Clear Sky Science · en
Robust neurogenesis in chronic stroke monkeys following mesenchymal stem cell transplantation plus intermittent theta-burst stimulation
New Hope for Long-Term Stroke Survivors
Many people who survive a stroke are left with lasting weakness in an arm or leg, and after the first several months their recovery often stalls. This study in monkeys explores an inventive two-part treatment that aims to “reawaken” the brain’s ability to repair itself, even years after a stroke. By combining transplanted stem cells with gentle magnetic pulses to the skull, the researchers show that an apparently stable, damaged brain can once again grow new nerve cells and rebuild connections that improve movement.
Why Chronic Stroke Is So Hard to Treat
Stroke cuts off blood flow to parts of the brain, killing nerve cells and leaving behind a scarred region that controls movement, speech, or other functions. Standard rehabilitation works best in the first six months, when the brain is naturally most flexible. After that, patients enter a chronic phase in which improvements slow dramatically, leaving many with permanent disability. Existing tools such as magnetic brain stimulation can offer modest benefits, but their effects tend to fade. Stem cells, taken from tissues like bone marrow or umbilical cord, have shown promise in calming inflammation and supporting surviving neurons, yet they rarely take root long-term or fully restore function. This work tests whether these two approaches might be stronger together than either one alone.
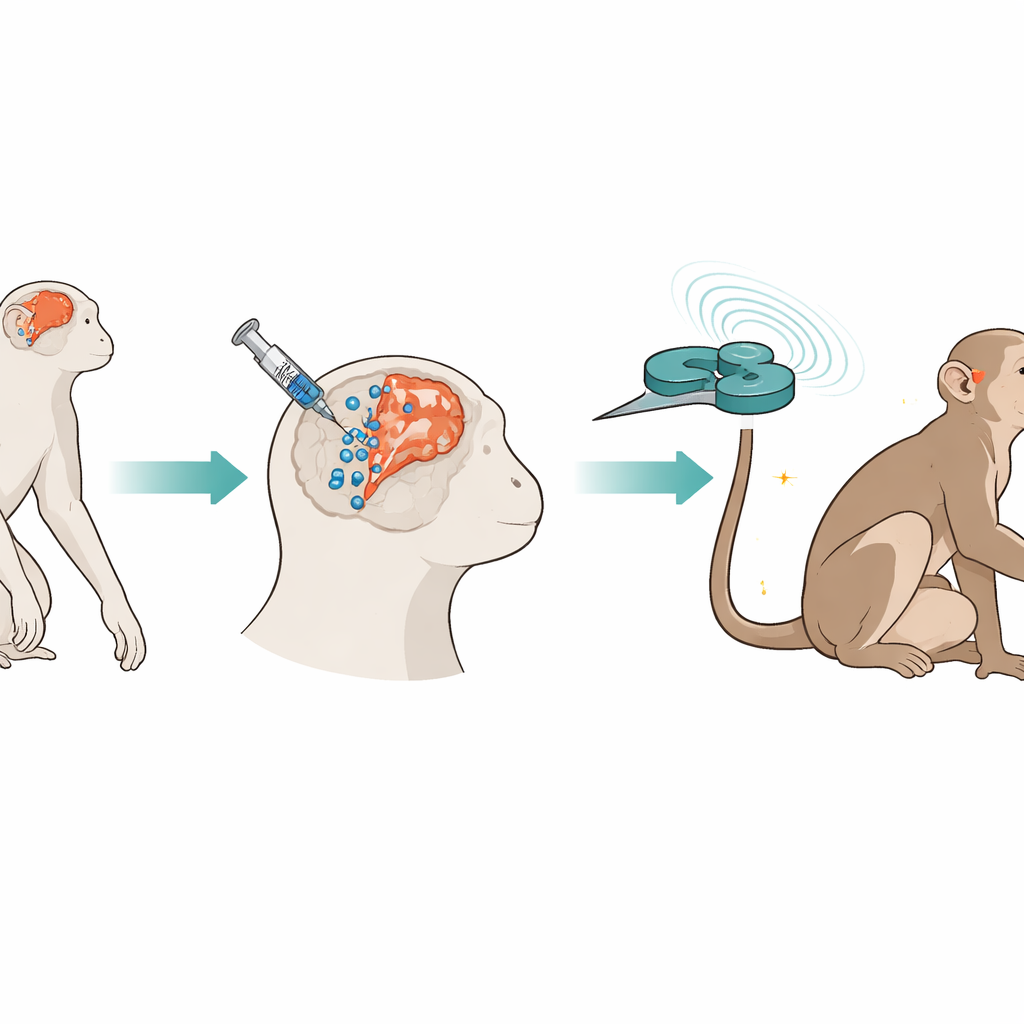
Building a Two-Step Repair Strategy
The researchers used cynomolgus monkeys, whose brains and stroke responses resemble those of humans. Two years after deliberately blocking a brain artery to create a long-lasting stroke, they stereotactically injected human umbilical cord–derived mesenchymal stem cells into several sites around the damaged area and along a nearby stem cell–rich zone. The cells were