Clear Sky Science · en
Reversible DNA condensation drives natural transformation
How Bacteria Borrow DNA
Antibiotic resistance can spread when bacteria pick up loose DNA from their surroundings, a trick known as natural transformation. This study reveals how a single protein helps pull incoming DNA across the tough outer barrier of certain bacteria, using a subtle push–pull routine rather than a powerful motor. Understanding this microscopic tug-of-war could inform future strategies to slow the spread of resistance genes.
A Molecular Doorway for New Genes
Many bacteria can naturally take up fragments of DNA from their environment and add useful genes to their own chromosomes. To do this, the DNA must cross the cell wall and reach the interior of the cell. In Gram-positive bacteria, which have a thick cell wall, a membrane protein called ComEA sits in the space between the wall and the inner membrane, where it grabs onto incoming DNA. Until now, scientists knew ComEA was essential for this process, but they did not understand how it actually helped move DNA inward.
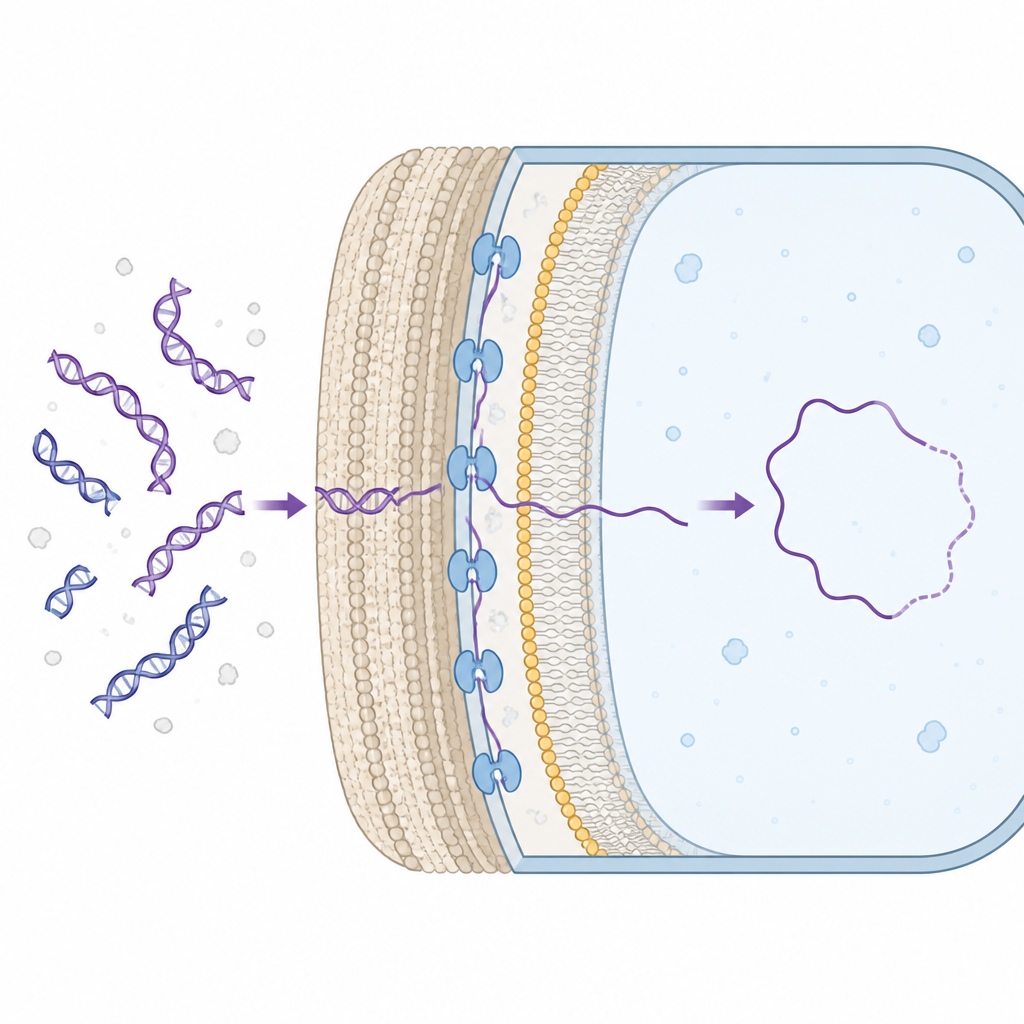
Gentle DNA Tugs Measured One Molecule at a Time
The researchers used single-molecule optical tweezers, a technique that holds a single piece of DNA between two tiny beads using laser light. By adding purified ComEA and pulling on the DNA, they could see when ComEA molecules linked distant parts of the strand together, forming small loops. As more of these bridges formed, the overall length of DNA effectively shrank, showing that ComEA was condensing it. Breaking these bridges caused sudden jumps in DNA length, allowing the team to calculate how much DNA had been looped and how much mechanical force was generated. They found that ComEA can pull with very small, sub-piconewton forces, enough to bias DNA movement inward but far weaker than the forces produced by classical molecular motors.
