Clear Sky Science · en
Single-cell spatial map of cis-regulatory elements for disease-related genes in the macaque cortex
Why this brain map matters
Many brain diseases, from schizophrenia to Alzheimer’s, run in families—but the DNA changes that raise risk often sit in mysterious stretches of the genome that do not code for proteins. This study tackles that puzzle by creating a detailed map of how millions of tiny DNA switches operate in different cell types and locations across the cortex of macaque monkeys, our close primate relatives. By linking these switches to both cell identity and human disease risk, the work offers a new window into why certain brain cells and regions are especially vulnerable in psychiatric and neurodegenerative disorders.
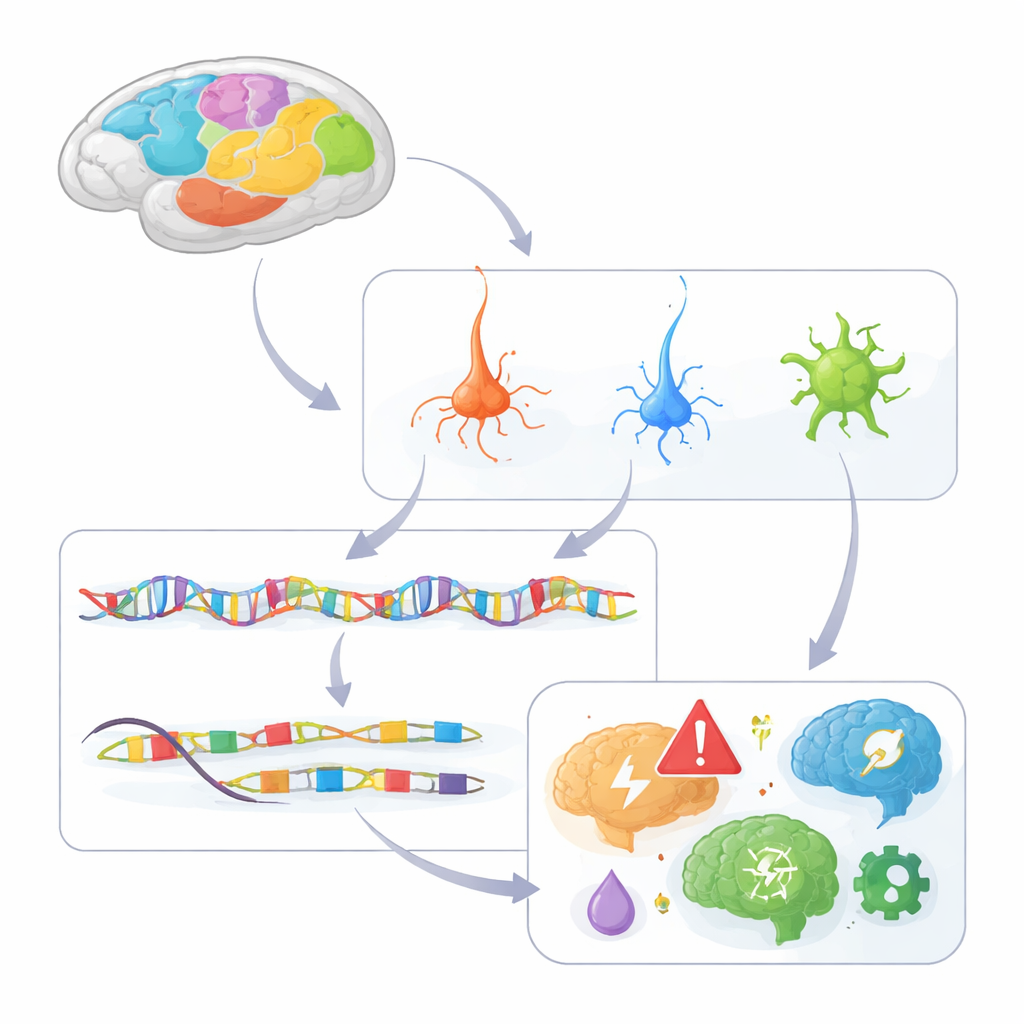
Building a cell-level atlas of the primate cortex
The researchers analyzed chromatin accessibility—how open or closed DNA is—in about 1.6 million individual nuclei taken from 142 regions across the macaque cortex. Open stretches of DNA mark cis-regulatory elements, the “on–off” switches that control nearby genes. Using a droplet-based method called single-nucleus ATAC-seq, they grouped cells into 230 distinct types, spanning excitatory glutamatergic neurons, inhibitory GABAergic neurons, and various support cells such as astrocytes, oligodendrocytes, and microglia. They identified more than 600,000 candidate regulatory elements and linked many of them to specific target genes, building a first draft of the cortex’s regulatory wiring diagram.
