Clear Sky Science · en
HERC1 oncogene enhances stemness and tumorigenic potential in CD44+-derived organoids of head and neck squamous cell carcinoma through IL-6/STAT3 signaling
Why stubborn head and neck cancers matter
Head and neck cancers are often treated with surgery, radiation, and chemotherapy, yet many tumors come back, spread, or stop responding to drugs. A big reason is a small but powerful group of cells inside the tumor that behave like stem cells: they renew themselves, seed new growth, and shrug off treatment. This study uncovers a new molecular "switch" called HERC1 that helps these dangerous cells thrive, and shows how signals from nearby support cells in the tumor’s neighborhood make matters worse. Understanding this switch could point to smarter ways to stop relapses and improve survival.
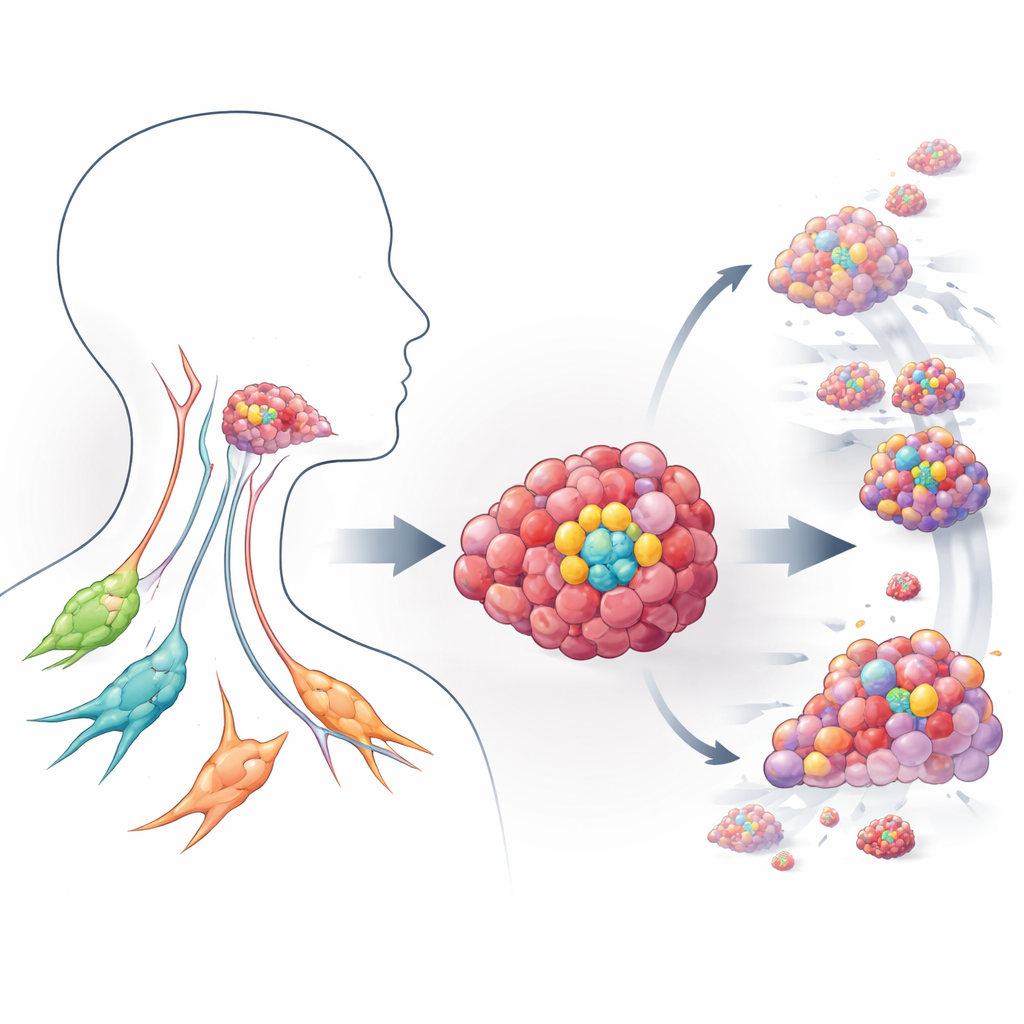
A closer look at dangerous tumor starter cells
The researchers focused on head and neck squamous cell carcinoma, one of the most common and difficult-to-treat cancers worldwide. Within these tumors, a subset of cells marked by a surface protein called CD44 behaves like stem cells: they can both self-renew and give rise to many other tumor cells, driving growth, spread, and resistance to therapy. Using patient-relevant three-dimensional cultures known as organoids, grown from CD44-positive tumor cells, the team built miniature versions of head and neck cancers in the lab. These organoids mimic real tumors far better than flat cell layers, allowing the scientists to watch how stem-like cancer cells interact with their surroundings and respond to drugs.
