Clear Sky Science · en
The acid-sensing ion channel 1a modulates anxiety- and depression-related behaviors via its influencing on the activity of corticotropin-releasing hormone-expressing neurons in the hypothalamic paraventricular nucleus in male mice
How a Tiny Brain Switch Shapes Our Stress Levels
Stress is a familiar part of modern life, but under the skin it is driven by precise electrical and chemical signals in the brain. This study in male mice uncovers a previously hidden player in that system: a tiny “acid-sensing” channel in specific stress-command neurons. By showing how this channel boosts stress hormones and anxious or depressed behaviors—and how blocking it calms both brain and body—the work points toward new kinds of treatments for anxiety, depression, and other stress-linked illnesses.
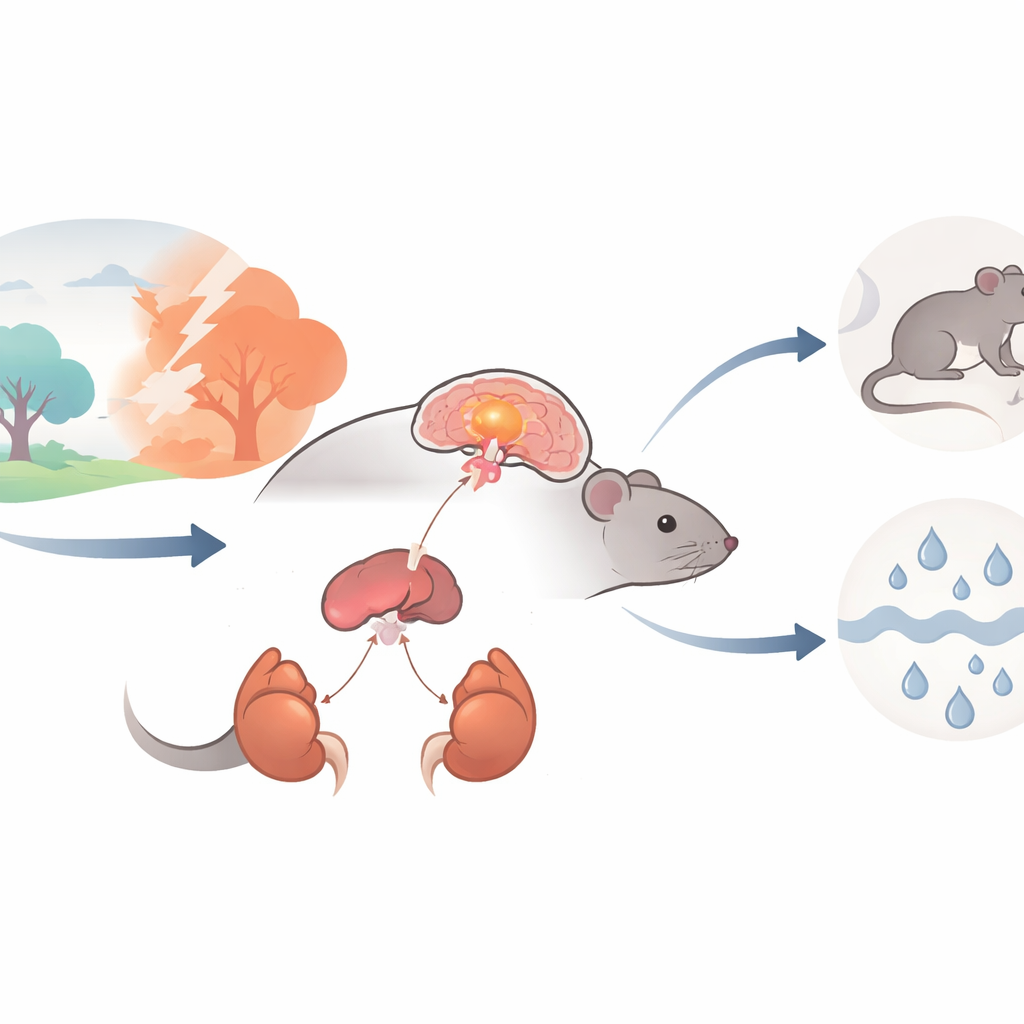
The Body’s Main Stress Alarm
When we face a challenge, a chain of command called the hypothalamic–pituitary–adrenal (HPA) axis springs into action. It begins in a small brain region, the hypothalamic paraventricular nucleus (PVN), where special nerve cells release corticotropin-releasing hormone (CRH). CRH signals the pituitary gland, which then tells the adrenal glands atop the kidneys to flood the bloodstream with stress hormones such as corticosterone. These hormones are essential for survival in short bursts, but if this alarm is switched on too strongly or too often, it can contribute to anxiety, depression, metabolic disease, and high blood pressure. The authors asked whether a particular ion channel, called ASIC1a, inside CRH-producing PVN neurons helps set the gain on this stress alarm.