labeled with tiny iron particles and fluorescent tags so they could be tracked over time with MRI scans and under the microscope. One week later, the team began applying intermittent theta-burst stimulation, a rapid pattern of magnetic pulses delivered through a figure-eight coil held over the injured motor cortex. This stimulation was given five days a week for 17 weeks while the animals’ movement, brain activity, and blood chemistry were monitored.
Stronger Movements and Healthier Brain Signals
Before treatment, the monkeys’ affected arms had reached a performance plateau on tasks such as climbing small staircases to retrieve food or pulling treats from a tricky box. After the combined therapy, their scores improved: success rates rose, completion times dropped, and the once-weakened limbs were used more often. Electrical testing showed that less stimulation was needed to trigger muscle responses from the injured side of the brain, and signals traveled faster along motor pathways, hinting at repaired wiring. Brain scans echoed these changes. Functional MRI revealed that the damaged motor cortex became more active and better synchronized with neighboring regions, while the strength of communication between movement-related areas increased. Diffusion imaging suggested that the white matter “cables” carrying signals grew more orderly, and magnetic resonance spectroscopy showed shifts in brain chemicals consistent with healthier neurons, better membrane repair, and improved energy use.
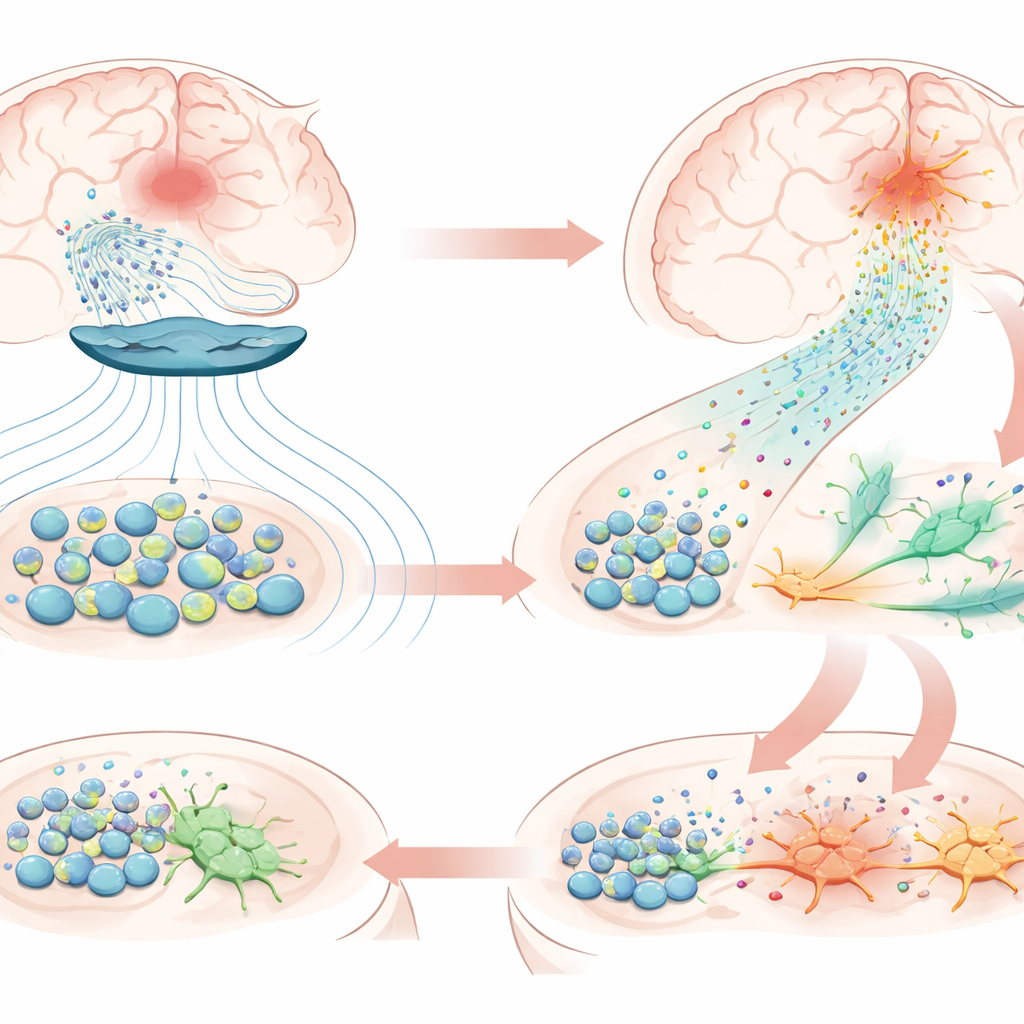
Awakening the Brain’s Own Stem Cells
When the study ended, the transplanted stem cells were still present near the stroke site, confirming unusually long survival. Detailed protein surveys of blood and brain tissue showed that the treatment boosted molecules linked to nerve growth, synapse formation, and reduced inflammation, but also prominently switched on pathways tied to neurogenesis—the birth of new neurons—and chemotaxis, the guided movement of