Clear Sky Science · en
PLAAT2 suppresses gastric cancer progression by facilitating cMyc ubiquitination and inhibiting MEK/ERK signaling
Why This Study Matters for Stomach Cancer
Stomach (gastric) cancer remains one of the world’s deadliest cancers, in part because it is often detected late and can spread quickly. This study explores a little-known natural defender inside our cells, a protein called PLAAT2, and shows how it helps keep stomach cancer in check. By uncovering how this molecule works and why it is lost in many tumors, the research points to a new weak spot in gastric cancer that future drugs might be able to target.
A Missing Protector in Tumor Tissue
To see whether PLAAT2 plays a role in stomach cancer, the researchers began by comparing tumor samples with nearby noncancerous tissue from more than a hundred patients, as well as data from large public cancer databases. They found a clear pattern: PLAAT2 levels were much lower in cancerous tissue than in healthy stomach lining. Patients whose tumors had especially low amounts of PLAAT2 tended to have more advanced disease, more lymph node spread, and poorer overall survival. Similar reductions were seen when they examined stomach cancer cell lines in the lab, suggesting that loss of this protein is a common feature of the disease.
How Silencing and Restoring PLAAT2 Changes Cancer Behavior
The team then tested what happens when PLAAT2 levels are artificially dialed down or boosted in cancer cells. When PLAAT2 was switched off, tumor cells grew faster, formed more colonies, moved more easily through laboratory membranes, and were less likely to undergo programmed cell death. When PLAAT2 was restored to higher levels, the opposite happened: cell growth slowed, movement and invasion were reduced, and more cells died. These changes were also seen in mice, where tumors lacking PLAAT2 grew larger, while those with extra PLAAT2 stayed smaller, confirming that this molecule acts as a genuine brake on cancer progression.
Short-Circuiting a Growth Switch Inside the Cell
Digging deeper, the scientists discovered that PLAAT2 controls a powerful growth driver protein called cMyc, which is abnormally active in many cancers. Rather than changing how much cMyc is produced, PLAAT2 affected how quickly cMyc is broken down. With more PLAAT2 present, cMyc proteins were tagged for disposal and cleared away faster; with less PLAAT2, cMyc lingered and accumulated. This breakdown process relies on a cellular “recycling” system in which small molecular tags signal which proteins should be destroyed. PLAAT2 was found to help bring cMyc into contact with TRIM32, an enzyme that attaches these tags. TRIM32 then places specific tags on cMyc at two key positions, marking it for destruction and keeping its growth-promoting activity in check.
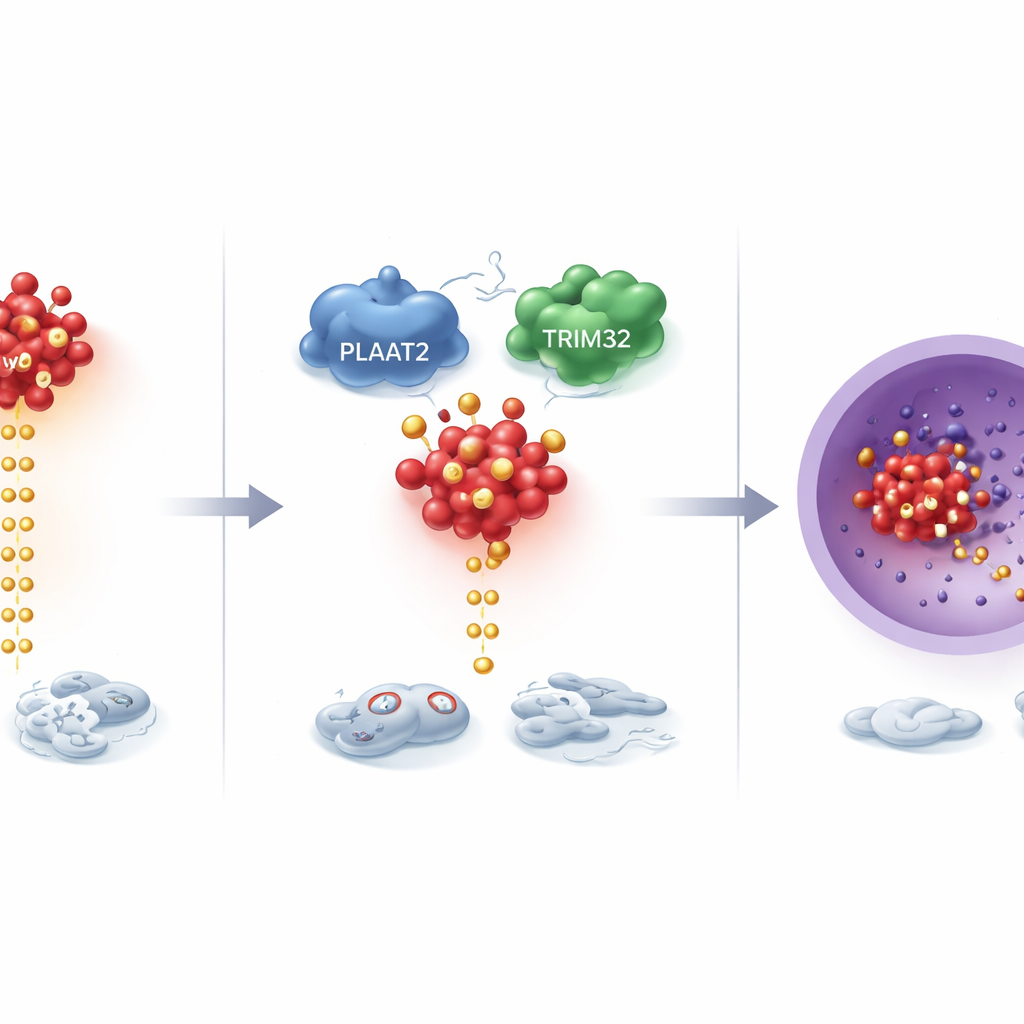
Turning Down a Major Cancer Signaling Pathway
Because cMyc can activate a chain of signals that encourage cells to divide, migrate, and change identity, the researchers looked at one major route it controls, known as the MEK/ERK pathway. When PLAAT2 levels were low, this pathway became more active, and cancer cells showed features of a process called epithelial–mesenchymal transition, in which normally well-behaved cells loosen their connections and become more mobile and invasive. Restoring PLAAT2 reversed these changes: the MEK/ERK pathway quieted down, markers of invasive behavior dropped, and cells behaved less aggressively. Blocking or boosting cMyc directly could undo the effects of PLAAT2, underscoring that this protective protein works largely by reining in cMyc and the signals it sends.
What This Means for Future Treatments
Taken together, the findings suggest that PLAAT2 is an important natural safeguard against stomach cancer, helping to recruit TRIM32 to strip away excess cMyc and, in turn, dampen a major growth and spread pathway. When PLAAT2 is lost or silenced, cMyc and MEK/ERK signaling run unchecked, allowing tumors to grow and invade more easily. By highlighting this PLAAT2–TRIM32–cMyc chain as a critical control point, the study opens up new possibilities: future therapies might aim to restore PLAAT2, boost TRIM32’s action on cMyc, or directly mimic this degradation process to slow or stop gastric cancer progression.
Citation: Chu, M., Shi, X., Shi, Z. et al. PLAAT2 suppresses gastric cancer progression by facilitating cMyc ubiquitination and inhibiting MEK/ERK signaling. Cell Death Dis 17, 314 (2026). https://doi.org/10.1038/s41419-026-08546-y
Keywords: gastric cancer, PLAAT2, cMyc, ubiquitination, MEK ERK signaling