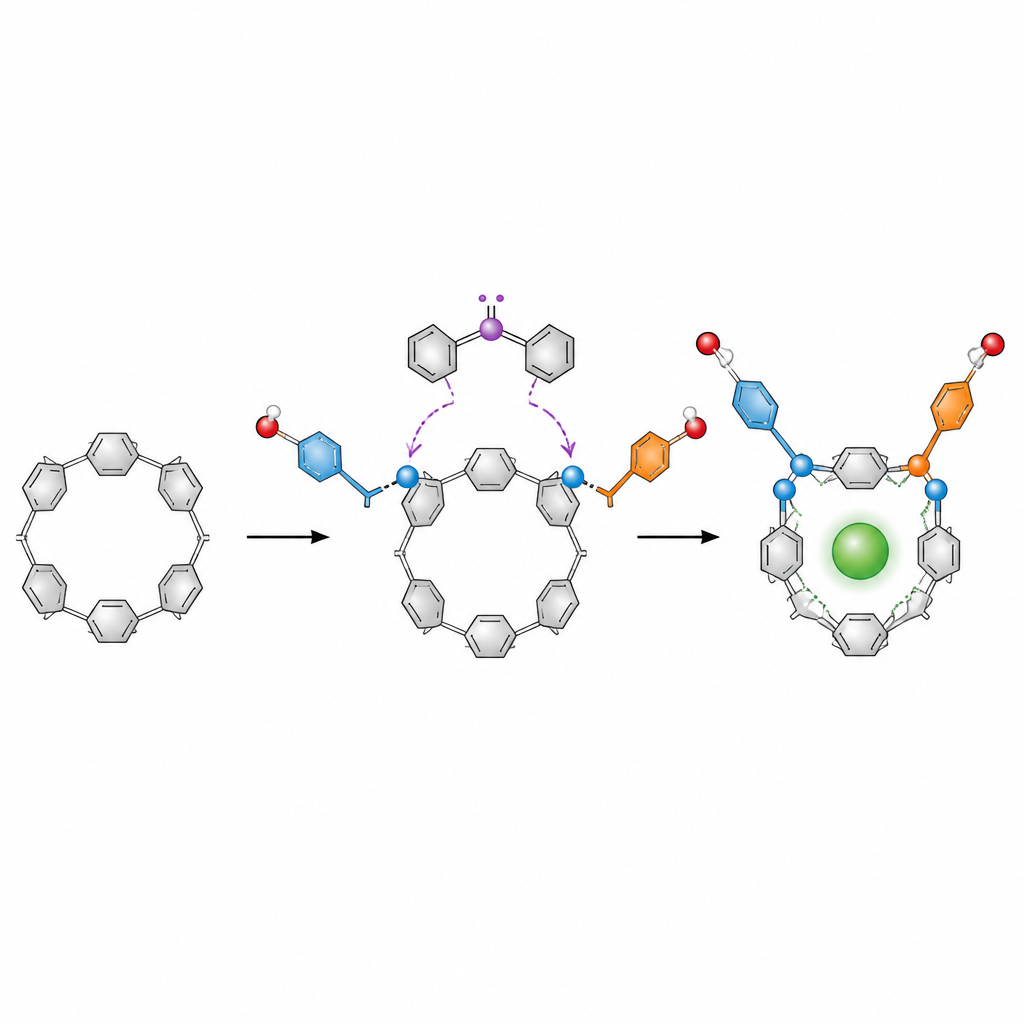
Why the shape of a ring matters
The stars of the story are pillar[5]arenes, doughnut‑like rings built from five repeating aromatic units. These rings are popular in host–guest chemistry, where they act as tiny containers for ions, drugs or dyes. On paper, a pillar[5]arene can twist into several mirror‑image forms that swap back and forth. To use them as precise tools, chemists need to freeze one “handed” form in place and enrich one mirror image over the other, but existing methods often rely on metals, multiple steps and tedious separation of mixtures.
A metal‑free shortcut to locked rings
The authors designed a much simpler approach based on organocatalysis, which uses small organic molecules instead of metals to steer reactions. They started from a diformyl pillar[5]arene, a version of the ring with two reactive aldehyde groups. In the presence of a chiral carbene catalyst, an oxidant and a base, these groups undergo a double esterification with aromatic alcohols such as naphthols. Each reaction attaches an ester “arm” to the ring. Once two bulky arms are in place, the ring can no longer twist freely, so one chiral form becomes locked and strongly favored.
Tuning the reaction’s reach
Through systematic testing, the team discovered conditions that give both high yields and strong preference for one mirror image. They examined how the nature of the catalyst, solvent, base and oxidant influences the outcome, and found that sodium acetate in dichloromethane at moderate temperature works particularly well. The process tolerates many different aromatic partners, including naphthols with electron‑rich or electron‑poor groups and a range of phenols. Using a clever steric analysis, they showed that only substituents long and bulky enough truly prevent the ring from flipping, which explains why small groups like methyl do not deliver stable chiral products.
