Clear Sky Science · en
CCK2R regulates METH-induced CPP acquisition within VTA-BLA-BNST circuit in male mice
Why this matters for people and society
Methamphetamine addiction tears apart lives, yet current treatments often fail to stop powerful cravings and relapse. This study looks deep inside the brain to find a specific “switch” that helps meth link pleasurable drug effects to places and cues in the environment. By pinpointing this switch in a precise chain of brain regions in mice, the work suggests new, highly targeted ways to weaken drug memories and reduce the pull of meth, while leaving normal mood and thinking largely untouched. 
A brain pathway that turns context into craving
The researchers focused on a three-step pathway connecting key reward and emotion hubs: the ventral tegmental area (VTA), the basolateral amygdala (BLA), and the bed nucleus of the stria terminalis (BNST). The VTA contains dopamine-releasing cells that signal reward; the BLA helps attach emotional value to experiences; and the BNST integrates stress and motivation. When mice were given meth in one chamber of a two-room box, they later preferred that side, a classic sign that the drug’s pleasurable effects had become tied to that place. This “conditioned place preference” model allowed the team to track how activity in the VTA–BLA–BNST chain changed as drug memories formed.
A digestive hormone moonlighting in the brain
Surprisingly, a molecule best known from the gut—cholecystokinin (CCK)—turned out to be central. In the brain, certain VTA dopamine neurons also release CCK. The team used genetically engineered mice to delete CCK only from these cells. Without CCK, mice no longer developed a strong preference for the meth-paired chamber, and the usual meth-driven increase in VTA cell firing was blunted. Yet these mice behaved normally in tests of movement, memory, anxiety, and depression-like behavior. This suggests that VTA CCK is not needed for everyday brain function, but is crucial when meth tries to stamp in a powerful reward memory.
A single receptor as a critical gate in the amygdala
CCK acts by binding to receptors on other neurons, and the authors found that one of them, called CCK2R, becomes especially abundant in the BLA after meth exposure. When they deleted CCK2R specifically from the BLA’s excitatory cells, meth could no longer create strong place preference, and the abnormal burst of activity in these cells was largely normalized. Fine-scale recordings showed that meth usually boosts excitatory connections and tiny protrusions called dendritic spines in the BLA—changes linked to stronger, longer-lasting memories. Removing CCK2R prevented these structural and electrical shifts, while again leaving general learning and mood intact.
Extending the signal to a stress hub
The story did not end in the amygdala. Using sophisticated viral tracing, the researchers showed that VTA dopamine neurons feed into BLA excitatory cells, which in turn project to the BNST, a region tied to anxiety and addiction. Silencing this VTA–BLA–BNST chain during meth training sharply reduced place preference and calmed the overactive BNST neurons. Crucially, knocking out CCK2R only in the subset of BLA cells that both receive VTA input and send signals to the BNST blocked meth’s effects on BNST activity, synaptic strengthening, and dendritic spine growth. Circuits that did not feed into the BNST were far less affected, underscoring how precisely the CCK2R “switch” is wired. 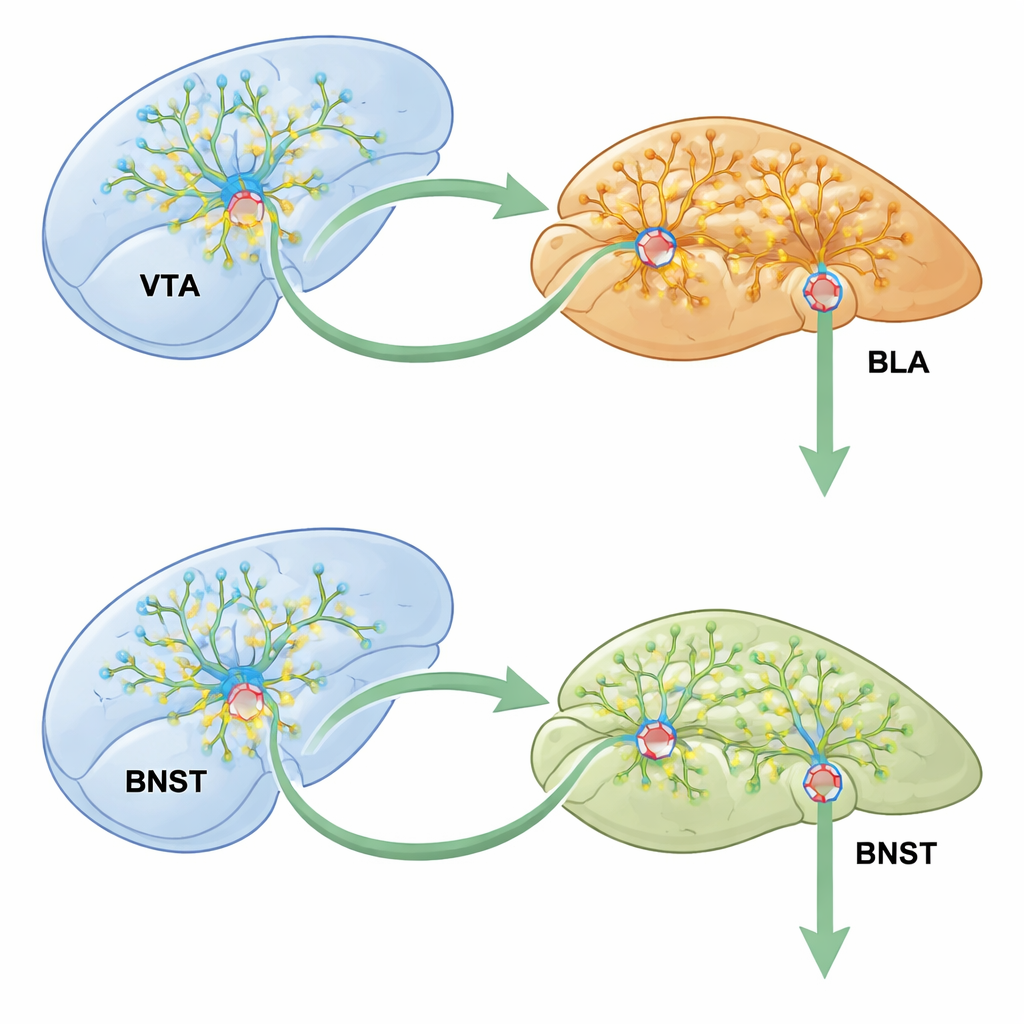
What this could mean for future treatment
Overall, the study shows that meth hijacks a specific gut–brain messenger system—CCK acting on CCK2R—within a defined VTA–BLA–BNST pathway to harden drug-related memories and drive preference for drug-associated places. Turning off CCK2R in this circuit erased many hallmarks of addiction in the mice, from heightened firing of neurons to extra synapses and altered brain rhythms, without broadly disturbing emotion or cognition. For a layperson, this means scientists have identified a very selective “circuit breaker” for meth reward. Drugs that safely block CCK2R in the right brain pathways might one day help weaken meth cravings and relapse risk, offering a more focused therapy than current approaches.
Citation: Wang, J., Zhang, M., Qiao, L. et al. CCK2R regulates METH-induced CPP acquisition within VTA-BLA-BNST circuit in male mice. Transl Psychiatry 16, 210 (2026). https://doi.org/10.1038/s41398-026-03982-y
Keywords: methamphetamine addiction, brain reward circuits, cholecystokinin, amygdala and BNST, drug memory