Clear Sky Science · en
ASB7 promotes osteosarcoma lung metastasis through ubiquitin-mediated degradation of ATF2
Why runaway bone tumors matter
Osteosarcoma is a rare but aggressive bone cancer that strikes mainly children, teenagers, and older adults. Its deadliest feature is its tendency to spread to the lungs, often turning a treatable local disease into a life‑threatening one. This study asks a simple but crucial question: what molecular "switches" help osteosarcoma cells break away from bone, travel through the body, and colonize the lungs—and can those switches be turned back off?
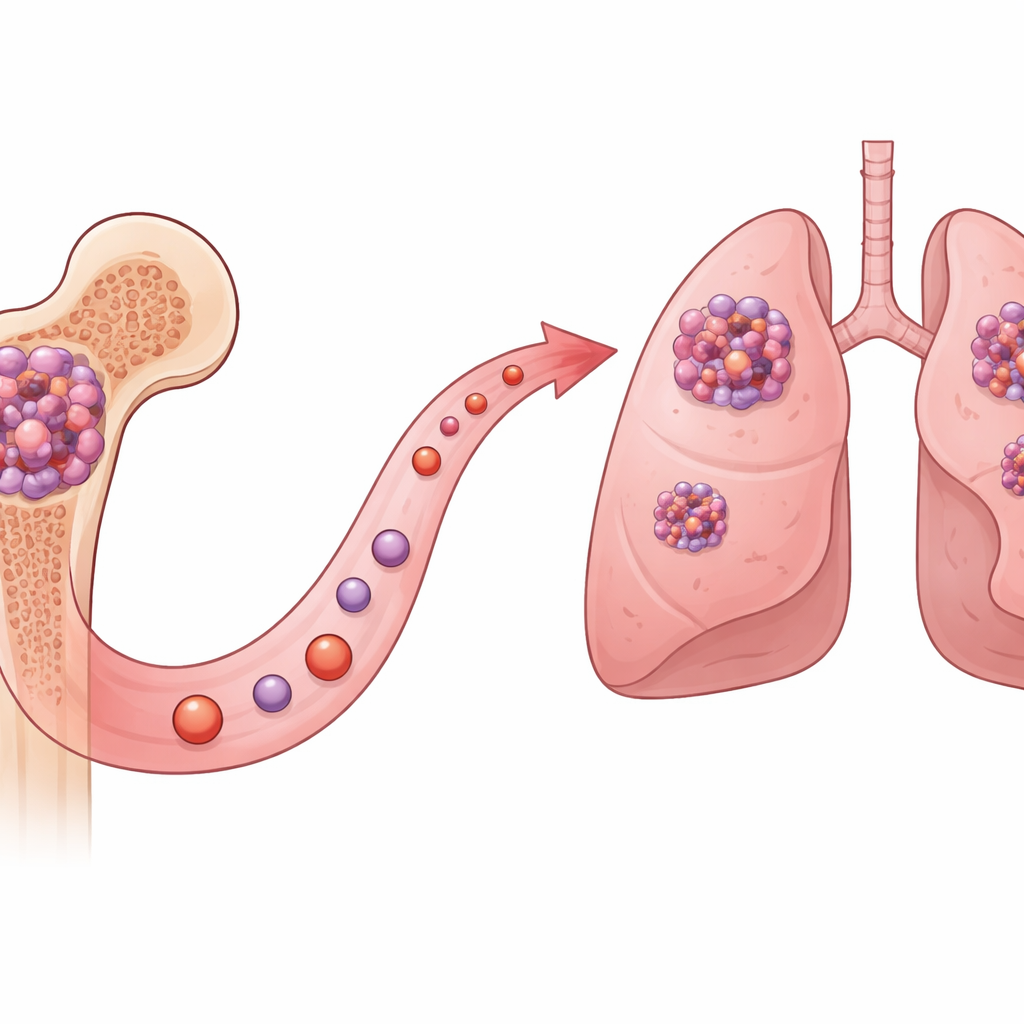
A hidden helper of cancer spread
The researchers focused on a protein called ASB7, previously known for helping cells dispose of other proteins. By mining large public cancer datasets and analyzing patient tumor samples, they found that osteosarcoma cells often carry extra copies of the ASB7 gene and make unusually high amounts of ASB7 protein. Patients whose tumors were rich in ASB7 tended to have worse survival, hinting that this molecule does more than routine housekeeping—it seems to help tumors behave more aggressively.
From smooth cells to roaming invaders
To see what ASB7 actually does in cancer cells, the team engineered osteosarcoma cells to either overproduce or lack ASB7. When ASB7 levels were cranked up, cells sprouted many thin, finger‑like and fan‑shaped protrusions built from actin, the same scaffolding that normal cells use to crawl. These ASB7‑high cells moved faster through lab test chambers and invaded through gel that mimics body tissue. When implanted into the leg bones of mice, they were far more likely to seed numerous colonies in the lungs. Removing ASB7 had the opposite effect: cells lost their protrusions, migrated poorly, and formed far fewer lung metastases, without major changes in how quickly they divided.
