Clear Sky Science · en
NIR-II-triggered plasmonic catalysis with tip-localized enhancement: a strategy for hypoxic biofilm eradication on orthopedic implants
Why stubborn implant infections matter
When people receive metal screws, plates, or joint replacements, invisible colonies of bacteria can quietly build slimy fortresses, called biofilms, on the surface. These biofilms shrug off antibiotics and heat, often forcing patients to endure repeat surgeries and long hospital stays. This study describes a new kind of smart coating for titanium bone implants that uses deep-penetrating near‑infrared light to both dismantle these bacterial strongholds and encourage surrounding bone to grow, aiming to make implants safer and longer‑lasting.
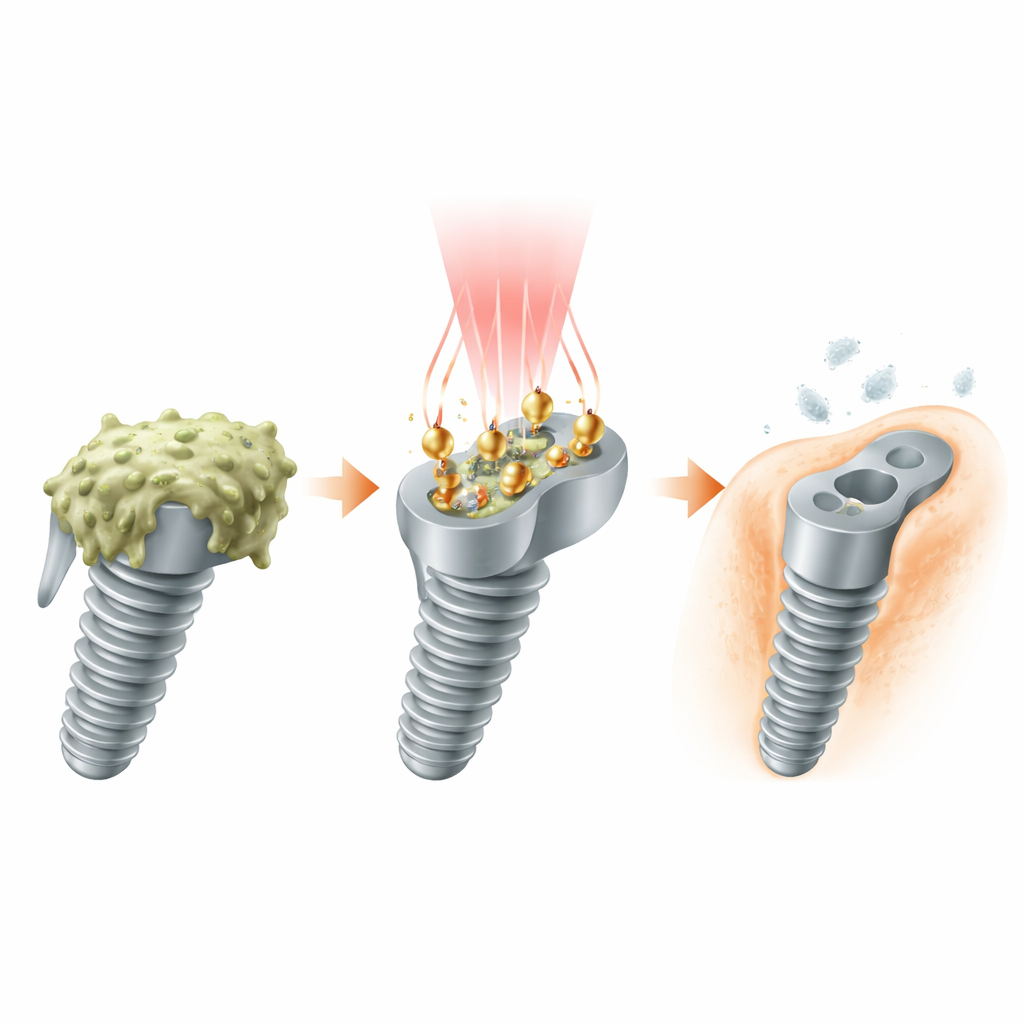
A light-powered cleanup crew
The researchers engineered tiny “worker” particles made of gold and platinum that sit on the implant surface. Each worker is shaped like a gold double-ended pyramid with a sprinkling of platinum dots at its tips. This design is not just aesthetic: it tunes the particles to respond to a special window of light, called NIR‑II, which can pass through body tissues more efficiently than visible light. When a NIR‑II laser shines on the coated implant, the gold–platinum structures soak up the light and convert it into both heat and energetic electrons, turning the surface into a light-activated cleaning field that targets bacteria where they hide.
From light to microscopic “scissors”
Under the right conditions, the energized particles act like artificial enzymes. Working together with a small amount of hydrogen peroxide, they generate highly reactive hydroxyl radicals—short‑lived chemical “scissors” that attack the molecules holding biofilms together. The platinum-tipped gold shape helps separate and guide the hot charges created by light, preventing them from canceling each other out and making the reaction much more efficient. As a result, the implant surface heats moderately while continuously producing radicals that slice apart the bacteria’s protective mesh and weaken their membranes, even in low‑oxygen environments where many other light-based treatments struggle.