Introducing HERC1, a hidden helper of cancer stem cells
HERC1 is a large protein that helps tag other proteins for destruction inside cells and has been linked to brain development and genome stability, but its role in cancer was poorly understood. By mining large cancer genome databases, the team found that tumors with high levels of both HERC1 and CD44 were more common in advanced head and neck cancers and were linked to worse patient survival. In lab-grown CD44-positive spheroids and organoids, shutting down HERC1 with genetic tools sharply reduced their ability to form new spheres and organoids, and lowered key stemness markers such as Sox2 and other self-renewal factors. Even cells that originally lacked CD44 became more stem-like and formed more spheroids when HERC1 was artificially boosted, suggesting that HERC1 helps create and maintain this aggressive cell population.
How the tumor neighborhood fuels invasion and spread
Tumors do not grow in isolation. They are surrounded by support cells, including fibroblasts, which can be reprogrammed into cancer-associated fibroblasts that actively aid the tumor. The study shows that these fibroblasts release the inflammatory messenger IL-6, which switches on a signaling protein called STAT3 inside CD44-positive cancer cells. This, in turn, raises HERC1 activity and triggers a downstream pathway involving ERK, a well-known driver of cell growth and movement. Together, this IL-6–STAT3–HERC1–ERK chain boosts the stem-like traits of the cancer cells, encourages them to undergo a shape-change process associated with invasion, and helps them migrate and seed new tumors in the lungs in mouse models. When HERC1 was blocked, tumors in animals had smoother, less invasive edges and produced far fewer metastatic spots in distant organs.
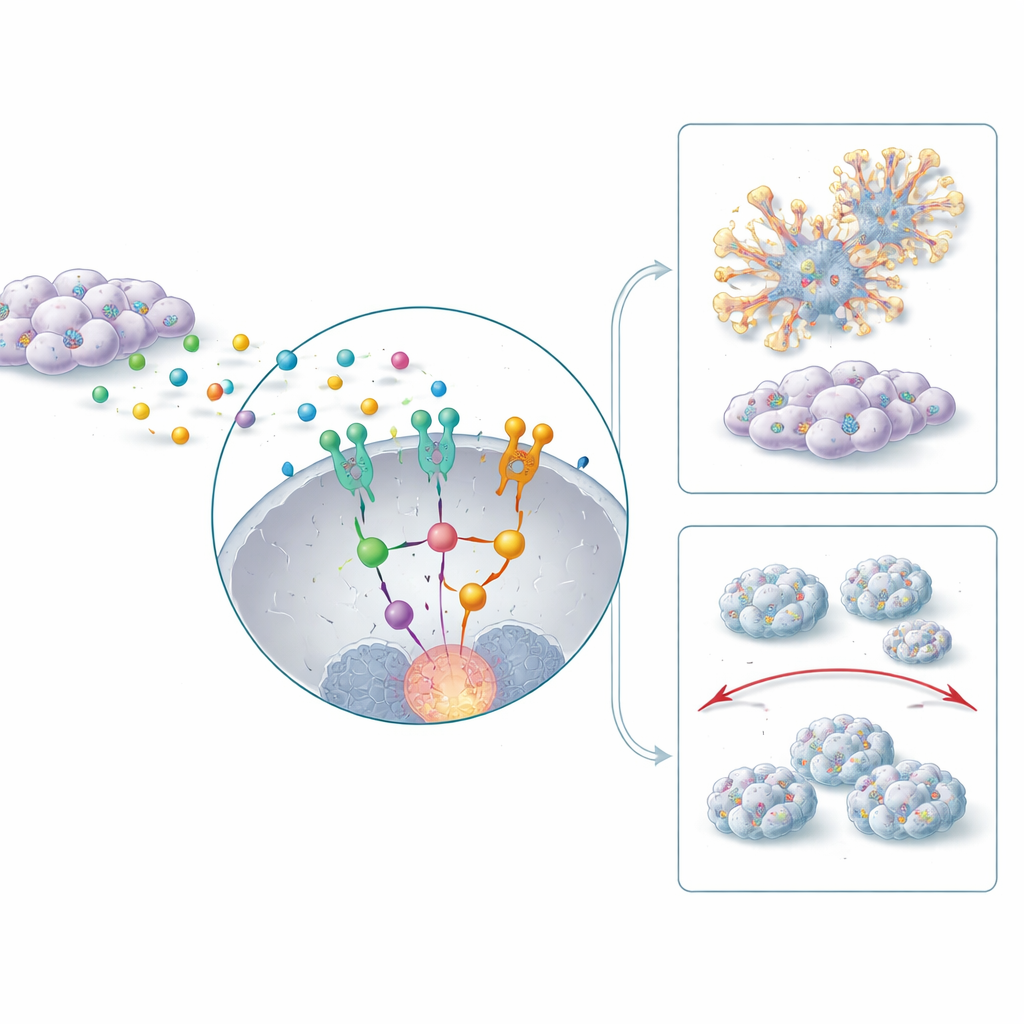
Why some tumors resist chemotherapy
