Clear Sky Science · en
Cognitive trajectories in Parkinson’s disease patients, a review on the impact of subthalamic deep brain stimulation (STN-DBS) and emerging adaptive strategies
Why Brain Stimulation in Parkinson’s Matters
Parkinson’s disease is best known for shaking and slowed movement, but many people are just as troubled by memory slips, word‑finding problems, and trouble focusing. To ease movement problems, doctors increasingly use deep brain stimulation (DBS), in which thin electrodes are implanted deep in the brain. This review asks a question patients and families often have: what does this treatment do to thinking and mood over the long run, and could new “smart” forms of DBS help protect the mind as well as the body?
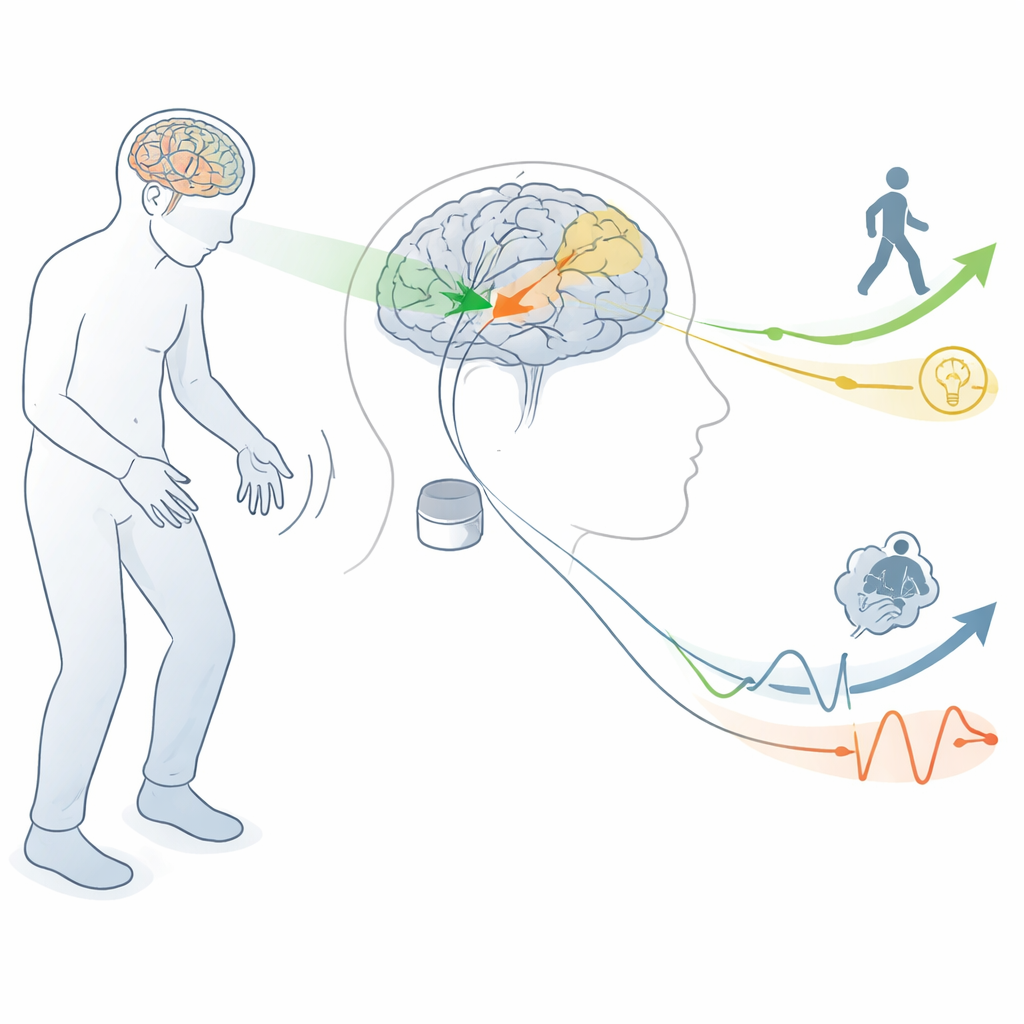
The Brain Circuit Behind Movement and Thought
The authors start by explaining how Parkinson’s disrupts brain circuits that normally help us start and control movement. A small region called the subthalamic nucleus, buried deep in the brain, becomes overactive when the chemical messenger dopamine is lost. High‑frequency DBS of this region (STN‑DBS) can calm abnormal activity and greatly improve tremor, stiffness, and unwanted movements. But this same area also connects to brain regions that support language, planning, emotion, and motivation. That overlap means that when doctors stimulate to help movement, they may also nudge circuits involved in thinking and feeling.
What Happens to Thinking After DBS
Drawing on randomized trials, long‑term follow‑ups, and meta‑analyses, the review finds a clear pattern. Overall intelligence and everyday thinking are usually preserved, and many patients feel better emotionally after surgery. However, one specific weakness appears again and again: difficulty coming up with words quickly, a skill tested by asking people to name as many items as they can from a category. Some studies also report mild changes in planning, mental flexibility, and memory over many years. These effects are typically modest, and many patients still report improved quality of life because their movement and mood are better. Importantly, long‑term data suggest that broader decline in thinking mostly reflects the natural progression of Parkinson’s rather than the stimulation itself.