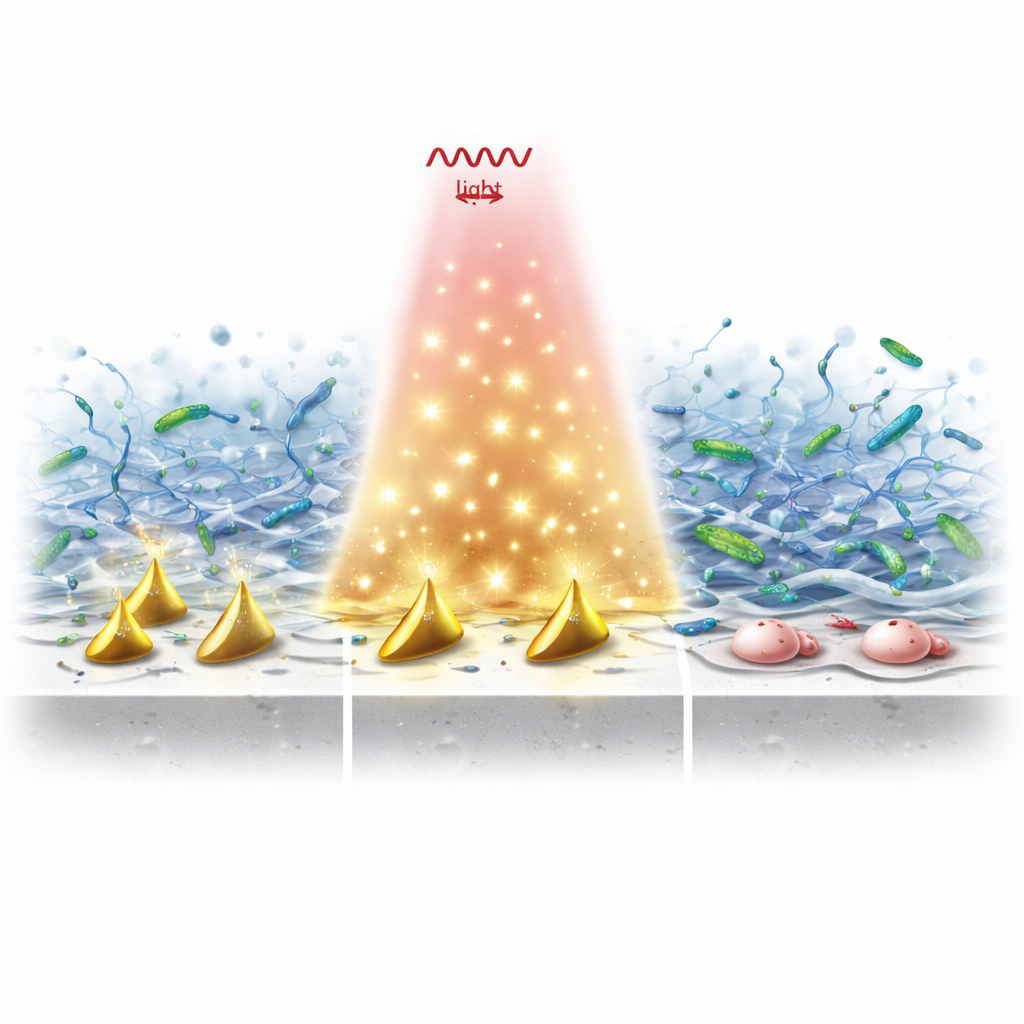
Breaking the slime shield and killing the bacteria
In lab tests, implants coated with these particles and then exposed to NIR‑II light showed a dramatic drop in bacterial survival compared with plain titanium. The dual action of warmth and radicals did more than kill free‑floating microbes: it disrupted mature biofilms by cutting their extracellular DNA, punching holes in bacterial walls, leaking out vital proteins, and overwhelming their internal antioxidant defenses. Even when oxygen was scarce—a typical condition around long‑standing infections—the system continued to churn out enough reactive species to destroy both the biofilm scaffold and the bacteria protected inside it.
Helping bone grow back, not just killing germs
Because an implant must bond firmly with bone to succeed, the team added a short peptide, called RGDC, to the nanoparticle layer. This peptide mimics natural adhesion signals in the body, giving bone‑forming cells something to grip. In cell culture, more bone precursor cells attached, spread, and multiplied on the modified surfaces than on bare titanium. Over time, these cells switched on genes associated with building bone matrix and mineral deposits. In rats with infected bone defects, the coated implants not only suppressed infection and inflammation but also showed more new bone forming snugly around the metal, indicating better integration with the skeleton.
Toward smarter, safer bone implants
Overall, the study demonstrates a strategy in which a surgeon’s metal implant can double as a controllable treatment device: a brief dose of invisible light from outside the body activates the coating to chemically and thermally dismantle biofilms while staying gentle enough for surrounding tissues. At the same time, the surface chemistry encourages bone cells to colonize and rebuild damaged areas. For patients, such light‑responsive, bacteria‑fighting, bone‑friendly coatings could one day reduce the need for repeat surgeries and long antibiotic courses, bringing us closer to implants that defend themselves while helping the body heal.
Citation: Sun, Y., Sheng, F., Liang, Y. et al. NIR-II-triggered plasmonic catalysis with tip-localized enhancement: a strategy for hypoxic biofilm eradication on orthopedic implants. Light Sci Appl 15, 204 (2026). https://doi.org/10.1038/s41377-026-02279-5
Keywords: orthopedic implants, bacterial biofilms, near infrared light, plasmonic nanozymes, bone regeneration