A Two-Mode Switch: Condense, Then Relax
Electron microscopy gave the scientists snapshots of what the DNA looked like when mixed with ComEA. At modest protein levels, they saw loops and partial compaction of the DNA, consistent with bridging interactions. As the concentration of ComEA increased further, these loops disappeared, even though the DNA surface became more thickly coated with protein. Additional measurements showed that at high coverage, ComEA still forms assemblies along DNA but now in a non-bridging manner that stabilizes stretched DNA rather than looping it. In this crowded state, the DNA becomes decondensed and no longer experiences pulling forces, revealing a built-in switch controlled by how much ComEA is present locally.
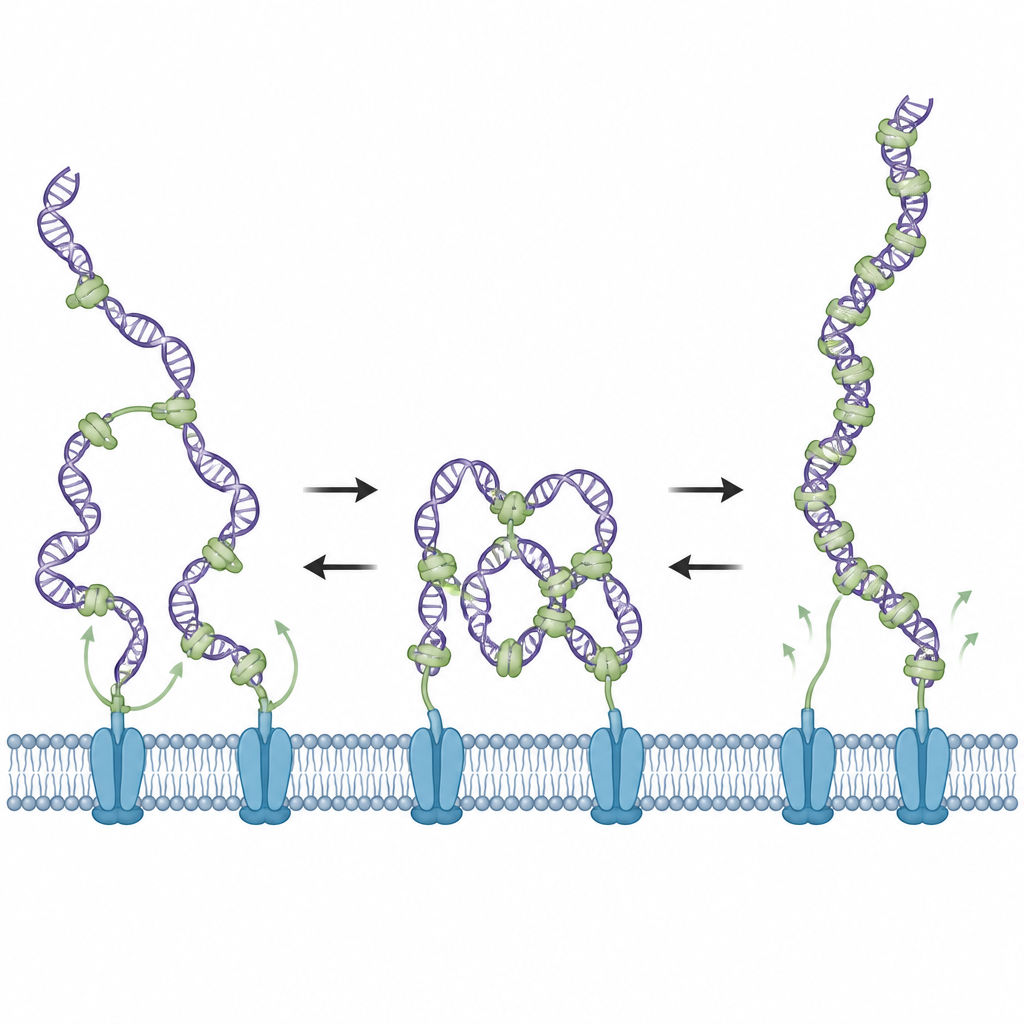
Engineering the Protein’s Flexible Link
ComEA contains a flexible linker that connects its DNA-gripping portion to the part that allows it to cluster with other ComEA molecules. The team shortened or lengthened this linker to test how it affected behavior. Short-linker mutants favored the relaxed, non-bridging state and failed to generate condensation forces, while long-linker mutants favored persistent bridging and force generation even at high protein levels. Both types of mutants bound DNA, but they were each poor at transforming real Bacillus subtilis cells, showing that successful DNA uptake requires ComEA to be able to both condense DNA at first and later let it relax.
Why This Matters for Antibiotic Resistance
Taken together, the results support a two-step model for how Gram-positive bacteria pull in foreign DNA. Early in uptake, sparsely bound ComEA proteins bridge separate stretches of DNA, gently condensing and drawing the strand through the thick cell wall toward the membrane. As more ComEA accumulates on the same DNA, it switches into a non-bridging mode that stops pulling and allows the DNA to decondense, making it easier for other transport proteins to feed a single DNA strand into the cell’s interior. This reversible condensation provides just enough directional bias to move DNA without fighting the later transport step, helping explain how bacteria efficiently acquire new genes that can include antibiotic resistance.
Citation: Santiago, J.I., Ahmed, I., Hahn, J. et al. Reversible DNA condensation drives natural transformation. Nat Commun 17, 4242 (2026). https://doi.org/10.1038/s41467-026-70559-0
Keywords: natural transformation, DNA uptake, ComEA, antibiotic resistance, bacterial cell wall