Clear Sky Science · en
A hierarchical theranostic nanoagent for multimodal imaging and targeted foam cell intervention in atherosclerosis
Why clogged arteries matter
Atherosclerosis—the gradual clogging and weakening of our arteries—is the hidden engine behind heart attacks and strokes. Deep inside artery walls, immune cells swell with fat and turn into so‑called foam cells, which help plaques grow and become fragile. Doctors can see big, late‑stage plaques with current scanners, but they struggle to spot and treat dangerous plaques early, when intervention could prevent catastrophe. This study introduces a smart, layered nanoparticle that can both light up these risky areas with multiple imaging techniques and directly calm and repair them in diseased arteries of mice.
Seeing trouble in arteries from several angles
The researchers first designed a special dye molecule that acts like a three‑in‑one beacon. It glows in a deep near‑infrared range of light, generates ultrasound signals when pulsed with a laser (photoacoustic imaging), and enhances magnetic resonance imaging (MRI). Each of these methods sees the body in a different way: the infrared glow is very sensitive and fast, photoacoustics provides sharp structural detail a few centimeters deep, and MRI maps anatomy throughout the body without radiation. By carefully tuning the dye’s structure and attaching a gadolinium unit used in MRI, the team created a single molecule that gives strong signals in all three systems, remaining stable even in harsh biological conditions.
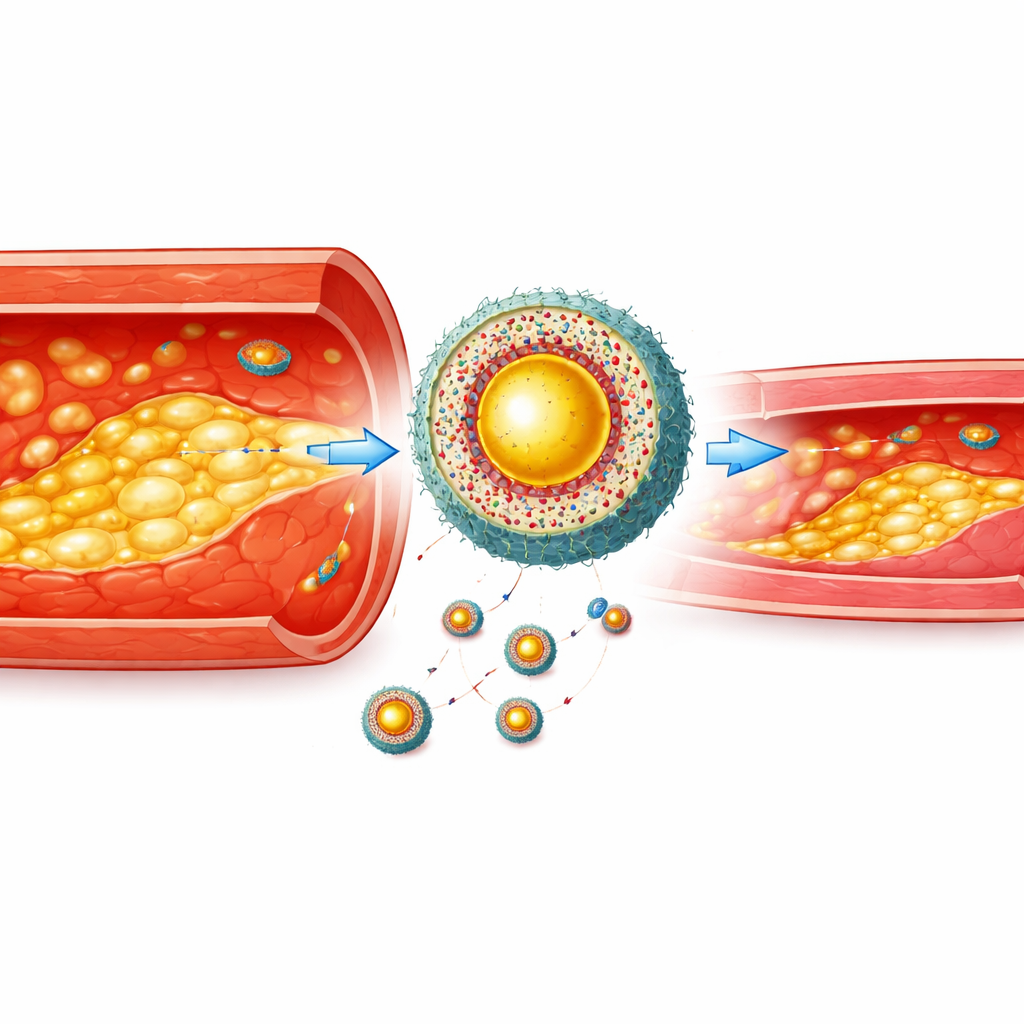
Building a homing capsule for diseased cells