Why Effects Differ From Person to Person
Not every patient shows the same cognitive changes after STN‑DBS. The review highlights several reasons for this variability. Age and baseline thinking skills matter: older people and those who already show mild cognitive problems are more likely to decline later on. The precise spot where the electrode sits in the subthalamic nucleus is also crucial. Contacts closer to regions linked with language and mood are more likely to affect word‑finding and emotional state. Beyond surgery and stimulation, features of the disease itself—such as Parkinson’s subtype, sleep problems, overlapping brain pathologies, genes, and general brain health—shape how vulnerable someone is. Depression, anxiety, and medication changes can further cloud the picture, making careful pre‑surgical assessment and follow‑up essential.
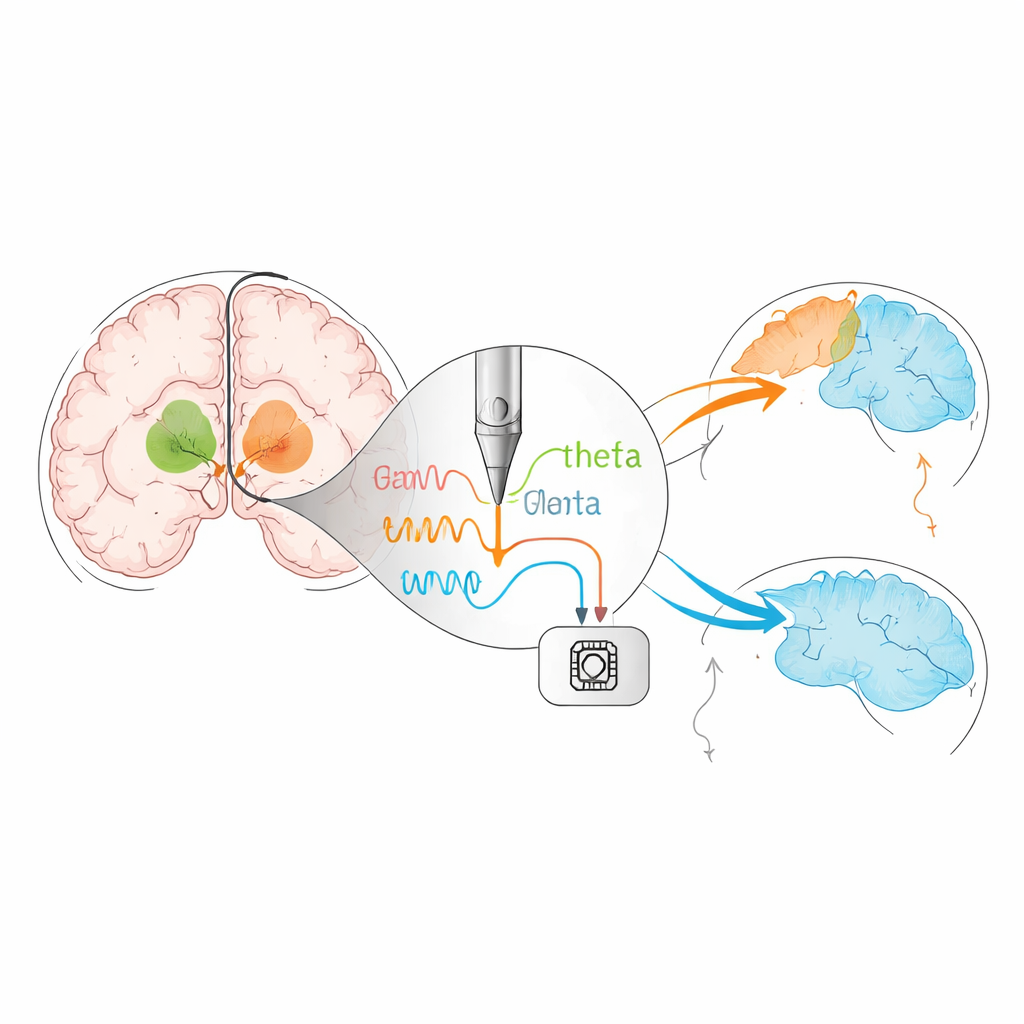
From Fixed to “Smart” Stimulation
Traditional DBS delivers steady pulses around the clock, regardless of what the person is doing or how their brain activity is changing. The review describes a next step: closed‑loop, or adaptive, DBS. In this approach, the device listens to the brain’s own electrical rhythms and adjusts stimulation in real time. Early systems track beta‑band activity, a brain rhythm linked to stiffness and slowness, to fine‑tune stimulation for movement. Newer work suggests that other rhythms, such as slower theta waves, may reflect attention, self‑control, or sleep state. Carefully timed stimulation at these frequencies has been shown in small studies to make decisions more cautious and improve working memory without harming movement, hinting that future devices might ease cognitive and sleep problems as well as motor symptoms.
Promises and Hurdles for Protecting the Mind
The authors conclude that for most people with Parkinson’s, STN‑DBS brings clear and lasting movement benefits, with thinking changes that are usually limited to specific skills like rapid word generation. The real opportunity ahead lies in using smarter, feedback‑guided stimulation to better balance motor gains with protection of mood and cognition. To get there, researchers must identify reliable brain signals that reflect non‑motor symptoms, refine imaging and surgical techniques to spare sensitive circuits, and run long‑term trials that track both movement and mental outcomes. If these challenges can be met, future DBS systems may not only steady the body but also help preserve independence, memory, language, and decision‑making over the course of Parkinson’s disease.
Citation: Almeida, V., Herz, D.M., Blech, J. et al. Cognitive trajectories in Parkinson’s disease patients, a review on the impact of subthalamic deep brain stimulation (STN-DBS) and emerging adaptive strategies. Transl Psychiatry 16, 233 (2026). https://doi.org/10.1038/s41398-026-04013-6
Keywords: Parkinson’s disease, deep brain stimulation, cognition, closed-loop neuromodulation, subthalamic nucleus