Clear Sky Science · en
Paired electrolysis enables para-C–H amination of phenols with nitroarenes and mechanistic visualization via multifunctional electrochemical mass spectrometry
Turning Simple Rings into Useful Molecules
Chemists rely on carbon–nitrogen bonds to build many of the medicines, dyes, and materials we use every day. Traditionally, stitching these atoms together requires precious metals, multiple reaction steps, and harsh chemicals. This study introduces a gentler, electricity-powered way to connect two very common building blocks—phenols and nitro compounds—into valuable products used in dyes, pesticides, and fuel additives. It also showcases a kind of “live video feed” of the reaction at the molecular level, captured by a specialized mass spectrometry setup.
Why This New Bond-Building Trick Matters
The target molecules in this work, called p-hydroxy diphenylamines, sit at the heart of many functional products, from colorants to antioxidants in fuels. Conventional routes to them usually start from an already reduced form of nitro compounds and often require protecting groups, extra reagents, and metal catalysts. Each added step increases cost, waste, and energy use. The authors set out to find a direct way to join a simple nitro compound with a phenol ring in one shot, ideally without expensive metals or external oxidizing and reducing agents.
Using Electricity as the Invisible Reagent
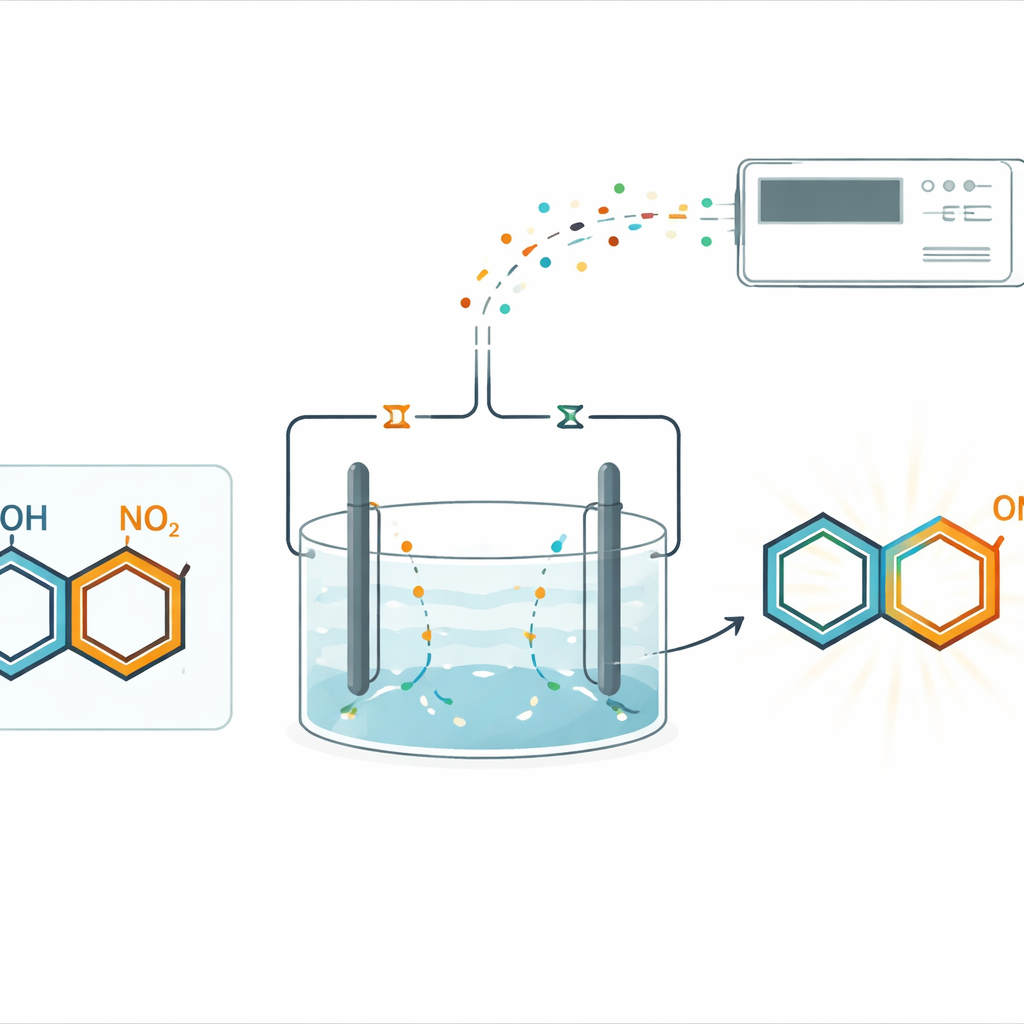
Instead of relying on bottled chemicals to add or remove electrons, the team used paired electrolysis in a single, undivided cell. In this setup, electrical current drives two complementary changes at once: reduction at one electrode and oxidation at the other. Nitro-containing rings are gently reduced on the cathode side, while phenols are activated on the anode side. Under carefully chosen conditions, the two activated partners meet and form a new carbon–nitrogen bond at a specific position on the phenol ring, known as the para site. This approach avoids metal catalysts and external reducing agents, yet still delivers the desired p-hydroxy diphenylamines in good yields.
Probing What Happens Between Start and Finish
To understand and optimize this reaction, the researchers built an automated electrochemical mass spectrometry (AIEC-MS) platform. Reaction mixtures containing only tiny amounts of material are fed through a miniature flow cell, where they experience an applied voltage and are then sprayed directly into a mass spectrometer. This allows the team to rapidly test many combinations of nitro compounds and phenols, while also capturing short-lived intermediates that appear only during electrolysis. With this tool, they showed that a wide variety of nitroarenes—bearing halogens, electron-withdrawing groups, deuterium labels, and even multiple nitro groups—as well as many substituted phenols, all participate effectively in the new bond-forming process.

Watching a Reactive “Ghost” Do the Work
The real power of the method lies in its ability to make visible the usually unseen steps of the reaction. Using high-resolution and tandem mass spectrometry, the authors detected a sequence of fleeting species formed as the nitro compound travels down the electrochemical “ladder” of reduction. Among them is a highly reactive aryl nitrene–like intermediate, a nitrogen-containing fragment that can insert directly into the carbon framework of the activated phenol ring. Additional experiments—including isotope labeling, kinetic measurements, voltage control, and trapping reactions with other partners—support a stepwise picture: nitro compounds are reduced through nitroso and related species to a nitrene; phenols are oxidized to more reactive forms; the nitrene inserts into the para C–H bond of the phenol-derived partner; and a final set of electron transfers restores aromatic stability to yield the product.
From Fundamental Insight to Future Applications
By combining a mild, electricity-driven reaction with a powerful in situ analytical platform, this work achieves both practical synthesis and a clear view of the underlying mechanism. In everyday terms, the team has devised a cleaner “one-plug” method to assemble useful nitrogen-containing molecules from simple, readily available rings, while simultaneously filming the molecular choreography as it unfolds. Their paired electrolysis strategy demonstrates that direct use of nitro compounds, guided by real-time mass spectrometry, can broaden what is possible in organic synthesis. Looking ahead, similar approaches may help chemists design more efficient, selective, and sustainable routes to a host of complex molecules important to medicine, agriculture, and materials science.
Citation: Peng, T., Chen, Z., Cui, X. et al. Paired electrolysis enables para-C–H amination of phenols with nitroarenes and mechanistic visualization via multifunctional electrochemical mass spectrometry. Nat Commun 17, 4143 (2026). https://doi.org/10.1038/s41467-026-70691-x
Keywords: electroorganic synthesis, C–N bond formation, paired electrolysis, mass spectrometry, nitroarene amination