Layered and regional patterns of gene control
By combining this DNA-switch atlas with a high-resolution spatial map of gene activity, the team could see where, within the six cortical layers and multiple lobes, different regulatory elements were active. Many switches showed strong layer and region preferences. For example, elements tied to genes that mark upper-layer neurons were active mainly in layers 2 and 3, while others were confined to deeper layers. Certain switches were highly active only in the primary visual cortex (V1), helping explain why some neuron types and gene programs are unique to this region. The study also revealed contrasts between the evolutionarily older piriform cortex and the neocortex, with different sets of switches active in glial cells versus deep-layer neurons, pointing to distinct regulatory programs that shape these architectures.
Primate innovations in key neuron types
Comparing macaque data with similar maps from human and mouse cortex, the authors found neuron subclasses that exist only in primates, especially certain layer-4 glutamatergic neurons and a specialized group of inhibitory cells called LAMP5/LHX6 interneurons. These cells carried many regulatory elements that are open in humans and macaques but not mice, and some of these elements appear to have arisen alongside transposable elements—mobile DNA sequences that can help create new gene switches. The primate-only switches regulate genes involved in synaptic function, neurodevelopment, and neuron communication, suggesting that relatively recent regulatory changes helped diversify cortical circuits unique to primates, particularly in layers linked to complex perception and cognition.
Support cells and the wiring behind plasticity
The map also sheds light on non-neuronal cells. Oligodendrocyte precursor cells and mature oligodendrocytes showed opposing abundance across cortical hierarchy: higher-level regions had more precursors and fewer mature myelinating cells, hinting at greater flexibility in adjusting wiring there. Their regulatory elements tracked this gradient and were enriched in primate-biased switches tied to synapse and visual system genes. Astrocytes split into interlaminar cells concentrated in the top layer and protoplasmic cells distributed through deeper layers, each controlled by distinct sets of switches and transcription factors. These patterns suggest that not only neurons, but also the cells that nourish, insulate, and remodel them, are tuned by region-specific regulatory programs that may influence learning capacity and vulnerability to damage.
