Clear Sky Science · en
Depression of tryptophan may contribute to adverse effects of naproxen
Why common pain pills matter for your whole body
Millions of people take non-steroidal anti-inflammatory drugs (NSAIDs) like naproxen for headaches, joint pain, or injuries, often assuming they are harmless over-the-counter helpers. Yet these drugs can quietly raise the risk of stomach bleeding and heart problems. This study asks a simple but important question: what do these medicines do to the body’s chemistry, and could a natural nutrient called tryptophan help explain, and perhaps soften, some of their hidden dangers?
A closer look at two similar pain relievers
The researchers compared naproxen, a traditional NSAID that blocks two related enzymes, with celecoxib, a newer drug that targets just one of them. Sixteen healthy volunteers took each treatment—naproxen, celecoxib, or a placebo—for a week in a carefully controlled crossover study. Blood and urine samples were then analyzed using detailed chemical profiling to see how thousands of small molecules changed in response to the drugs. Although both medicines successfully reduced the fatty-acid messengers that drive pain and fever, naproxen stood out for causing a clear drop in tryptophan and its main breakdown product, kynurenine, while celecoxib did not. This pattern suggested that naproxen was disturbing an important metabolic pathway beyond its intended target.
The surprising role of an everyday amino acid
Tryptophan is best known as the building block the body uses to make proteins and certain brain chemicals, but it also feeds a web of pathways linked to immunity, blood pressure, and energy use. In both people and mice, naproxen consistently lowered circulating tryptophan and kynurenine, while leaving other related molecules largely unchanged. Follow-up experiments showed that this effect did not depend on blocking the enzymes that naproxen is designed to inhibit. Even mice genetically engineered to lack those enzymes still lost tryptophan when given the drug. Instead, naproxen appeared to compete directly with tryptophan for binding sites on albumin, a major blood protein that ferries many molecules around the body. By clinging more tightly to these sites, naproxen pushed tryptophan and kynurenine off the protein and out into circulation, where they were more easily broken down and cleared.
Gut microbes and inflamed organs enter the story
The team also explored how this chemical shift might ripple through the body. In humans and mice, naproxen treatment subtly reshaped the gut microbiome, favoring certain bacterial groups previously linked to tryptophan breakdown into indole compounds. At the same time, mice fed naproxen showed small but telling signs of harm: more hidden blood in their stool and gene activity in the heart and gut that pointed to inflammation and tissue damage. Many of these alarm signals traced back to pathways driven by the immune messenger IL-1β and to a molecular complex called the inflammasome, both of which are involved in serious cardiovascular and intestinal disease.
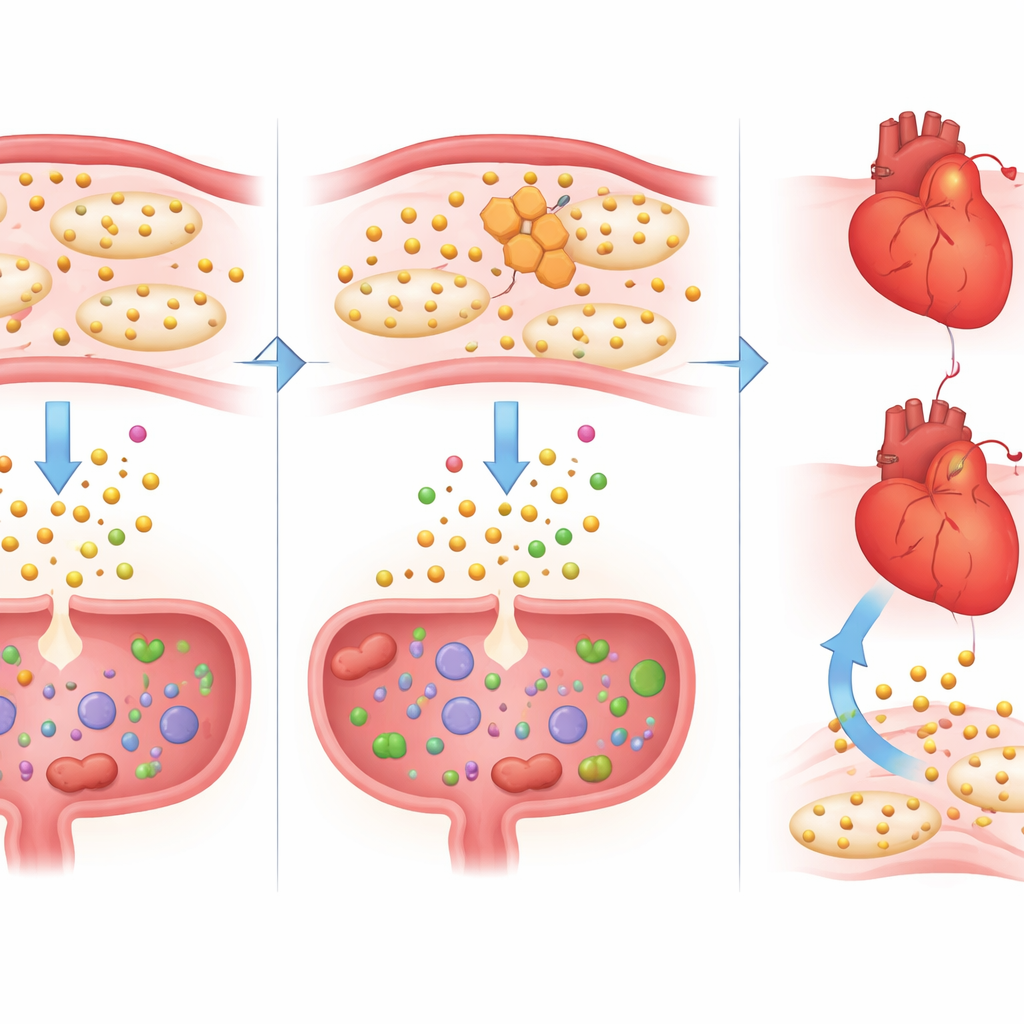
Restoring balance with extra tryptophan
To test whether loss of tryptophan was part of the problem rather than a harmless side effect, the scientists gave some naproxen-treated mice a controlled dose of extra tryptophan. This supplementation restored blood tryptophan levels to normal and had striking benefits. In the heart, hundreds of genes that naproxen had pushed toward a more inflammatory, injury-prone state swung back toward a healthier pattern. Signals tied to cytokine storms, heart enlargement, and poor pumping function quieted down, and IL-1β activity fell at both the RNA and protein levels. In the gut, tryptophan reduced stool blood and reversed changes in genes involved in barrier integrity, wound healing, and immune control. Notably, this rescue did not undo naproxen’s effect on platelets, indicating that it eased damage without interfering with the intended anti-clotting action.
What this means for people using pain relievers
Together, these findings suggest that naproxen’s tendency to deplete tryptophan—and to channel it toward breakdown by gut microbes—may help drive the drug’s unwanted effects on the heart and digestive tract. While more work is needed in larger, longer human studies, the results hint that tracking blood tryptophan could serve as an early warning marker of risk, and that carefully designed tryptophan supplementation might someday soften the collateral damage of widely used painkillers. For now, the study underscores that even familiar over-the-counter drugs can nudge our body’s chemistry in unexpected ways, and that understanding those shifts may open avenues to make everyday medicines safer.
Citation: Ghosh, S., Lahens, N.F., Barekat, K. et al. Depression of tryptophan may contribute to adverse effects of naproxen. Nat Commun 17, 2776 (2026). https://doi.org/10.1038/s41467-026-69684-7
Keywords: naproxen, tryptophan, NSAID side effects, gut microbiome, cardiovascular inflammation