cells. In the injured hemisphere, the team found abundant cells carrying markers of neural stem cells, which were largely absent on the uninjured side, along with early-stage neurons beginning to take on nerve-like features. A key player appeared to be the chemokine CXCL12, a signaling molecule produced by the transplanted stem cells. Its receptors were present on the brain’s own stem cells, and lab experiments showed that magnetic stimulation prompted stem cells to secrete more CXCL12 and other guiding factors. This suggests a chain reaction: magnetic pulses nudge transplanted cells to release stronger “chemical beacons,” drawing resident stem cells from nearby niches into the damaged zone, where they begin transforming into new neurons.
What This Could Mean for Future Patients
For people living with long-standing stroke disabilities, this work offers a cautiously optimistic message: even a chronically injured brain may not be beyond repair. In monkeys, the combination of stem cell grafts and patterned magnetic stimulation did more than fine-tune existing circuits—it appeared to restart the brain’s own factory for new nerve cells and guide them into rebuilding damaged areas, with measurable improvements in movement. While it will take larger studies, careful safety testing, and less invasive cell-delivery methods before this strategy can be tried widely in humans, the study lays out a clear blueprint. By pairing a biological therapy that supplies growth signals with a physical therapy that reshapes activity patterns, it may be possible to extend the window of stroke recovery well beyond what was once thought fixed.
Citation: Ma, YH., Chen, GB., Wu, MF. et al. Robust neurogenesis in chronic stroke monkeys following mesenchymal stem cell transplantation plus intermittent theta-burst stimulation. Sig Transduct Target Ther 11, 153 (2026). https://doi.org/10.1038/s41392-026-02694-5
Keywords: chronic stroke, stem cell therapy, brain stimulation, neurogenesis, nonhuman primate