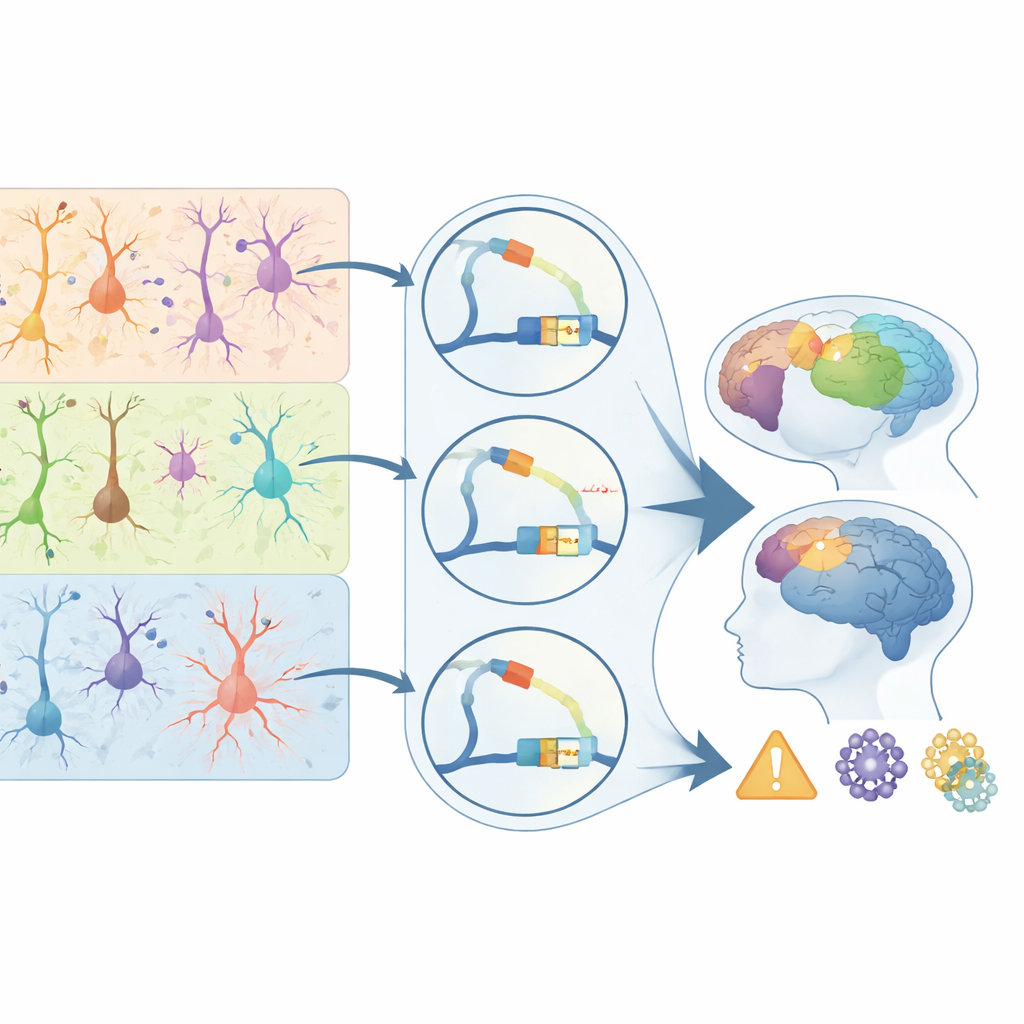
Connecting DNA switches to brain disease risk
To bridge their primate atlas to human health, the authors overlaid it with genetic studies of 28 brain-related disorders and traits. They found that disease-linked variants fall disproportionately into regulatory elements active in specific cell types. Schizophrenia and bipolar disorder risk variants cluster in switches used by upper-layer excitatory neurons; epilepsy signals are enriched in certain inhibitory cells; and Alzheimer’s risk variants concentrate strikingly in microglial regulatory elements. Many of these disease-associated switches are primate-biased, especially in layer-4 neurons for schizophrenia and in microglia for Alzheimer’s, and they target genes known to influence synapses, immune responses, and protein clearance. For a general reader, the key message is that disease risk often acts not by breaking proteins directly, but by subtly rewiring when and where genes turn on in particular cells. This study delivers a landmark map of those control points in the primate cortex, clarifying why some cell types and regions are disease hotspots and offering a guide for future therapies that aim to correct the brain’s regulatory code rather than its protein parts.
Citation: Meng, J., Chen, C., Zhu, Z. et al. Single-cell spatial map of cis-regulatory elements for disease-related genes in the macaque cortex. Nat Commun 17, 4041 (2026). https://doi.org/10.1038/s41467-026-70497-x
Keywords: single-cell epigenomics, primate cortex, gene regulation, brain disorders, cis-regulatory elements