On its own, a bright imaging beacon is not enough—it must reach the right place and stay there. The scientists embedded the multimodal dye and the cholesterol‑lowering drug atorvastatin inside a polymer shell that falls apart in the presence of high levels of reactive oxygen species, a chemical hallmark of inflamed foam cells. To help the particles slip through inflamed vessel linings and home in on foam cells, they wrapped the particles in real macrophage (immune cell) membranes and added a short peptide that binds to a “eat me” signal exposed on foam cell surfaces. This created a hierarchical nanoagent: first drawn to inflamed arterial walls, then more precisely to foam cells buried in the plaque, and finally activated by the stressed chemical environment inside those cells.
Testing safety, precision, and impact in cells
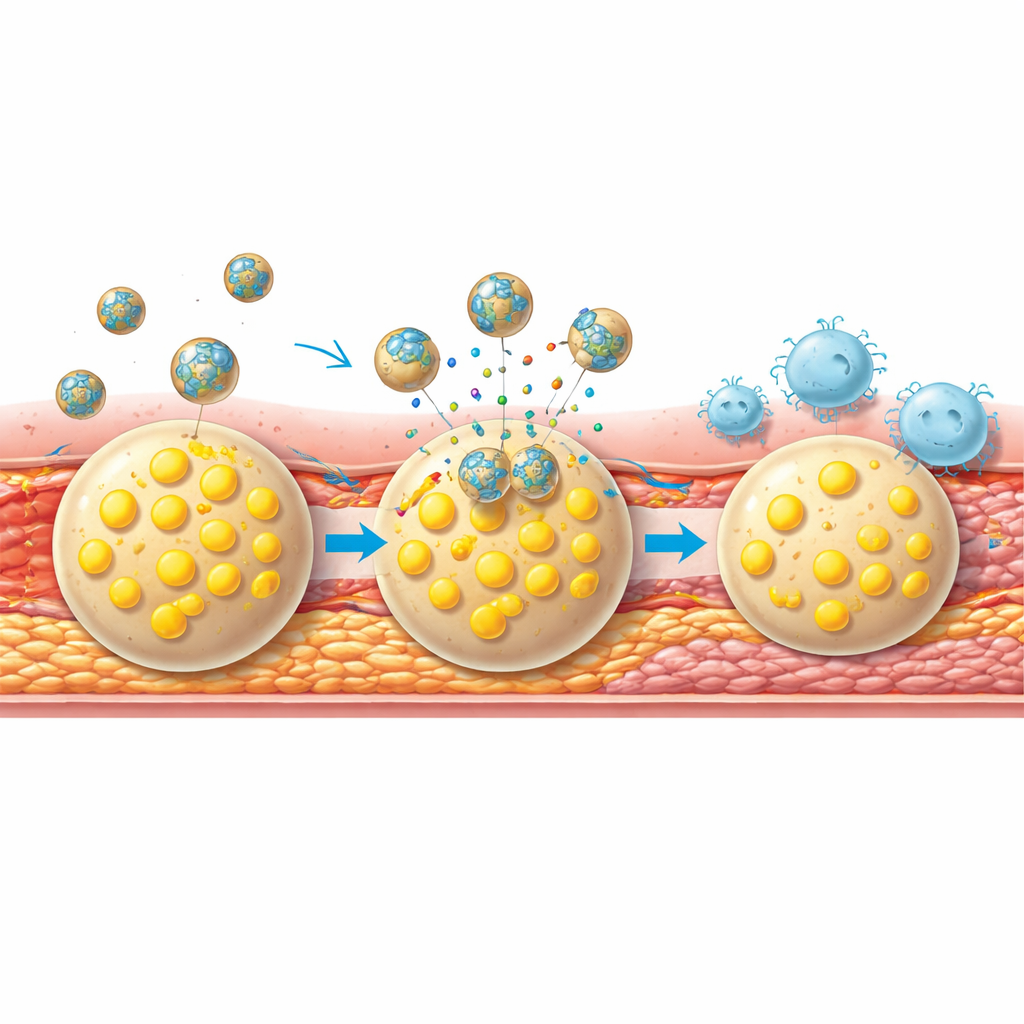
In cell culture, the nanoagent proved gentle to healthy cells, showing little toxicity or membrane damage. When exposed to foam cells, however, the particles were taken up far more efficiently than simpler versions lacking the membrane cloak or targeting peptide. Once inside these diseased cells, the oxidative environment triggered the polymer shell to break down and release atorvastatin. This reduced harmful reactive oxygen levels, curbed the uptake of fatty particles, and boosted the export of cholesterol by turning on natural transport proteins. At the same time, the treatment lowered levels of CD47, a surface molecule that normally tells immune cells “don’t eat me,” making the foam cells easier for macrophages to clear away.
Revealing and repairing plaques in living animals
In mice genetically prone to atherosclerosis and fed a high‑fat diet, the nanoagent circulated in the bloodstream for hours and accumulated strongly in arterial plaques. The team showed that infrared, photoacoustic, and MRI signals all rose together in plaque‑rich regions and closely matched tissue measures of plaque size, inflammation, and fragility. More advanced, unstable plaques produced stronger signals than milder ones, suggesting the platform can grade disease severity. When