Clear Sky Science · en
NCX1 reverse mode promotes calcium-dependent Neutrophil Extracellular Trap formation and lung damage in chronic obstructive pulmonary disease
Why tiny cells in smoker’s lungs matter
Chronic obstructive pulmonary disease (COPD) is a leading cause of death worldwide, most often linked to years of cigarette smoking. Many patients live with daily breathlessness and face frequent flare-ups, yet current treatments do little to halt the slow destruction of lung tissue. This study dives deep into one particular type of white blood cell—neutrophils—and a single molecular “gate” in their surface to explain how cigarette smoke turns helpful immune cells into engines of long‑lasting lung damage. By pinpointing this switch, the researchers reveal a potential way to calm harmful inflammation without shutting down the body’s defenses.
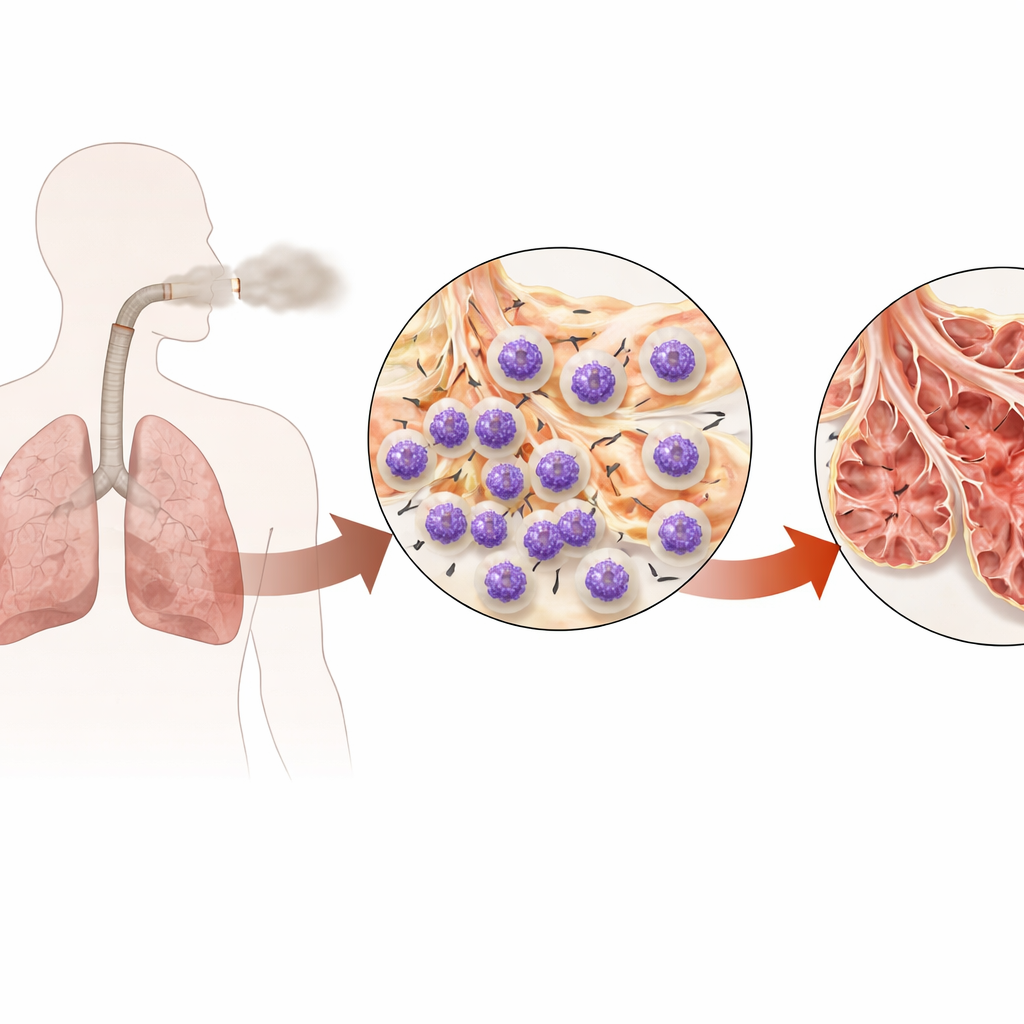
A stubborn lung disease fueled by overactive defenders
COPD slowly scars and hollows out the lungs, leaving thickened airways and enlarged, fragile air sacs. Neutrophils, first‑responder immune cells, are abundant in the sputum and lung tissue of people with COPD, and their presence tracks with worse airflow and lung function. But simply blunting neutrophils across the whole body risks serious infections. The authors therefore asked a more targeted question: is there a specific internal control in neutrophils that cigarette smoke hijacks to keep them activated, clustered in the lungs, and injuring tissue over the long term?
A calcium gate in neutrophils is turned up in COPD
The team focused on NCX1, a protein that swaps sodium and calcium ions across the cell membrane. In human lung samples, fluid from the airways, and circulating blood, NCX1 levels were markedly higher in neutrophils from patients with mixed chronic bronchitis and emphysema than in people without chronic lung disease. This increase tracked closely with markers of neutrophil activation, suggesting that NCX1 might be more than a bystander. In mice exposed to long‑term cigarette smoke, neutrophils in the lungs likewise showed elevated NCX1, mirroring the human findings.
How smoke pushes neutrophils into a destructive state
Using mouse models and cultured cells, the researchers uncovered how NCX1 helps convert cigarette smoke exposure into damaging inflammation. Normally, NCX1 can move calcium out of cells; under certain conditions it flips and brings calcium in. Cigarette smoke extract strongly pushed neutrophils into this “reverse mode,” driving a surge of calcium inside the cells. This calcium overload was essential for the formation of neutrophil extracellular traps (NETs)—webs of DNA studded with toxic enzymes that can trap microbes but also shred nearby tissue. When NCX1 was genetically deleted only in neutrophils, or when its reverse activity was blocked with a drug, smoke‑exposed neutrophils showed far less calcium influx and released far fewer NETs.