A protective brake that gets dismantled
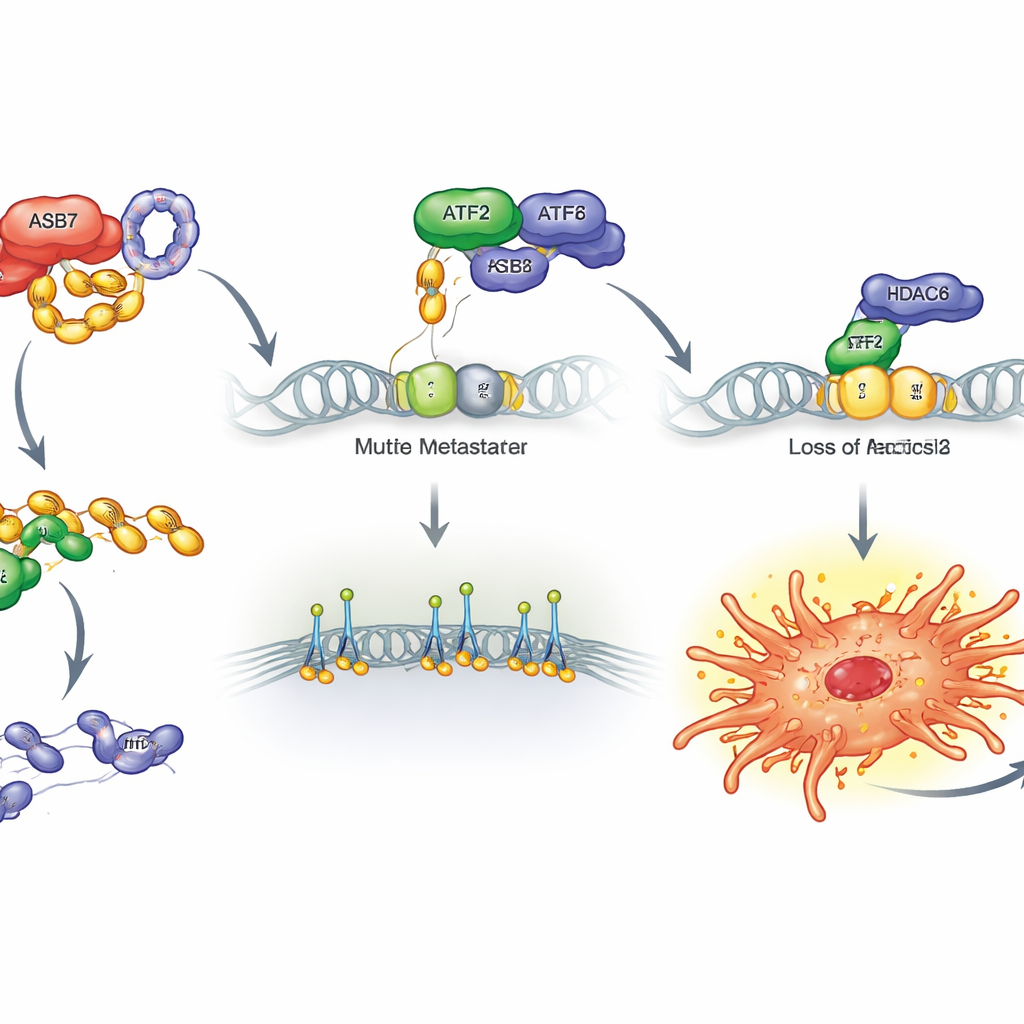
Digging deeper, the scientists identified another protein, ATF2, as a key brake on metastasis. ATF2 is a transcription factor, a protein that binds DNA and helps control which genes are turned on. In patient samples and large protein datasets, tumors with lots of ASB7 had little ATF2, and patients with low ATF2 did poorly. Boosting ATF2 in osteosarcoma cells reduced protrusions, slowed migration and invasion, and cut lung metastases in mice; deleting ATF2 did the reverse. Biochemical experiments revealed why: ASB7 teams up with a partner, CUL5, to tag ATF2 at a specific spot with small ubiquitin molecules, marking it for destruction by the cell’s protein‑shredding machinery. With ATF2 constantly broken down, its ability to restrain dangerous genes disappears.
Releasing a key grip on the surroundings
The team then asked which genes ATF2 normally keeps in check. By combining gene‑activity profiles with public DNA‑binding maps, they homed in on ITGB2, a member of the integrin family of proteins that physically connect cells to their surroundings and help them pull themselves forward. ATF2 normally sits near the ITGB2 gene and, together with another protein called HDAC6, keeps it quiet. When ASB7 destroys ATF2, this partnership is disrupted, HDAC6 binds less, and ITGB2 is freed to turn on strongly. Experiments confirmed this chain: raising ASB7 or deleting ATF2 increased ITGB2, while raising ATF2 or removing ASB7 lowered it. Forcing cells to overproduce ITGB2 alone was enough to boost protrusions, migration, invasion, and lung colonization in mice, whereas removing ITGB2 blunted these behaviors.
What this means for future treatment
Taken together, the work outlines a clear route by which osteosarcoma cells gain the ability to spread. ASB7 acts as a molecular "hit man" that destroys ATF2, weakening its partnership with HDAC6 at the ITGB2 gene. This releases ITGB2 and related genes, giving tumor cells extra grip and mobility as they invade blood vessels and seed the lungs. Because this pathway seems especially active in bone sarcomas, it highlights several promising therapeutic ideas: blocking ASB7’s activity, stabilizing ATF2, dampening ITGB2, or tuning the connected DNA‑silencing machinery might all help keep osteosarcoma from metastasizing, turning a frequently deadly disease into one that is far more manageable.
Citation: Zou, Y., Zhong, J., Huo, L. et al. ASB7 promotes osteosarcoma lung metastasis through ubiquitin-mediated degradation of ATF2. Cell Discov 12, 31 (2026). https://doi.org/10.1038/s41421-026-00890-9
Keywords: osteosarcoma, metastasis, ASB7, ATF2, integrins