used repeatedly as a treatment, the targeted nanoagent shrank overall plaque burden far more than free atorvastatin or less sophisticated nanoparticle versions. It reduced oxidative stress, lowered inflammatory messenger levels, and shifted immune cell populations toward a calmer, more protective balance.
Making plaques tougher and tracking progress
Beyond shrinking plaques, the therapy made them harder to rupture. Treated mice showed less of an enzyme that erodes the fibrous cap over plaques, more collagen and smooth muscle cells that strengthen this cap, and fewer fragile new blood vessels that can leak and destabilize the lesion. Importantly, the same nanoparticles used for treatment also served as real‑time reporters of success: after weeks of therapy, repeat imaging sessions showed much weaker multimodal signals in the carotid arteries, mirroring the healthier plaque structure seen under the microscope.
What this could mean for patients
To a layperson, this work points toward future “smart contrast agents” that do far more than just help radiologists see blockages. This layered nanoagent finds the most dangerous, inflamed plaques, delivers drug precisely where it is needed, helps the body clear harmful foam cells, and then reports back on how well the treatment is working—all without surgery. While these results are in mice and human translation will require careful testing, the study outlines a powerful strategy for turning silent, unstable artery plaques into better‑behaved, sturdier structures before they cause heart attacks or strokes.
Citation: Song, J., Kang, X., Yang, S. et al. A hierarchical theranostic nanoagent for multimodal imaging and targeted foam cell intervention in atherosclerosis. Nat Commun 17, 3794 (2026). https://doi.org/10.1038/s41467-026-70463-7
Keywords: atherosclerosis, nanoparticles, multimodal imaging, foam cells, plaque stabilization