A Hidden Valve in Stress Neurons
ASIC1a is a protein channel in cell membranes that opens when the nearby fluid becomes more acidic, allowing positively charged ions, including calcium, to rush into the cell. The team first confirmed that ASIC1a is abundant in CRH-producing PVN neurons in mice. They then used two complementary strategies to turn this channel down. In one, they infused a highly selective ASIC1a blocker directly into the PVN. In the other, they used engineered viruses in CRH-Cre mice to silence the ASIC1a gene specifically in CRH neurons. In both cases, mice behaved less anxiously in exploration tests and showed fewer depression-like signs in classic behavioral assays, even though their overall movement and speed were unchanged. At the same time, their blood levels of stress hormones ACTH and corticosterone after an acute stressor dropped, while baseline hormone levels remained normal. Together, these findings suggest that ASIC1a is not required for ordinary function but helps drive the overreaction to stress.
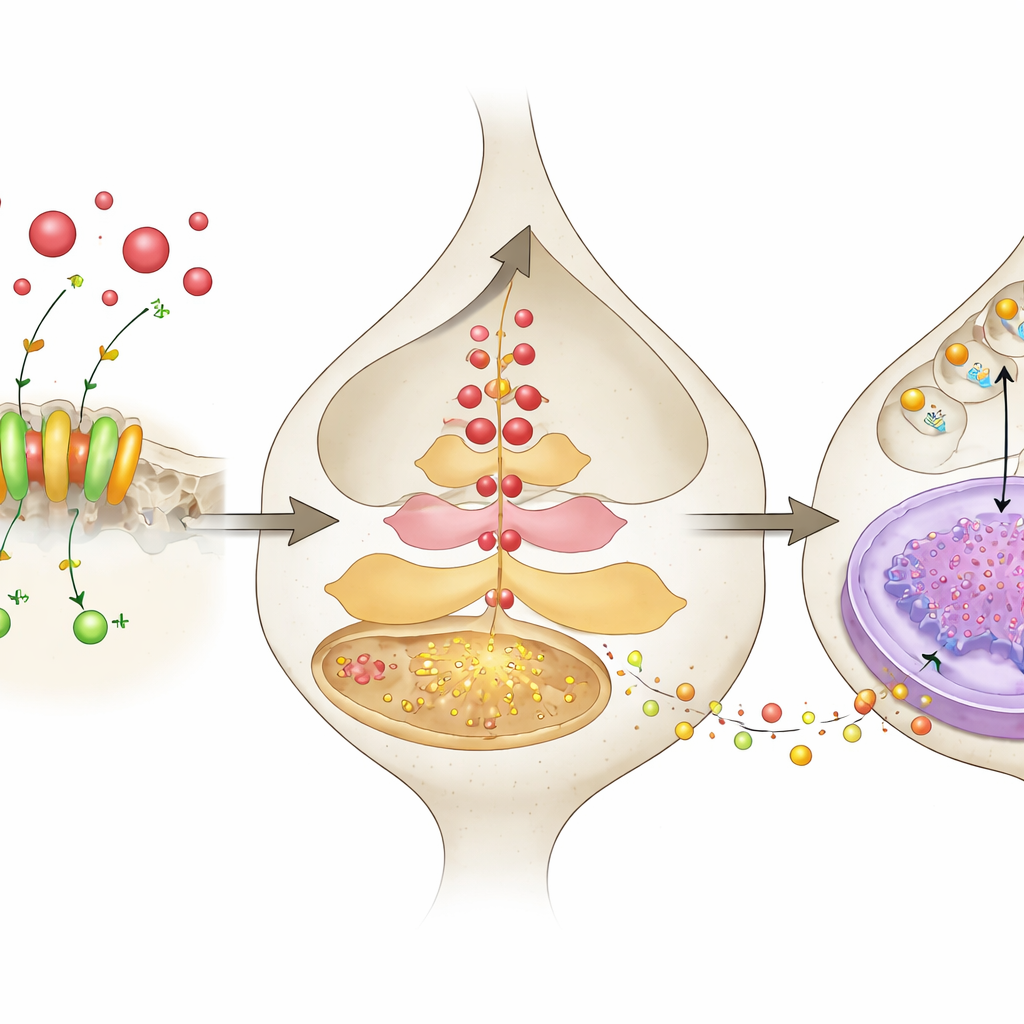
Zooming In: From Acid Signals to Stress Hormone Release
To probe what happens inside the neurons, the researchers recorded real-time calcium activity from CRH PVN neurons using a fluorescent reporter delivered by virus and read out with optical fibers in awake mice. During stressful events such as a foot shock or a forced swim, neurons with normal ASIC1a showed strong bursts of calcium signals, whereas neurons lacking ASIC1a responded much more weakly. In cell cultures, briefly lowering the pH of the surrounding solution—mimicking a more acidic environment—sharply increased CRH release, but this effect was blunted when ASIC1a was blocked or knocked down. The acid pulse also boosted CRH production inside cells, at both protein and messenger RNA levels, again in an ASIC1a-dependent manner. These experiments link acidity outside the neuron, ASIC1a opening, calcium entry, and increased CRH output as parts of a single cascade.
The Inner Relay: A Calcium-Driven Signaling Chain