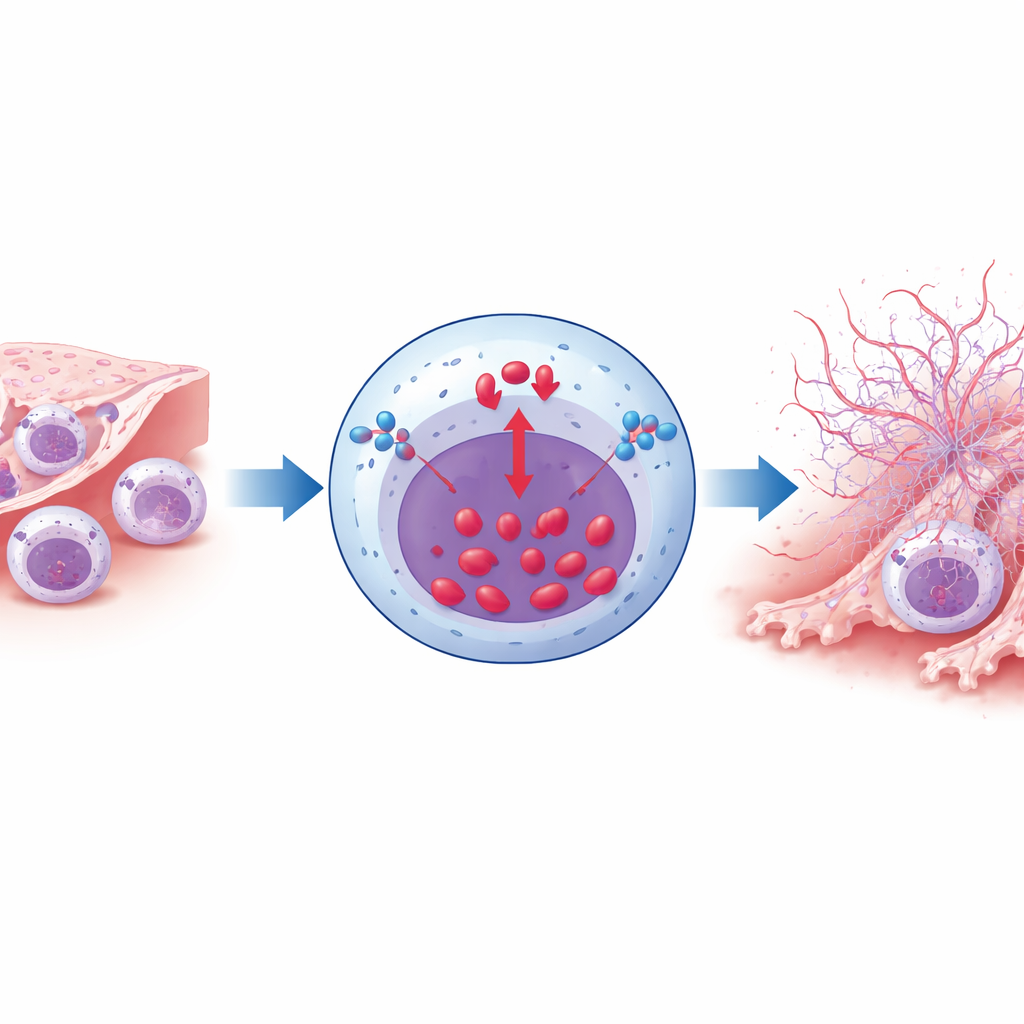
Less trapping, less scarring, better lungs
The downstream effects of reducing NCX1‑driven NET formation were striking. In smoke‑exposed mice, neutrophil‑specific NCX1 deletion led to fewer neutrophils clogging the lungs, less collagen buildup and smooth‑muscle thickening around airways, and milder destruction of air sacs. Measures of lung function, including airflow and the ability of lungs to expand, were partially preserved compared with control animals. Independently blocking NETs with a different drug, Cl‑amidine, produced similar benefits: fewer neutrophils, less structural damage, and better breathing mechanics. Importantly, removing NCX1 from neutrophils did not alter how these cells developed, migrated, or swallowed bacteria under normal conditions, suggesting that the intervention mainly curbs their pathological behavior in a smoke‑filled environment.
A new, more precise target for COPD treatment
To a lay reader, the message is that the study has identified a key molecular switch—NCX1 running in reverse—that helps explain why neutrophils linger and cause ongoing harm in smokers’ lungs. By feeding excess calcium into these cells, NCX1 promotes the release of sticky, enzyme‑coated DNA nets that injure lung structures and maintain a chemical environment that keeps neutrophils coming back. Turning down this switch, either by removing NCX1 from neutrophils or by blocking its reverse activity, breaks this vicious cycle in mice and protects lung function without broadly weakening the immune system. While more work is needed to understand exactly how cigarette components flip NCX1 into reverse and to test safety in humans, the findings point to NCX1 and NET formation as promising, more selective drug targets for slowing or preventing COPD‑related lung damage.
Citation: Liao, SX., Wang, YW., Shi, LM. et al. NCX1 reverse mode promotes calcium-dependent Neutrophil Extracellular Trap formation and lung damage in chronic obstructive pulmonary disease. Nat Commun 17, 3801 (2026). https://doi.org/10.1038/s41467-026-69636-1
Keywords: COPD, neutrophils, calcium signaling, NETs, cigarette smoke