From simple rings to complex guests
The method reaches beyond simple building blocks. The researchers successfully attached fragments from natural and bioactive molecules, such as estrone and the cholesterol‑lowering drug ezetimibe, while preserving high “handedness” control. They further demonstrated that the locked rings withstand heat and chemical modification without losing their chiral purity. On gram scale, the key product forms in almost quantitative yield. Additional reactions on remaining functional groups create more complex structures, including a rotaxane, in which a dumbbell‑shaped molecule is threaded through the pillar[5]arene ring and trapped by bulky stoppers.
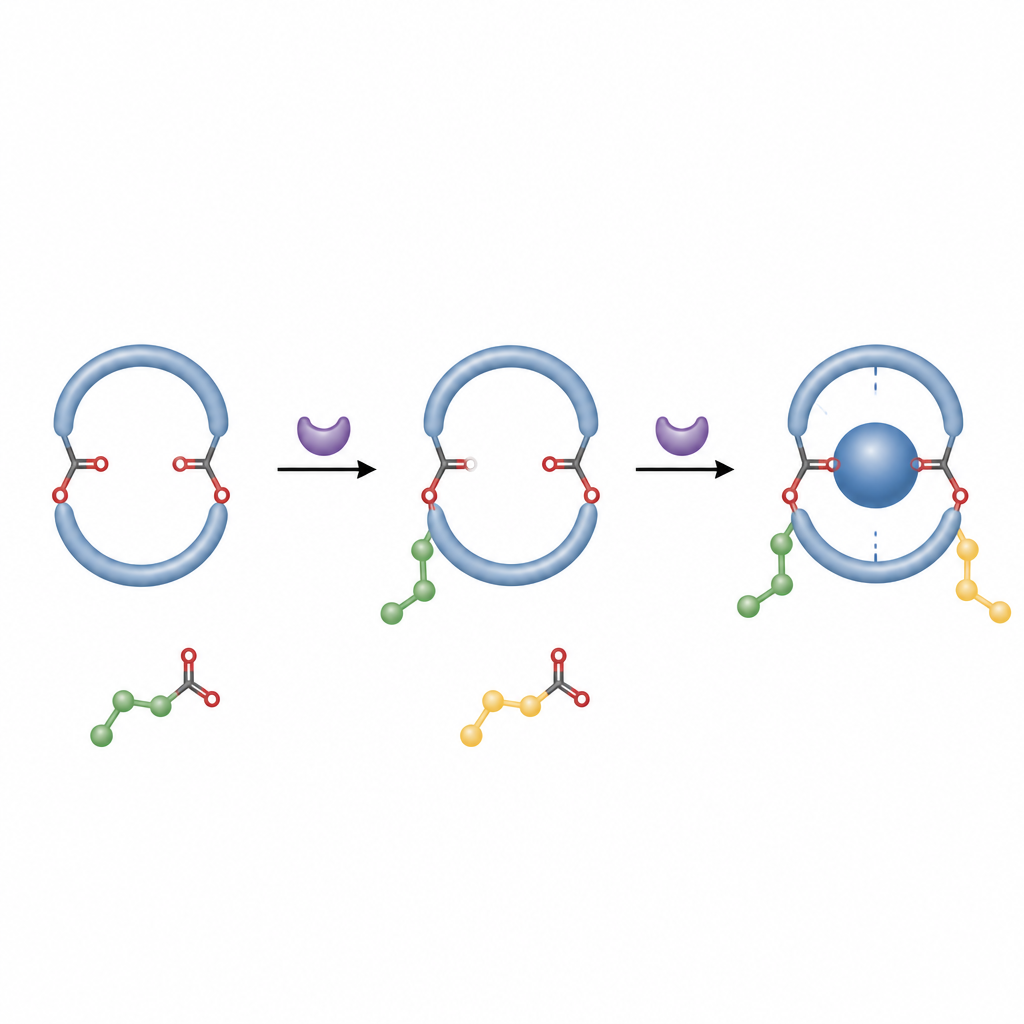
What this means for future molecular machines
In everyday terms, the authors have found a way to snap a molecular ring into a fixed left‑ or right‑handed pose using a simple, metal‑free process. This makes it much easier to prepare pure, shape‑defined hosts that can grip other molecules in predictable ways. Such control over handedness and stability is important for designing responsive materials, chiral sensors and molecular devices, and this straightforward double esterification offers a practical new route to those advanced structures.
Citation: Dočekal, V., Hladík, O., Lóška, L. et al. Carbene-catalyzed double esterification enables enantioselective conformational self-locking of pillar[5]arenes. Nat Commun 17, 4253 (2026). https://doi.org/10.1038/s41467-026-70809-1
Keywords: pillararenes, organocatalysis, chiral hosts, esterification, supramolecular chemistry