Standard drugs such as cisplatin and 5-fluorouracil are mainstays of treatment for head and neck cancer, but CD44-positive organoids proved unusually hard to kill with these agents. The researchers discovered that high HERC1 levels are tied to an anti-death program inside the cells, involving molecules like Bcl-2 that help cells survive stress. When HERC1 was knocked down, organoids became much more sensitive to chemotherapy, showing increased activation of cell-death enzymes and losing their structural integrity. Blocking IL-6 or STAT3, which lie upstream of HERC1, further enhanced the killing effect of chemotherapy. In mice, combining HERC1 inhibition with 5-fluorouracil shrank tumors by more than three-quarters, far more than either approach alone.
What this means for future treatments
By linking inflammatory signals from the tumor’s neighborhood to a molecular switch inside cancer stem-like cells, this work identifies HERC1 as a central player in tumor growth, invasion, and drug resistance in head and neck cancer. In simple terms, fibroblasts around the tumor send IL-6 signals that flip on STAT3, which then boosts HERC1 and activates growth and survival pathways in CD44-positive cells. These cells become better at spreading and harder to kill. The findings suggest that drugs aimed at HERC1 itself, or at the IL-6/STAT3/HERC1 chain, could weaken these stubborn cells, make standard chemotherapies more effective, and ultimately reduce relapse and metastasis in patients with aggressive head and neck cancers.
Citation: Jeong, E., Kim, H.L., Park, S. et al. HERC1 oncogene enhances stemness and tumorigenic potential in CD44+-derived organoids of head and neck squamous cell carcinoma through IL-6/STAT3 signaling. Oncogene 45, 1840–1855 (2026). https://doi.org/10.1038/s41388-026-03725-9
Keywords: head and neck cancer, cancer stem cells, tumor microenvironment, chemoresistance, IL-6 STAT3 signaling