The team then dissected the signaling chain between calcium entry and CRH gene activation. When ASIC1a was stimulated by acidic conditions, levels of an activated form of the enzyme CaMKII rose, as did levels of c-Fos, a fast-acting transcription factor known to turn on stress-related genes. Blocking ASIC1a or its ion flow reduced these changes. Using a drug that interferes with AP-1, the protein complex that includes c-Fos, the researchers dampened the acid-triggered rise in CRH production and release in cultured neurons. Infusing this AP-1 blocker into the PVN of mice also eased anxiety- and depression-like behaviors. These results support a model in which ASIC1a-driven calcium signals activate CaMKII and c-Fos, which then push the CRH gene into higher gear, amplifying the stress response.
What This Means for Future Treatments
Put simply, the study identifies ASIC1a in CRH-producing PVN neurons as a molecular “volume knob” on the brain’s main stress pathway. When ASIC1a is highly active, stress neurons fire more strongly, stress hormones surge higher, and animals behave in more anxious and depression-like ways. Turning ASIC1a down—either with precise genetic tools or with drugs—softens this reaction without shutting down normal behavior. While the work was done in male mice and focused on acute stress, it suggests that medicines targeting ASIC1a or its calcium-driven signaling partners could one day offer a new route to treating stress-related disorders such as anxiety, depression, and even some metabolic diseases.
Citation: Yue, J., Zhang, Q., Wang, M. et al. The acid-sensing ion channel 1a modulates anxiety- and depression-related behaviors via its influencing on the activity of corticotropin-releasing hormone-expressing neurons in the hypothalamic paraventricular nucleus in male mice. Transl Psychiatry 16, 189 (2026). https://doi.org/10.1038/s41398-026-03946-2
Keywords: stress response, acid-sensing ion channels, corticotropin-releasing hormone, anxiety and depression, hypothalamic-pituitary-adrenal axis